Abstract
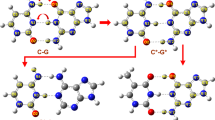
The substituent effects on the geometrical parameters and the individual hydrogen bond (HB) energies of base pairs such as X–adenine–thymine (X–A–T), X–thymine–adenine (X–T–A), X–guanine–cytosine (X–G–C), and X–cytosine–guanine (X–C–G) have been studied by the quantum mechanical calculations at the B3LYP and MP2 levels with the 6–311++G(d,p) basis set. The electron withdrawing (EW) substituents (F and NO2) increase the total binding energy (ΔE) of X–G–C derivatives and the electron donating (ED) substituent (CH3) decreases it when they are introduced in the 8 and 9 positions of G. The effects of substituents are reversed when they are located in the 1, 5, and 6 positions of C, with exception of CH3 in the 1 position and F in the 5 position, which in both cases the ΔE value decreases negligibly small. With minor exceptions (X=8–CH3, 8–F, and 9–NO2), both ED and EW substituents increase slightly the ΔE values of X–A–T derivatives. The individual HB energies (∆E HBs) have been estimated using electron densities that calculated at the hydrogen bond critical points (HBCPs) by the atoms in molecules (AIM) method. Most of changes of individual HBs are in consistent with the ED/EW nature of substituents and the role of atoms entered H-bonding. The remarkable change is observed for NO2 substituted derivative in each case.



Similar content being viewed by others
References
Xue C, Popelier PLA (2008) Computational study of substituent effects on the interaction energies of hydrogen–bonded Watson–Crick cytosine–guanine base pairs. J Phys Chem B 112:5257–5264
Watson JD, Crick FHC (1953) A structure for deoxyribose nucleic acid. Nature 171:737–738
Brenner S, Jacob F, Meselson M (1961) An unstable intermediate carrying information from genes to ribosomes for protein synthesis. Nature 190:576–581
Guerra CF, Bickelhaupt FM, Snijders JG, Baerends EJ (1999) The nature of the hydrogen bond in DNA base pairs: the role of charge transfer and resonance assistance. Chem A Eur J 5:3581–3594
Hobza P, Sponer J (1999) Structure, energetics, and dynamics of the nucleic acid base pairs: nonempirical ab initio calculations. Chem Rev 99:3247–3276
Hobza P, Zahradnik R, Mueller–Dethlefs K (2006) The world of non–covalent interactions. Chem Commun 71:443–531
Shishkin OV, Sponer J, Hobza P (1999) Intramolecular flexibility of DNA bases in adenine–thymine and guanine–cytosine Watson–Crick base pairs. J Mol Struct 477:15–21
Meng F, Liu C, Xu W (2003) Substituent effects of R (R=CH3, CH3O, F and NO2) on the A–T and G–C base pairs: a theoretical study. Chem Phys Lett 373:72–78
Kawahara SI, Uchimaru T, Taira K, Sekine M (2002) An ab initio study of the hydrogen bond energy of base pairs formed between substituted 9–methylguanine derivatives and 1–methylcytosine. J Phys Chem A 106:3207–3212
Kawahara SI, Kobori A, Sekine M, Uchimaru T (2001) Theoretical study of the hydrogen bond energy of base pairs formed between substituted 1–methylcytosine derivatives and 9–methylguanine. J Phys Chem A 105:10596–10601
Kawahara SI, Wada T, Kawauchi S, Uchimaru T, Sekine M (1999) Ab initio and density functional studies of substituent effects of an A–U base pair on the stability of hydrogen bonding. J Phys Chem A 103:8516–8523
Kawahara SI, Uchimaru T, Taira K, Sekine M (2001) Ab initio evaluation of the substitution effect of the hydrogen bond energy of the Watson–Crick type base pair between 1–methyluracil and substituted 9–methyladenine derivatives. J Phys Chem A 105:3894–3898
Xue CX, Popelier PLA (2009) Prediction of interaction energies of substituted hydrogen–bonded Watson–Crick cytosine–guanine (8X) base pairs. J Phys Chem B 113:3245–3250
Moser A, Guza R, Tretyakova N (2009) Density functional study of the influence of C5 cytosine substitution in base pairs with guanine. Theor Chem Account 122:179–188
Burda JV, Sponer J, Hobza P (1996) Ab initio study of the interaction of guanine and adenine with various mono–and bivalent metal cations (Li+, Na+, K+, Cs+; Cu+, Ag+, Au+; Mg2+, Ca2+, Sr2+, Ba2+; Zn2+, Cd2+, and Hg2+). J Phys Chem 100:7250–7255
Fonseca Guerra C, Bickelhaupt FM, Snijders JG, Baerends EJ (2000) Hydrogen bonding in DNA base pairs: reconciliation of theory and experiment. J Am Chem Soc 122:4117–4128
Russo N, Toscano M, Grand A (2001) Bond energies and attachments sites of sodium and potassium cations to DNA and RNA nucleic acid bases in the gas phase. J Am Chem Soc 123:10272–10279
Petrov AS, Lamm G, Pack GR (2002) Water–mediated magnesium–guanine interactions. J Phys Chem B 106:3294–3300
Cerda BA, Wesdemiotis C (1996) Li+, Na+, and K+ binding to the DNA and RNA nucleobases bond energies and attachment sites from the dissociation of metal ion–bound heterodimers. J Am Chem Soc 118:11884–11892
Munoz J, Sponer J, Hobza P, Orozco M, Luque FJ (2001) Interactions of hydrated Mg2+ cation with bases, base pairs, and nucleotides electron topology, natural bond orbital, electrostatic, and vibrational study. J Phys Chem B 105:6051–6060
Gresh N (1999) Complexes of pentahydrated Zn2+ with guanine, adenine, and the guanine–cytosine and adenine–thymine base pairs structures and energies characterized by polarizable molecular mechanics and ab initio calculations. J Phys Chem B 103:11415–11427
Sponer J, Sabat M, Gorb L, Leszczynski J, Lippert B, Hobza P (2000) The effect of metal binding to the N7 Site of purine nucleotides on their structure, energy, and involvement in base pairing. J Phys Chem B 104:7535–7544
Gadre SR, Pundlik SS, Limaye AC, Rendell AP (1998) Electrostatic investigation of metal cation binding to DNA bases and base pairs. Chem Commun 5:573–574
Sun L, Bu Y (2005) Marked variations of dissociation energy and H–bond character of the guanine–cytosine base pair induced by one–electron oxidation and Li+ cation coupling. J Phys Chem B 109:593–600
Destexhe A, Smets J, Adamowicz L, Maes G (1994) Matrix isolation FT–IR studies and ab initio calculations of hydrogen–bonded complexes of molecules modeling cytosine or isocytosine tautomers. 1. pyridine and pyrimidine complexes with water in argon matrixes. J Phys Chem 98:1506–1514
Gould IR, Kollman PA (1994) Theoretical investigation of the hydrogen bond strengths in guanine–cytosine and adenine–thymine base pairs. J Am Chem Soc 116:2493–2499
Hobza P, Sponer J, Polasek M (1995) H–bonded and stacked DNA base pairs: cytosine dimer An ab initio second–order moller–plesset study. J Am Chem Soc 117:792–798
Sponer J, Hobza P (1996) Nonempirical ab initio calculations on DNA base pairs. Chem Phys 204:365–372
Sponer J, Hobza P (1996) Thermodynamic characteristics for the formation of H–bonded DNA base pairs. Chem Phys Lett 261:379–384
Sponer J, Leszczynski J, Hobza P (1996) Nature of nucleic acid–base stacking: nonempirical ab initio and empirical potential characterization of 10 stacked base dimers comparison of stacked and H–bonded base pairs. J Phys Chem 100:5590–5596
Sponer J, Hobza P (2000) Interaction energies of hydrogen–bonded formamide dimer, formamidine dimer, and selected DNA base pairs obtained with large basis sets of atomic orbitals. J Phys Chem A 104:4592–4597
Asensio A, Kobko N, Dannenberg JJ (2003) Cooperative hydrogen–bonding in adenine–thymine and guanine–cytosine base pairs density functional theory and moller–plesset molecular orbital study. J Phys Chem A 107:6441–6443
Grunenberg J (2004) Direct assessment of interresidue forces in Watson–Crick base pairs using theoretical compliance constants. J Am Chem Soc 126:16310–16311
Dong H, Hua W, Li S (2007) Estimation on the individual hydrogen–bond strength in molecules with multiple hydrogen bonds. J Phys Chem A 111:2941–2945
Ebrahimi A, Habibi khorasani SM, Delarami H (2009) Estimation of individual binding energies in some dimers involving multiple hydrogen bonds using topological properties of electron charge density. Chem Phys 365:18–23
Meng F, Wang H, Xu W, Liu C, Wang H, Xu W, Liu C (2005) Theoretical study of GC+/GC base pair derivatives. Chem Phys 308:117–123
Bader RFW (1990) Atoms in molecules: a quantum theory. Oxford University Press, Oxford
Backe AD (1993) Density–functional thermochemistry. III. The role of exact exchange. J Chem Phys 98:5648–5652
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Montgomery JA Jr, Vreven T, Kudin KN, Burant JC, Millam JM, Iyengar SS, Tomasi J, Barone V, Mennucci B, Cossi M, Scalmani G, Rega N, Petersson GA, Nakatsuji H, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Klene M, Li X, Knox JE, Hratchian HP, Cross JB, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Ayala PY, Morokuma K, Voth GA, Salvador P, Dannenberg JJ, Zakrzewski VG, Dapprich S, Daniels AD, Strain MC, Farkas O, Malick DK, Rabuck AD, Raghavachari K, Foresman JB, Ortiz JV, Cui Q, Baboul AG, Clifford S, Cioslowski J, Stefanov BB, Liu G, Liashenko A, Piskorz P, Komaromi I, Martin RL, Fox DJ, Keith T, Al-Laham MA, Peng CY, Nanayakkara A, Challacombe M, Gill PMW, Johnson B, Chen W, Wong MW, Gonzalez C, Pople JA (2003) Gaussian03 (Revision B03). Gaussian, Inc, Pitts burgh, PA
Boys SB, Bernardi F (1970) The calculations of small molecular interaction by the difference of separate total energies some procedures with reduce error. Mol Phys 19:553–566
Biegler Konig FW, Schonbohm J, Bayles D (2001) Software news and updates AIM2000. J Comput Chem 22:545–559
Reed AE, Curtiss LA, Weinhold F (1988) Intramolecular interactions from a natural bond orbital, donor–acceptor viewpoint. Chem Rev 88:899–936
Glendening D, Reed AE, Carpenter JE, Weinhold F, NBO version 3.1
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Ebrahimi, A., Habibi Khorassani, S.M., Delarami, H. et al. The effect of CH3, F and NO2 substituents on the individual hydrogen bond energies in the adenine–thymine and guanine–cytosine base pairs. J Comput Aided Mol Des 24, 409–416 (2010). https://doi.org/10.1007/s10822-010-9348-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-010-9348-2