Abstract
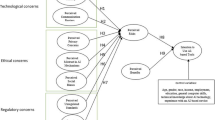
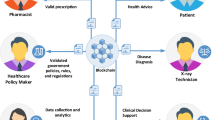
Recent ONCHIT mandates call for increased individual health data collection efforts as well as heightened security measures. To date most healthcare organizations have been reluctant to exchange information, citing confidentiality concerns and unshared costs incurred by specific organizations. Implantable monitoring and treatment devices are rapidly emerging as data collection interface tools in response to such mandates. Proposed here is a translational, device-independent consumer-based solution, which focuses on information controlled by specific patients, and functions within a distributed (organization neutral) environment. While the conceptual applications employed in this technology set are provided by way of illustration, they may also serve as a transformative model for emerging EMR/EHR requirements.





Similar content being viewed by others
References
ONC, Office of the National Coordinator, Federal Health IT Strategic Plan (ONC): 2008–2012. June 3, 2008.
Schulman, J., Dell, R., Gord, J., System of Implantable Devices For Monitoring and/or Affecting Body Parameters. US Pat. No. 6,164,284 (issued December 26, 2000).
Sakoda, K., Communication Method, Communication Device, And Computer Program. US Patent Application 20080201627 (issued August 9, 2007).
Schulman, J., Gord, J., DeRocco, P., Karr, L., Folkman, D., Barber, A., Multichannel Communication Protocol Configured to Extend the Battery Life of an Implantable Device. US Pat. No. 6,472,991 (issued October 29, 2002).
DelMain, G., Folkman, D., DeRocco, P., Karr, L., System and Method for Sharing a Common Communication Channel Between Multiple Systems of Implantable Medical Devices. US Pat. No. 7,406,105 (issued July 29, 2008).
Schulman, J., Dell, R., Gord, J. U., Battery Powered Patient Implantable Device. US Pat. No. 6,185,452 (issued February 6, 2001).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lorence, D., Lee, J. & Richards, M. ONCHIT Security in Distributed Environments: A Proposed Model for Implantable Devices. J Med Syst 34, 601–608 (2010). https://doi.org/10.1007/s10916-009-9273-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10916-009-9273-x