Abstract
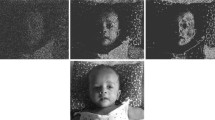
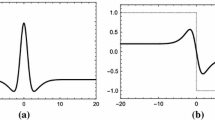
Traditionally the intensity discontinuities in an image are detected as zero-crossings of the second derivative with the help of a Laplacian of Gaussian (LOG) operator that models the receptive field of retinal Ganglion cells. Such zero-crossings supposedly form a raw primal sketch edge map of the external world in the primary visual cortex of the brain. Based on a new operator which is a linear combination of the LOG and a Dirac-delta function that models the extra-classical receptive field of the ganglion cells, we find that zero-crossing points thus generated, store in presence of noise, apart from the edge information, the shading information of the image in the form of density variation of these points. We have also shown that an optimal image contrast produces best mapping of the shading information to such zero-crossing density variation for a given amount of noise contamination. Furthermore, we have observed that an optimal amount of noise contamination reproduces the minimum optimal contrast and hence gives rise to the best representation of the original image. We show that this phenomenon is similar in nature to that of stochastic resonance phenomenon observed in psychophysical experiments.
Similar content being viewed by others
References
Benzi R, Parisi G, Sutera A, Vulpiani A (1982) Stochastic resonance in climatic change. Tellus 34: 10–16
Canny J (1986) A computational approach to edge detection. IEEE Trans. PAMI 8: 679–698
Cordo P, Inglis JT, Verschueren S, Collins JJ, Merfeld DM, Rosenblum S, Buckley S, Moss F (1996) Noise in human muscle spindles. Nature (London) 383: 769–770
Douglass JK, Wilkens L, Pantazelou E, Moss F (1993) Noise enhancement of information transfer in crayfish mechanoreceptors by stochastic resonance. Nature (London) 365: 337–340
Freund JA (2000) Stochastic Resonance with images and spatially correlated stochastic patterns. In: Freund JA, Poeschel T (eds) Stochastic processes in physics, chemistry and biology. Springer, Berlin, pp 160–171
Ghosh K, Sarkar S, Bhaumik K (2005) A possible mechanism of zero-crossing detection using the concept of extended classical receptive field of retinal ganglion cells. Biol Cybern 93: 1–5
Ghosh K, Sarkar S, Bhaumik K (2006) A possible explanation of the low-level brightness-contrast illusions in the light of an extended classical receptive field model of retinal ganglion cells. Biol Cybern 94: 89–96
Ghosh K, Sarkar S, Bhaumik K (2007) Understanding image structure from a new multi-scale representation of higher order derivative filters. Image Vis Comput 25: 1228–1238
Hochstein S, Spitzer H (1984) Zero crossing detectors in primary visual cortex?. Biol Cybern 51: 195–199
Ikeda H, Wright MJ (1972) Functional organization of the periphery effect in retinal ganglion cells. Vis Res 12: 1857–1879
Kuffler SW (1953) Discharge patterns and functional organizations of mammalian retina. J Neurophysiol 16: 37–68
Kaplan E, Benardete E (2001) The dynamics of primate retinal ganglion cells. Prog Brain Res 134: 1–18
Kitajo K, Nozaki D, Ward LM, Yamamoto Y (2003) Behavioral stochastic resonance within human brain. Phys Rev Lett 90: 218103–218106
Levin JE, Miller JP (1996) Broadband neural encoding in the cricket cereal sensory system enhanced by stochastic resonance. Nature (London) 380: 165–168
Ma SD, Li B (1998) Derivative computation by multiscale filters. Image Vis Comput 16: 43–53
Marr D, Hildreth E (1980) Theory of edge detection. Proc. R. Soc. Lond. B 207: 187–217
Marr D, Ullman S (1981) Directional selectivity and its use in early visual processing. Proc. R. Soc. Lond. B 211: 151–180
McIlwain JT (1966) Some evidence concerning the periphery effect in cat’s retina. Exp Brain Res 1: 265–271
Nicolis C (1982) Stochastic aspects of climatic transitions—response to a periodic forcing. Tellus 34: 1–9
Passaglia CL, Enroth-Cugell C, Troy JB (2001) Effects of remote stimulation on the mean firing rate of cat retinal ganglion cells. J Neurosci 21: 5794–5803
Robinson HPC (2004) The biophysical basis of firing variability in cortical neurons. In: Feng JF (eds) Computational neuroscience: a comprehensive approach. Chapman & Hall/CRC, London, pp 159–183
Richter J, Ullman S (1986) Non-linearities in cortical simple cell and the possible detection of zero crossings. Biol Cybern 53: 195–202
Rodieck RW, Stone J (1965) Analysis of receptive fields of cat retinal ganglion cells. J Neurophysiol 28: 833–849
Stocks NG (2000) Suprathreshold stochastic resonance in multilevel threshold systems. Phys Rev Lett 84: 2310–2313
Shalden MN, Newsome WT (1994) Noise, neural codes and cortical organization. Curr Opin Neurobiol 4: 569–579
Simonotto E, Riani M, Seife C, Roberts M, Twitty J, Moss F (1997) Visual perception of stochastic resonance. Phys Rev Lett 78: 1186–1189
Tham C (2007) Stochastic resonances in vision: models and data. Ph.D. Thesis, The University of Manchester, MIMS Eprint 2007.7 (2007). http://www.manchester.ac.uk/mims/eprints
Usui S, Stark L (1978) Sensory and motor mechanism interact to control amplitude of pupil noise. Vis Res 18: 505–507
Wiesel TN (1960) Receptive fields of ganglion cells in cat’s retina. J Physiol (Lond) 153: 583–594
Wiesenfeld K, Moss F (1995) Stochastic resonance and the benefits of noise: from ice ages to crayfish and SQUIDs. Nature (London) 373: 33–36
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ghosh, K., Sarkar, S. & Bhaumik, K. A possible mechanism of stochastic resonance in the light of an extra-classical receptive field model of retinal ganglion cells. Biol Cybern 100, 351–359 (2009). https://doi.org/10.1007/s00422-009-0306-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00422-009-0306-9