Abstract
Purpose
Cell nuclei classification in breast cancer histopathology images plays an important role in effective diagnose since breast cancer can often be characterized by its expression in cell nuclei. However, due to the small and variant sizes of cell nuclei, and heavy noise in histopathology images, traditional machine learning methods cannot achieve desirable recognition accuracy. To address this challenge, this paper aims to present a novel deep neural network which performs representation learning and cell nuclei recognition in an end-to-end manner.
Methods
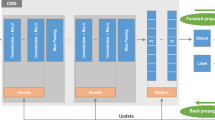
The proposed model hierarchically maps raw medical images into a latent space in which robustness is achieved by employing a stacked denoising autoencoder. A supervised classifier is further developed to improve the discrimination of the model by maximizing inter-subject separability in the latent space. The proposed method involves a cascade model which jointly learns a set of nonlinear mappings and a classifier from the given raw medical images. Such an on-the-shelf learning strategy makes obtaining discriminative features possible, thus leading to better recognition performance.
Results
Extensive experiments with benign and malignant breast cancer datasets are conducted to verify the effectiveness of the proposed method. Better performance was obtained when compared with other feature extraction methods, and higher recognition rate was achieved when compared with other seven classification methods.
Conclusions
We propose an end-to-end DNN model for cell nuclei and non-nuclei classification of histopathology images. It demonstrates that the proposed method can achieve promising performance in cell nuclei classification, and the proposed method is suitable for the cell nuclei classification task.












Similar content being viewed by others
Notes
The BCC database is downloaded from the website at http://bioimage.ucsb.edu/research/bio-segmentation.
References
Basavanhally A, Xu J, Madabhushi A, Ganesan S (2009) Computer-aided prognosis of er+ breast cancer histopathology and correlating survival outcome with oncotype dx assay. In: IEEE international symposium on Biomedical imaging: from nano to macro (ISBI’09). IEEE pp 851–854
Breiman L (2001) Random forests. Mach Learn 45(1):5–32
Chen X, Zhou X, Wong ST (2006) Automated segmentation, classification, and tracking of cancer cell nuclei in time-lapse microscopy. IEEE Trans Biomed Eng 53(4):762–766
Cirean DC, Giusti A, Gambardella LM, Schmidhuber J (2013) Mitosis detection in breast cancer histology images with deep neural networks. Springer, Berlin, pp 411–418
Dempster AP, Laird NM, Rubin DB (1977) Maximum likelihood from incomplete data via the EM algorithm. J Roy Stat Soc B 39(1):1–38
Fatakdawala H, Xu J, Basavanhally A, Bhanot G, Ganesan S, Feldman M, Tomaszewski JE, Madabhushi A (2010) Expectation maximization-driven geodesic active contour with overlap resolution (emagacor): application to lymphocyte segmentation on breast cancer histopathology. IEEE Trans Biomed Eng 57(7):1676–1689
Fukunaga K, Hostetler LD (1975) K-nearest-neighbor bayes-risk estimation. IEEE Trans Inf Theory 21(3):285–293
Gelasca E.D, Byun J, Obara B, Manjunath B (2008) Evaluation and benchmark for biological image segmentation. In: 15th IEEE International Conference on Image Processing (ICIP 2008). IEEE, pp 1816–1819
Glorot X, Bengio Y (2010) Understanding the difficulty of training deep feedforward neural networks. Aistats 9:249–256
Hatipoglu N, Bilgin G (2014) Classification of histopathological images using convolutional neural network. In: 2014 4th international conference on image processing theory, tools and applications (IPTA). IEEE, pp 1–6
Hinton GE, Osindero S, Teh YW (2006) A fast learning algorithm for deep belief nets. Neural Comput 18(7):1527–1554
Huang PW, Lee CH (2009) Automatic classification for pathological prostate images based on fractal analysis. IEEE Trans Med Imag 28(7):1037–1050
Kowal M (2014) Computer-aided diagnosis for breast tumor classification using microscopic images of fine needle biopsy. Springer, Berlin, pp 213–224
Kullback S, Leibler RA (1951) On information and sufficiency. Anna Math Stat 22(1):79–86
Larochelle H, Erhan D, Courville A, Bergstra J, Bengio Y (2007) An empirical evaluation of deep architectures on problems with many factors of variation. In: Proceedings of the 24th international conference on Machine learning. ACM, pp 473–480
LeCun Y, Jackel L, Bottou L, Brunot A, Cortes C, Denker J, Drucker H, Guyon I, Muller U, Sackinger E (1995) Comparison of learning algorithms for handwritten digit recognition. In: International conference on artificial neural networks, vol 60. pp 53–60
Liang M, Li Z, Chen T, Zeng J (2015) Integrative data analysis of multi-platform cancer data with a multimodal deep learning approach. IEEE/ACM Trans Comput Biol Bioinf 12(4):928–937. doi:10.1109/TCBB.2014.2377729
Lowe DG (2004) Distinctive image features from scale-invariant keypoints. Int J Comput Vis 60(2):91–110
McLachlan G (2004) Discriminant analysis and statistical pattern recognition. Wiley, Hoboken
Naik S, Doyle S, Agner S, Madabhushi A, Feldman M, Tomaszewski J (2008) Automated gland and nuclei segmentation for grading of prostate and breast cancer histopathology. In: 5th IEEE international symposium on biomedical imaging: from nano to macro (ISBI 2008). IEEE, pp 284–287
Ojansivu V, Linder N, Rahtu E, Pietikinen M, Lundin M, Joensuu H, Lundin J (2013) Automated classification of breast cancer morphology in histopathological images. Diagn Pathol 8(1):1–4
Pang B, Zhang Y, Chen Q, Gao Z, Peng Q, You X Cell nucleus segmentation in color histopathological imagery using convolutional networks. In: 2010 Chinese conference on pattern recognition (CCPR). IEEE, pp 1–5
Peng X, Yi Z, Tang H (2015) Robust subspace clustering via thresholding ridge regression. In: AAAI, pp 3827–3833
Peng X, Zhao B, Yan R, Tang H, Yi Z (2016) Bag of events: an efficient probability-based feature extraction method for aer image sensors. IEEE Trans Neural Netw Learn Syst 99:1–13
Poultney C, Chopra S, Cun Y.L (2007) Efficient learning of sparse representations with an energy-based model. In: Advances in neural information processing systems, pp 1137–1144
Ranzato MA, Huang FJ, Boureau YL, LeCun Y (2007) Unsupervised learning of invariant feature hierarchies with applications to object recognition. In: IEEE conference on computer vision and pattern recognition (CVPR’07). IEEE, pp 1–8
Schlkopf B, Smola A, Mller KR (1997) Kernel principal component analysis. Springer, Berlin, pp 583–588
Schmah T, Hinton G.E, Small S.L, Strother S, Zemel R.S (2009) Generative versus discriminative training of RBMS for classification of fmri images. In: Advances in neural information processing systems. pp 1409–1416
Scholkopf B, Smola AJ (2001) Learning with kernels: support vector machines, regularization, optimization, and beyond. MIT press, Cambridge
Sertel O, Kong J, Shimada H, Catalyurek U, Saltz JH, Gurcan MN (2009) Computer-aided prognosis of neuroblastoma on whole-slide images: classification of stromal development. Pattern recognition 42(6):1093–1103
Shin HC, Orton MR, Collins DJ, Doran SJ, Leach MO (2013) Stacked autoencoders for unsupervised feature learning and multiple organ detection in a pilot study using 4D patient data. IEEE Trans Pattern Anal Mach Intell 35(8):1930–1943
Shin M, Jang D, Nam H, Lee K.H., Lee D (2016) Predicting the absorption potential of chemical compounds through a deep learning approach. IEEE/ACM Trans Comput Biol Bioinf 99:1. doi:10.1109/TCBB.2016.2535233
Shlens J (2014) A tutorial on principal component analysis. arXiv preprint arXiv:1404.1100
Szegedy C, Liu W, Jia Y, Sermanet P, Reed S, Anguelov D, Erhan D, Vanhoucke V, Rabinovich A (2015) Going deeper with convolutions. In: Proceedings of the IEEE conference on computer vision and pattern recognition. pp 1–9
Vapnik VN, Vapnik V (1998) Statistical learning theory, vol 1. Wiley, New York
Vincent P, Larochelle H, Bengio Y, Manzagol P.A Extracting and composing robust features with denoising autoencoders. In: Proceedings of the 25th international conference on machine learning. ACM, pp 1096–1103
Vincent P, Larochelle H, Lajoie I, Bengio Y, Manzagol PA (2010) Stacked denoising autoencoders: learning useful representations in a deep network with a local denoising criterion. J Mach Learn Res 11:3371–3408
Xu J, Xiang L, Hang R, Wu J (2014) Stacked sparse autoencoder (ssae) based framework for nuclei patch classification on breast cancer histopathology. In: 2014 IEEE 11th international symposium on biomedical imaging (ISBI). IEEE, pp 999–1002
Xu J, Xiang L, Liu Q, Gilmore H, Wu J, Tang J, Madabhushi A (2016) Stacked sparse autoencoder (ssae) for nuclei detection on breast cancer histopathology images. IEEE Trans Med Imag 35(1):119–130
Zhang Z, Lyons M, Schuster M, Akamatsu S (1998) Comparison between geometry-based and gabor-wavelets-based facial expression recognition using multi-layer perceptron. In: 1998 proceedings third IEEE international conference on automatic face and gesture recognition. IEEE, pp 454–459
Acknowledgements
This work is supported by the Fok Ying Tung Education Foundation under Grant 151068, the Foundation for Youth Science and Technology Innovation Research Team of Sichuan Province 2016TD0018, and the National Natural Science Foundation of China under Grant 61332002.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Human and animal rights
No animal or human experiments were conducted as part of this research.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Rights and permissions
About this article
Cite this article
Feng, Y., Zhang, L. & Yi, Z. Breast cancer cell nuclei classification in histopathology images using deep neural networks. Int J CARS 13, 179–191 (2018). https://doi.org/10.1007/s11548-017-1663-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-017-1663-9