Abstract
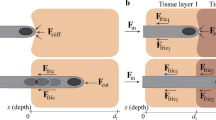
The estimation of needle deflection when the needle interacts with biological tissue is challenging in needle steering. Most previous studies have used homogeneous tissue models to estimate the deflection of a needle. However, biological tissue typically has a layered structure with variable mechanical properties and geometric features. In this study, we propose a needle deflection model with a double-layered elastic medium. Each layer possesses distinct mechanical properties, which result in inconsistent degrees of friction force on the needle surface, needle-cutting force, and forces caused by the stiffness of the elastic medium. The model uses the Rayleigh–Ritz method to analytically estimate the needle deflection. To validate the model, needle steering experiments were performed using double-layered tissue phantoms and porcine tissues. The experimental results revealed that changes in reaction force at the needle base occurred when the needle passed through the boundary between the two layers. The mean absolute error between the estimated and measured needle tip trajectory was 0.88 ± 0.30 mm for the double-layered tissue phantom and 1.85 ± 0.73 mm for the porcine tissue. These results are comparable with previous studies of homogeneous tissue. The proposed model could improve needle steering in biological tissues, which consist of multiple layers.






Similar content being viewed by others
References
Abayazid M, Roesthuis RJ, Reilink R, Misra S (2012) Integrating deflection models and image feedback for real-time flexible needle steering. Robot IEEE Trans 29(2):542–553
Asadian A, Kermani MR, Patel RV (2011) An analytical model for deflection of flexible needles during needle insertion. IEEE/RSJ international conference on intelligent robots and systems (IROS) 2551–2556
Barbe L, Bayle B, de Mathelin M, Gangi A (2007) In vivo model estimation and haptic characterization of needle insertions. Int J Robot Res 26(11–12):1283–1301
Blumenfeld P, Hata N, DiMaio S, Zou K, Haker S, Fichtinger G, Tempany C (2007) Transperineal prostate biopsy under magnetic resonance image guidance: a needle placement accuracy study. J Magn Reson Imaging 26(3):688–694
Carra A, Avila-Vilchis JC (2010) Needle insertion modeling through several tissue layers. 2nd International Asia Conference on Informatics in Control, Automation and Robotics pp 237–240
Cowan NJ, Goldberg K, Chirikjian GS, Fichtinger G, Alterovitz R, Reed KB, Kallem V, Park W, Misra S, Okamura AM (2010) Robotic needle steering: design, modeling, planning, and image guidance. Surg Robot Syst Appl Vis 557–582
DiMaio SP, Salcudean SE (2003) Needle insertion modeling and simulation. IEEE Trans Robot Autom 19(5):864–875
Egorov V, Tsyuryupa S, Kanilo S, Kogit M, Sarvazyan A (2008) Soft tissue elastometer. Med Eng Phys 30(2):206–212
Elgezua I, Kobayashi Y, Fujie MG (2013) Survey on current state-of-the-art in needle insertion robots: open challenges for application in real surgery. Procedia CIRP 5:94–99
Glozman D, Shoham M (2007) Image-guided robotic flexible needle steering. IEEE Trans Rob 23(3):459–467
Gokgol C, Basdogan C, Canadinc D (2012) Estimation of fracture toughness of liver tissue: experiments and validation. Med Eng Phys 34(7):882–891
Kataoka H, Washio T, Audette M, Mizuhara K (2001) A model for relations between needle deflection, force, and thickness on needle penetration. Med Image Comput Comput Assist Interv 966–974
Levental I, Georges PC, Janmey PA (2007) Soft biological materials and their impact on cell function. Soft Matter 3(3):299–306
Mahal AS, Knauer M, Gregory PB (1981) Bleeding after liver biopsy. West J Med 134(1):11
Mahvash M, Dupont PE (2010) Mechanics of dynamic needle insertion into a biological material. IEEE Trans Biomed Eng 57(4):934–943
Misra S, Reed K, Schafer B, Ramesh K, Okamura A (2010) Mechanics of flexible needles robotically steered through soft tissue. Int J Robot Res 29(13):1640–1660
Okamura AM, Simone C, O’Leary MD (2004) Force modeling for needle insertion into soft tissue. IEEE Trans Biomed Eng 51(10):1707–1716
Patil S, Burgner J, Webster RJ, Alterovitz R (2014) Needle steering in 3-D via rapid replanning. IEEE Transactions on Robotics, Online publish
Phee SJ, Yang K (2010) Interventional navigation systems for treatment of unresectable liver tumor. Med Biol Eng Comput 48(2):103–111
Roesthuis RJ, van Veen YRJ, Jahya A, Misra S (2011) Mechanics of needle-tissue interaction. pp 2557–2563
Shergold OA, Fleck NA, Radford D (2006) The uniaxial stress versus strain response of pig skin and silicone rubber at low and high strain rates. Int J Impact Eng 32(9):1384–1402
Shidham GB, Siddiqi N, Beres JA, Logan B, Nagaraja H, Shidham SG, Piering WF (2005) Clinical risk factors associated with bleeding after native kidney biopsy. Nephrology 10(3):305–310
Vrooijink GJ, Abayazid M, Patil S, Alterovitz R, Misra S (2014) Needle path planning and steering in a three-dimensional non-static environment using two-dimensional ultrasound images. Int J Robot Res (in proof)
Webster RJ, Kim JS, Cowan NJ, Chirikjian GS, Okamura AM (2006) Nonholonomic modeling of needle steering. Int J Robot Res 25(5–6):509–525
Wedlick TR, Okamura AM (2012) Characterization of robotic needle insertion and rotation in artificial and ex vivo tissues. Biomedical Robotics and Biomechatronics (BioRob), 4th IEEE RAS & EMBS International Conference, pp 62–68
Yan KG, Podder T, Yan Y, Tien IL, Cheng CWS, Wan Sing N (2009) Tissue interaction modeling with depth-varying mean parameter: preliminary study. Biomed Eng IEEE Trans 56(2):255–262
Acknowledgments
This work was supported by the Global Frontier R&D Program on funded by the National Research Foundation of Korea grand funded by the Korean Government (MSIP) (2012M3A6A3056424).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Lee, H., Kim, J. Estimation of flexible needle deflection in layered soft tissues with different elastic moduli. Med Biol Eng Comput 52, 729–740 (2014). https://doi.org/10.1007/s11517-014-1173-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-014-1173-7