Abstract
Objective
To obtain an accurate assessment of the percentage and depth of extra-capsular soft tissue removed with the prostate by the various surgical techniques in order to help surgeons in determining the appropriateness of different surgical approaches. This can be enhanced by an accurate and automated means of identifying the prostate gland contour.
Materials and Methods
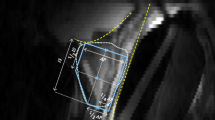
To facilitate 3D reconstruction and, ultimately, more accurate analyses, it is essential for us to identify the capsule boundary that separates the prostate gland tissue from its extra-capsular tissue. However, the capsule is sometimes unrecognizable due to the naturally occurring intrusion of muscle and connective tissue into the prostate gland. At these regions where the capsule disappears, its contour can be arbitrarily created with a continuing contour line based on the natural shape of the prostate. We utilize an algorithm based on a least squares curve fitting technique that uses a prostate shape equation to merge previously detected capsule parts with the shape equation to produce an approximated curve that represents the prostate capsule.
Results
We have tested our algorithm using three different shapes on 13 histologic prostate slices that are cut at different locations from the apex. The best result shows a 90% average contour match when compared to pathologist-drawn contours.
Conclusion
We believe that automatically identifying histologic prostate contours will lead to increased objective analyses of surgical margins and extracapsular spread of cancer. Our results show that this is achievable.
Similar content being viewed by others
References
Aarnik RG, Giesen RJB, Huynen AL, De La Rosete JJMCH, Debruyne FMJ and Wijkstra H (1994). A practical clinical method for contour determination in ultrasonographic prostate images. Ultrasound Med Biol 20(7): 705–717
Betrounia N, Vermandela M, Pasquierc D, Maoucheb S and Rousseaua J (2005). Segmentation of abdominal ultrasound images of the prostate using a priori information and an adapted noise filter. Comput Med Imaging Graph 29: 43–51
Chiu B, Freeman GH, Salama MMA and Fenster A (2004). Prostate segmentation algorithm using dyadic wavelet transform and discrete dynamic contour. Phys Med Biol 49: 4943–4960
Diamond J, Anderson HN, Bartels PH, Montironi R and Hamilton PW (2004). The use of morphological characteristics and texture analysis in the identification of tissue composition in prostatic neoplasia. Human Pathol 35(9): 1121–1131
Gong L, Pathak SD, Haynor DR, Cho PS and Kim Y (2004). Parametric Shape Modeling Using Deformable Superellipses for Prostate Segmentation. IEEE Trans Med Imaging 23: 340–349
Hamilton PW, Bartels PH and Thompson D (1997). Automated location of dysplastic fields in colorectal histology using image texture analysis. J Pathol 182: 68–75
Hussein R, McKenzie F and Joshi R (2004). Automating prostate capsule contour estimation for 3D model reconstruction using shape and histological features. SPIE Int Soc Opt Eng Proc SPIE 5367(1): 790–798
Kass M, Witkin A and Terzopoulos D (1987). Snakes: active contour models. Int J Comput Vis 1: 321–331
Knoll C, Alcaniz M, Grau V, Monserrat C and Juan MC (1999). Outlining of the prostate using snakes with shape restrictions based on the wavelet transform. Pattern Recognit 32(10): 1767–1781
Knoll C, Alcaniz M, Monserrat C, Grau V and Juan MC (1999). Multiresolution segmentation of medical images using shape-restricted snakes. Proc SPIE 3661: 222–233
Ladak HM, Mao F, Wang Y, Downey DB, Steinman DA and Fenster A (2000). Prostate segmentation from 2D ultrasound images. Med Phys 27: 1777–1788
Liu YJ, Ng WS, Teo MY and Lim HC (1997). Computerised prostate boundary estimation of ultrasound images using radial bas-relief method. Med Biol Eng Comput 35(5): 445–454
Lorenz CH and Ermert H (1997). Segmentation of ultrasonic prostate images using a probabilistic model based on markov random processes. Ultrason Imaging 19: 44–45
McKenzie FD, Hussein R, Seevinck J, Schellhammer P, Diaz J (2003) Prostate gland and extra-capsular tissue 3D reconstruction and measurement. In: The 3rd IEEE symposium on bioinformatics and bioengineering (BIBE), Bethesda, pp 246–250, March 10–12
McNeal JE, Villers AA, Redwine EA, Freiha FS and Stamey TA (1990). Capsular penetration in prostate cancer: significance for natural history and treatment. Am J Surg Pathol 14: 240–247
Nedzved P (2000). Morphological segmentation of histology cell images. IEEE ICPR’00 1: 1500
Pathak SD, Aarnink RG, Chalana V, Wijkstra H, Debruyne FMJ, Kim Y and Rosette JJ (1998). Quantitative three-dimensional transrectal ultrasound for prostate imaging. Proc SPIE 3335: 83–92
Pathak SD, Haynor DR and Kim Y (2000). Edge-guided boundary delineation in prostate ultrasound images. IEEE Trans Med Imaging 19(12): 1211–1219
Petushi S, Katsinis C, Coward C, Garcia F, Tozeren A (2004) Automated identification of microstructures on histology slides. In: 2nd IEEE international symposium on biomedical imaging: macro to nano (IEEE Cat No. 04EX821). IEEE Piscataway, vol 1, pp 424–471
Pilu M, Fitzgibbon AW, Fisher RB (1996) Ellipse-specific direct least-square fitting. In: Proceedings international conference on image processing (Cat. No.96CH35919). IEEE New York, vol 3, pp 599–602
Prater JS and Richard WD (1992). Segmenting ultrasound images of the prostate using neural networks. Ultrason Imaging 14: 159–185
Richard WD and Keen CG (1996). Automated texture-based segmentation of ultrasound images of the prostate. Comput Med Imaging Graph 20(3): 131–140
Sattar AA, Noël J, Vanderhaeghen J, Schulman C and Wespes E (1995). Prostate capsule: computerized morphometric analysis of its components. Urology 46(2): 178–181
Zhu Y, Williams S and Zwiggelaar R (2006). Computer technology in detection and staging of prostate carcinoma: a review. Med Image Anal 10(2): 178–199
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Hussein, R., McKenzie, F.D. Identifying ambiguous prostate gland contours from histology using capsule shape information and least squares curve fitting. Int J CARS 2, 143–150 (2007). https://doi.org/10.1007/s11548-007-0134-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-007-0134-0