Abstract
Many neuronal systems exhibit slow random alternations and sudden switches in activity states. Models with noisy relaxation dynamics (oscillatory, excitable or bistable) account for these temporal, slow wave, patterns and the fluctuations within states. The noise-induced transitions in a relaxation dynamics are analogous to escape by a particle in a slowly changing double-well potential. In this formalism, we obtain semi-analytically the first and second order statistical properties: the distributions of the slow process at the transitions and the temporal correlations of successive switching events. We find that the temporal correlations can be used to help distinguish among biophysical mechanisms for the slow negative feedback, such as divisive or subtractive. We develop our results in the context of models for cellular pacemaker neurons; they also apply to mean-field models for spontaneously active networks with slow wave dynamics.







Similar content being viewed by others
References
Baltanas, J. P., & Casado, J. M. (1998). Bursting behavior of the FitzHugh-Nagumo neuron model subject to quasi-monochromatic noise. Physica D, 122, 231–240.
Box, G. E. P., & Muller, M. E. (1958). A note on the generation of random normal deviates. The Annals of Mathematical Statistics, 29(2), 610–611.
Butera, R. J., Rinzel, J., & Smith, J. C. (1999). Models of respiratory rhythm generation in the pre-Bötzinger complex. I. Bursting pacemaker neurons. Journal of Neurophysiology, 81, 382–397.
Chow, C. C., & White, J. (1996). Spontaneous action potentials due to channel fluctuations. Biophysical Journal, 71, 3013–3021.
Cox, D. R., & Lewis, P. A. W. (1966). The statistical analysis of series of events. London: Methuen.
DeVille, R. E. L., Muratov, C., & Vanden-Eijnden, E. (2005). Two distinct mechanisms of coherence in randomly perturbed dynamical systems. Physical Review E, 72, 031105.
DeVille, R. E. L., Muratov, C., & Vanden-Eijnden, E. (2006). Non-meanfield deterministic limits in chemical reaction kinetics far from equilibrium. Journal of Chemical Physics, 124(23), 231102.
Elandt-Johnson, R. C., & Johnson, N. L. (1980). Survival models and data analysis. John Wiley & Sons.
Feldman, J. L., & Del Negro, C. A. (2006). Looking for inspiration: new perspectives on respiratory rhythm. Nature Reviews Neuroscience, 7, 232–242.
Freidlin, M. I. (2001). On stable oscillations and equilibriums induced by small noise. Journal of Statistical Physics, 103, 283–300.
Freidlin, M. I., & Wentzell, A. D. (1998). Random perturbations of dynamical systems (2nd ed.). New York: Springer.
Gardiner, C. W. (1985). Handbook of stochastic methods. Berlin: Springer-Verlag.
Grzywacz, N., & Sernagor, E. (2000). Spontaneous activity in developing turtle retinal ganglion cells. Visual Neuroscience, 17, 229–241.
Gutkin, B. S., & Ermentrout, G. B. (1998). Dynamics of membrane excitability determine interspike interval variability: a link between spike generation mechanisms and cortical spike train statistics. Neural Computation, 10(5), 1047–1065.
Hänggi, P., Talkner, P., & Borkovec, M. (1990). Reaction-rate theory: fifty years after Kramers. Reviews of Modern Physics, 62(2), 251–341.
Higham, D. J. (2001). An algorithmic introduction to numerical simulation of stochastic differential equations. SIAM Review, 43(3), 525–546.
Hitczenko, P., & Medvedev, G. S. (2009). Bursting oscillations induced by small noise. SIAM Journal on Applied Mathematics, 69, 1359–1392.
Koshiya, N., & Smith, J. C. (1999). Neuronal pacemaker for breathing visualized in vitro. Nature, 400, 360–363.
Kuske, R., & Baer, S. M. (2002). Asymptotic analysis of noise sensitivity in a neuronal burster. Bulletin of Mathematical Biology, 64, 447–481.
Laing, C. R., & Chow, C. C. (2002). A spiking neuron model for binocular rivalry. Journal of Computational Neuroscience, 12, 39–53.
Longtin, A., Bulsara, A., & Moss, F. (1991). Time-interval sequences in bistable systems and the noise-induced transmission of information by sensory neurons. Physical Review Letters, 67, 656–659.
Moreno-Bote, R., Rinzel, J., & Rubin, N. (2007). Noise-induced alternations in an attractor network model of perceptual bi-stability. Journal of Neurophysiology, 98(3), 1125–1139.
Muratov, C. B., Vanden-Eijnden, E., & Weinan, E. (2005). Self-induced stochastic resonance in excitable systems. Physica D, 210, 227–240.
Prinz, A. A., Abbott, L. F., & Marder, E. (2004). The dynamic clamp comes of age. Trends in Neuroscience, 27(4).
Rinzel, J. (1987). A formal classification of bursting mechanisms in excitable systems. In E. Teramoto, & M. Yamaguti (Eds.), Mathematical topics in population biology, morphogenesis, and neuroscience. Berlin: Springer-Verlag. vol 71 of Lecture notes in biomathematics: 267–281.
Rowat, P. F., & Elson, R. C. (2004). State-dependent effects of Na channel noise on neuronal burst generation. Journal of Computational Neuroscience, 16, 87–112.
Sharp, A. A., O’Neil, M. B., Abbott, L. F., & Marder, E. (1993). The dynamic clamp: artificial conductances in biological neurons. Trends in Neurosciences, 16(10), 389–394.
Shpiro, A., Curtu, R., Rinzel, J., & Rubin, N. (2007). Dynamical characteristics common to neuronal competition models. Journal of Neurophysiology, 97(1), 462–473.
Shpiro, A., Moreno-Bote, R., Rubin, N., Rinzel, J. (2009). Balance between noise and adaptation in competition models of perceptual bistability. Journal of Computational Neuroscience, 27(1), 37–54.
Staley, K. J., Longacher, M., Bains, J. S., & Yee, A. (1998). Presynaptic modulation of CA3 network activity. Nature Neuroscience, 1, 201–209.
Streit, J. (1993). Regular oscillations of synaptic activity in spinal networks in vitro. Journal of Neurophysiology, 70, 871–878.
Su, J., Rubin, J., & Terman, D. (2004). Effects of noise on elliptic bursters. Nonlinearity, 17, 133–157.
Tabak, J., Senn, W., O’Donovan, M. J., & Rinzel, J. (2000). Modeling of spontaneous activity in the developing spinal cord using activity-dependent depression in an excitatory network. Journal of Neuroscience, 20(8), 3041–3056.
Tabak, J., Rinzel, J., & O’Donovan, M. J. (2001). The role of activity-dependent network depression in the expression and self-regulation of spontaneous activity in the developing spinal cord. Journal of Neuroscience, 21(22), 8966–8978.
Tabak, J., O’Donovan, M. J., & Rinzel, J. (2006). Differential control of active and silent phases in relaxation models of neuronal rhythms. Journal of Computational Neuroscience, 21, 307–328.
Tabak, J., Mascagni, M., & Bertram, R. (2007). Spontaneous episodic activity: why episode duration is correlated with the length of preceding but not following interval. Soc Neurosci Abstr 925.7/QQ19.
White, J., Klink, R., Alonso, A., & Kay, A. R. (1998). Noise from voltage-gated ion channels may influence neuronal dynamics in the entorhinal cortex. Journal of Neurophysiology, 80, 262–269.
Acknowledgments
We thank Lee DeVille, Ruben Moreno-Bote, Jeffrey Smith and Eric Vanden-Eijnden for helpful discussions and Brent Doiron for suggesting survival analysis.
Author information
Authors and Affiliations
Corresponding author
Additional information
Action Editor: Alain Destexhe
Appendix
Appendix
1.1 Modification of the duration
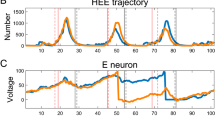
In Section 3.2, the durations of states are computed under the assumption that the system moves along the V-nullcline. However, as seen in Fig. 3(b), the trajectory during each state is widely spread along the V-nullcline due to noise. The deviation of the trajectory from the V-nullcline affects the speed of the slow variable in Eq. (3c) and thus, may lead to the discrepancy between the analysis and the simulation (Fig. 3(d)).
The statistics of this deviation can be obtained analytically by the stationary distribution of V for each h. The distribution of V at each h is the stationary solution for the Fokker-Planck equation (Gardiner 1985). When the system has a potential function, U(V,h), then the stationary solution is given as follows:
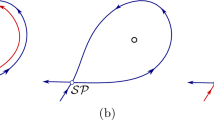
In a double-well potential, we can calculate the conditional probability and the mean given that the system stays in the left well by normalizing Eq. (10a). Here, V sep (h) is the separatrix between the left and right wells for given h.
With the modified V obtained from Eq. (10c), we can compute the duration. Figure 8 shows the mean deviation of the trajectory from the V-nullcline and comparison of the durations computed using the V-values on the V-nullcline and the modified V. The distributions of h at the entry and leaving points are obtained from the simulations so that the discrepancy between the simulation and analysis does not originate from the transition PDFs. With the modified V, there is a modest change in the distribution of durations. It agrees better with the simulation even though the distribution with the modified V still doesn’t agree with the simulation perfectly. The mean of the deviation is not enough to correct the distribution. However, we can see the influence of the deviation of V on the duration in Fig. 8(b).
Deviation of a trajectory from the slow manifold and its effect on the duration of the state. (a) Mean V of the noisy trajectory (dotted line) and the V-nullcline in the SP (solid line) are plotted in the V-h phase plane. Due to noise, the mean of the trajectory deviates from the V-nullcline. (b) Distribution of SP durations obtained from a simulation and an analysis. In an analysis, the distributions of starting and leaving locations in the SP are given as the transition PDFs in h obtained from the simulation. The solid line is computed with the V values on the V-nullcline and the dotted line is computed using the mean of the deviated V
Rights and permissions
About this article
Cite this article
Lim, S., Rinzel, J. Noise-induced transitions in slow wave neuronal dynamics. J Comput Neurosci 28, 1–17 (2010). https://doi.org/10.1007/s10827-009-0178-y
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10827-009-0178-y