Abstract
Objects
We present computer-assisted diagnosis (CAD) software designed to improve prostate cancer detection using perfusion MRI data.
Methods
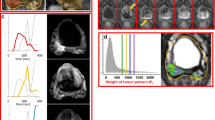
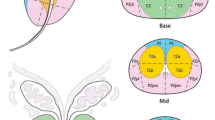
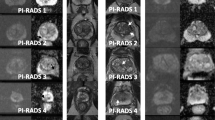
In addition to standard visualization features, this software allows for the 2D and multislice 2D contouring of suspicious areas based on a seeded region growing algorithm, and area labeling based on zonal anatomy. Tumor volume assessment and the semiquantitative analysis of DCE-MRI sequences can both be performed. We retrospectively analyzed DCE-MRI examinations of 100 patients and found 121 lesions showing a suspiciously high intensity with early enhancement in 84 of them. Seventy-one patients turned out to be malignant, whereas 50 were benign. Based on an analysis of the median wash-in and wash-out values of these foci, we designed a standardized 5-level cancer suspicion score (ranging from “probably benign” to “highly suspicious”). This comprehensive score provides a scaled likelihood of malignancy in the region of interest taking account of its location in relation to prostate zonal anatomy. We compared its accuracy with that of visual assessments of time–intensity curves performed by specialist and non-specialist radiologists.
Results
Parameters of the scoring algorithm were designed to provide the greatest possible sensitivity in our sample population. A re-substitution evaluation provided an Se/Sp of 100/45% for peripheral zone cancer, and 100/40% for transition zone cancer characterization. When identifying malignant areas using time–intensity curves data, this simple algorithm performed significantly better (AUC = 0.77) than a non-specialist (AUC = 0.57, P < 0.0001) radiologist, and better than a trained (AUC = 0.70) radiologist, although this difference was not significant.
Conclusion
Our new prostate MRI CAD software provides a standardized cancer suspicion score for suspicious foci detected in DCE-MRI T1-w images. Our results suggest that it may improve radiologists’ performances in prostate cancer identification, especially when they are not specialized in prostate imaging.
Similar content being viewed by others
References
Stamey TA, Caldwell M, McNeal JE, Nolley R, Hemenez M, Downs J (2004) The prostate specific antigen era in the United States is over for prostate cancer: what happened in the last 20 years?. J Urol 172: 1297–1301. doi:10.1097/01.ju.0000139993.51181.5d
Ito K, Ohi M, Yamamoto T, Miyamoto S, Kurokawa K, Fukabori Y et al (2002) The diagnostic accuracy of the age-adjusted and prostate volume-adjusted biopsy method in males with prostate specific antigen levels of 4.1–10.0 ng/mL. Cancer 95: 2112–2119. doi:10.1002/cncr.10941
Haider MA, van der Kwast TH, Tanguay J, Evans AJ, Hashmi AT, Lockwood G et al (2007) Combined T2-weighted and diffusion-weighted MRI for localization of prostate cancer. AJR Am J Roentgenol 189: 323–328. doi:10.2214/AJR.07.2211
Wetter A, Engl TA, Nadjmabadi D, Fliessbach K, Lehnert T, Gurung J et al (2006) Combined MRI and MR spectroscopy of the prostate before radical prostatectomy. AJR Am J Roentgenol 187: 724–730. doi:10.2214/AJR.05.0642
Kozlowski P, Chang SD, Jones EC, Berean KW, Chen H, Goldenberg SL (2006) Combined diffusion-weighted and dynamic contrast-enhanced MRI for prostate cancer diagnosis—correlation with biopsy and histopathology. J Magn Reson Imaging 24: 108–113. doi:10.1002/jmri.20626
Hricak H (2006) New horizons in genitourinary oncologic imaging. Abdom Imaging 31: 182–187. doi:10.1007/s00261-005-0385-6
Siegal JA, Yu E, Brawer MK (1995) Topography of neovascularity in human prostate carcinoma. Cancer 75: 2545–2551 doi:10.1002/1097-0142(19950515)75:10<2545::AID-CNCR2820751022>3.0.CO;2-X
Engelbrecht MR, Huisman HJ, Laheij RJ, Jager GJ, van Leenders GJ, Hulsbergen-Van De Kaa CA et al (2003) Discrimination of prostate cancer from normal peripheral zone and central gland tissue by using dynamic contrast-enhanced MR imaging. Radiology 229: 248–254. doi:10.1148/radiol.2291020200
Leach MO, Brindle KM, Evelhoch JL, Griffiths JR, Horsman MR, Jackson A et al (2005) The assessment of antiangiogenic and antivascular therapies in early-stage clinical trials using magnetic resonance imaging: issues and recommendations. Br J Cancer 92: 1599–1610. doi:10.1038/sj.bjc.6602550
Morgan VA, Kyriazi S, Ashley SE, DeSouza NM (2007) Evaluation of the potential of diffusion-weighted imaging in prostate cancer detection. Acta Radiol 48: 695–703. doi:10.1080/02841850701349257
Tanimoto A, Nakashima J, Kohno H, Shinmoto H, Kuribayashi S (2007) Prostate cancer screening: the clinical value of diffusionweighted imaging and dynamic MR imaging in combination with T2-weighted imaging. J Magn Reson Imaging 25(1): 146–152
Polascik TJ, Mouraviev V (2008) Focal therapy for prostate cancer. Curr Opin Urol 18: 269–274. doi:10.1097/MOU.0b013e3282f9b3a5
Beyersdorff D, Winkel A, Hamm B, Lenk S, Loening SA, Taupitz M (2005) MR imaging-guided prostate biopsy with a closed MR unit at 1.5 T: initial results. Radiology 234: 576–581. doi:10.1148/radiol.2342031887
Selli C, Caramella D, Giusti S, Conti A, Tognetti A, Mogorovich A et al (2007) Value of image fusion in the staging of prostatic carcinoma. La Radiologia medica 112 112(1): 74–81
Puech PA, Boussel L, Belfkih S, Lemaitre L, Douek P, Beuscart R (2007) DicomWorks: software for reviewing DICOM studies and promoting low-cost teleradiology. J Digit Imaging 20(2): 122–130
Adams R, Bischof L (1994) Seeded region growing. IEEE Trans Pattern Anal Mach Intell 16: 641–647. doi:10.1109/34.295913
Freeman H (1978) Application of the generalized chain coding scheme to map data processing. In: IEEE (ed) Proc IEEE Comp Soc Conf Patterns Recognition Map Data Processing
Preziosi P, Orlacchio A, Di Giambattista G, Di Renzi P, Bortolotti L, Fabiano A et al (2003) Enhancement patterns of prostate cancer in dynamic MRI. Eur Radiol 13: 925–930
Padhani AR, Gapinski CJ, Macvicar DA, Parker GJ, Suckling J, Revell PB et al (2000) Dynamic contrast enhanced MRI of prostate cancer: correlation with morphology and tumour stage, histological grade and PSA. Clin Radiol 55: 99–109. doi:10.1053/crad.1999.0327
Metz CE, Herman BA, Shen JH (1998) Maximum likelihood estimation of receiver operating characteristic (ROC) curves from continuously-distributed data. Stat Med 17: 1033–1053 doi: 10.1002/(SICI)1097-0258(19980515)17:9<1033::AID-SIM784>3.0.CO;2-Z
Girouin N, Mege-Lechevallier F, Tonina Senes A, Bissery A, Rabilloud M, Marechal JM et al (2007) Prostate dynamic contrast-enhanced MRI with simple visual diagnostic criteria: is it reasonable?. Eur Radiol 17: 1498–1509. doi:10.1007/s00330-006-0478-9
Noworolski SM, Henry RG, Vigneron DB, Kurhanewicz J (2005) Dynamic contrast-enhanced MRI in normal and abnormal prostate tissues as defined by biopsy, MRI, and 3D MRSI. Magn Reson Med 53: 249–255. doi:10.1002/mrm.20374
Turnbull LW, Buckley DL, Turnbull LS, Liney GP, Knowles AJ (1999) Differentiation of prostatic carcinoma and benign prostatic hyperplasia: correlation between dynamic Gd-DTPA-enhanced MR imaging and histopathology. J Magn Reson Imaging 9: 311–316 doi:10.1002/(SICI)1522-2586(199902)9:2<311::AID-JMRI24>3.0.CO;2-W
Kirkham AP, Emberton M, Allen C (2006) How good is MRI at detecting and characterising cancer within the prostate?. Eur Urol 50: 1163–1175. doi:10.1016/j.eururo.2006.06.025
Fichtinger G, DeWeese TL, Patriciu A, Tanacs A, Mazilu D, Anderson JH et al (2002) System for robotically assisted prostate biopsy and therapy with intraoperative CT guidance. Acad Radiol 9: 60–74. doi:10.1016/S1076-6332(03)80297-0
Fichtinger G, Burdette EC, Tanacs A, Patriciu A, Mazilu D, Whitcomb LL et al (2006) Robotically assisted prostate brachytherapy with transrectal ultrasound guidance—phantom experiments. Brachytherapy 5: 14–26. doi:10.1016/j.brachy.2005.10.003
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Puech, P., Betrouni, N., Makni, N. et al. Computer-assisted diagnosis of prostate cancer using DCE-MRI data: design, implementation and preliminary results. Int J CARS 4, 1–10 (2009). https://doi.org/10.1007/s11548-008-0261-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-008-0261-2