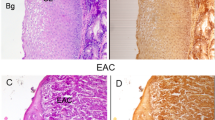
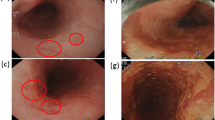
Abstract
Purpose
Esophageal carcinoma is the eighth most common cancer worldwide. Esophageal resection with gastric pull-up is a potentially curative therapeutic option. After this procedure, the specimen is examined by the pathologist to confirm complete removal of the cancer. An intraoperative analysis of the resectate would be less time-consuming and therefore improve patient safety.
Methods
Hyperspectral imaging (HSI) is a relatively new modality, which has shown promising results for the detection of tumors. Automatic approaches could support the surgeon in the visualization of tumor margins. Therefore, we evaluated four supervised classification algorithms: random forest, support vector machines (SVM), multilayer perceptron, and k-nearest neighbors to differentiate malignant from healthy tissue based on HSI recordings of esophago-gastric resectates in 11 patients.
Results
The best performances were obtained with a cancerous tissue detection of 63% sensitivity and 69% specificity with the SVM. In a leave-one patient-out cross-validation, the classification showed larger performance differences according to the patient data used. In less than 1 s, data classification and visualization was shown.
Conclusion
In this work, we successfully tested several classification algorithms for the automatic detection of esophageal carcinoma in resected tissue. A larger data set and a combination of several methods would probably increase the performance. Moreover, the implementation of software tools for intraoperative tumor boundary visualization will further support the surgeon during oncologic operations.



Similar content being viewed by others
References
Zhang Y (2013) Epidemiology of esophageal cancer. World J Gastroenterol 19:5598. https://doi.org/10.3748/wjg.v19.i34.5598
Flanagan JC, Batz R, Saboo SS, Nordeck SM, Abbara S, Kernstine K, Vasan V (2016) Esophagectomy and gastric pull-through procedures: surgical techniques, imaging features, and potential complications. RadioGraphics 36:107–121. https://doi.org/10.1148/rg.2016150126
Curtis NJ, Noble F, Bailey IS, Kelly JJ, Byrne JP, Underwood TJ (2014) The relevance of the Siewert classification in the era of multimodal therapy for adenocarcinoma of the gastro-oesophageal junction: Siewert Groups Retains Prognostic Value. J Surg Oncol 109:202–207. https://doi.org/10.1002/jso.23484
Ortega S, Fabelo H, Iakovidis D, Koulaouzidis A, Callico G (2019) Use of hyperspectral/multispectral imaging in gastroenterology. Shedding some–different–light into the dark. J Clin Med 8:36. https://doi.org/10.3390/jcm8010036
Nawn CD, Souhan BE, Carter R, Kneapler C, Fell N, Ye JY (2016) Distinguishing tracheal and esophageal tissues with hyperspectral imaging and fiber-optic sensing. J Biomed Opt 21:117004. https://doi.org/10.1117/1.JBO.21.11.117004
Fabelo H, Ortega S, Lazcano R, Madroñal DM, Callicó G, Juárez E, Salvador R, Bulters D, Bulstrode H, Szolna A, Piñeiro J, Sosa CJ, O’Shanahan A, Bisshopp S, Hernández M, Morera J, Ravi D, Kiran B, Vega A, Báez-Quevedo A, Yang G-Z, Stanciulescu B, Sarmiento R (2018) An intraoperative visualization system using hyperspectral imaging to aid in brain tumor delineation. Sensors 18:430. https://doi.org/10.3390/s18020430
Fabelo H, Ortega S, Kabwama S, Callico GM, Bulters D, Szolna A, Pineiro JF, Sarmiento R (2016) HELICoiD project: a new use of hyperspectral imaging for brain cancer detection in real-time during neurosurgical operations. In: Bannon DP (ed). Baltimore, Maryland, United States, p 986002
Ortega S, Fabelo H, Camacho R, de la Luz Plaza M, Callicó GM, Sarmiento R (2018) Detecting brain tumor in pathological slides using hyperspectral imaging. Biomed Opt Express 9:818. https://doi.org/10.1364/BOE.9.000818
Ravi D, Fabelo H, Callic GM, Yang G-Z (2017) Manifold embedding and semantic segmentation for intraoperative guidance with hyperspectral brain imaging. IEEE Trans Med Imaging 36:1845–1857. https://doi.org/10.1109/TMI.2017.2695523
Hohmann M, Kanawade R, Klämpfl F, Douplik A, Mudter J, Neurath MF, Albrecht H (2017) In-vivo multispectral video endoscopy towards in vivo hyperspectral video endoscopy. J Biophotonics 10:553–564. https://doi.org/10.1002/jbio.201600021
Ogihara H, Hamamoto Y, Fujita Y, Goto A, Nishikawa J, Sakaida I (2016) Development of a gastric cancer diagnostic support system with a pattern recognition method using a hyperspectral camera. J Sens 2016:1–6. https://doi.org/10.1155/2016/1803501
Akbari H, Halig LV, Schuster DM, Osunkoya A, Master V, Nieh PT, Chen GZ, Fei B (2012) Hyperspectral imaging and quantitative analysis for prostate cancer detection. J Biomed Opt 17:0760051. https://doi.org/10.1117/1.JBO.17.7.076005
Seibel EJ, Carroll RE, Dominitz JA, Johnston RS, Melville CD, Lee CM, Seitz SM, Kimmey MB (2008) Tethered capsule endoscopy, a low-cost and high-performance alternative technology for the screening of esophageal cancer and Barrett’s esophagus. IEEE Trans Biomed Eng 55:1032–1042. https://doi.org/10.1109/TBME.2008.915680
Hosking A, Coakley BJ, Chang D, Talebi-Liasi F, Lish S, Lee SW, Zong AM, Moore I, Browning J, Jacques SL, Krueger JG, Kelly KM, Linden KG, Gareau DS (2019) Hyperspectral imaging in automated digital dermoscopy screening for melanoma. Lasers Surg Med 51:214–222. https://doi.org/10.1002/lsm.23055
Baltussen EJM, Kok END, Brouwer de Koning SG, Sanders J, Aalbers AGJ, Kok NFM, Beets GL, Flohil CC, Bruin SC, Kuhlmann KFD, Sterenborg HJCM, Ruers TJM (2019) Hyperspectral imaging for tissue classification, a way toward smart laparoscopic colorectal surgery. J Biomed Opt 24:1. https://doi.org/10.1117/1.JBO.24.1.016002
Beaulieu RJ, Goldstein SD, Singh J, Safar B, Banerjee A, Ahuja N (2018) Automated diagnosis of colon cancer using hyperspectral sensing. Int J Med Robot 14:e1897. https://doi.org/10.1002/rcs.1897
Kulcke A, Holmer A, Wahl P, Siemers F, Wild T, Daeschlein G (2018) A compact hyperspectral camera for measurement of perfusion parameters in medicine. Biomed Tech (Berl) 63:519–527
Pedregosa F, Varoquaux G, Gramfort A, Michel V, Thirion B, Grisel O, Blondel M, Prettenhofer P, Weiss R, Dubourg V, Vanderplas J, Passos A, Cournapeau D, Brucher M, Perrot M, Duchesnay É (2011) Scikit-learn: machine learning in python. J Mach Learn Res 12:2825–2830
Lemaître G, Nogueira F, Aridas CK (2017) Imbalanced-learn: a python toolbox to tackle the curse of imbalanced datasets in machine learning. J Mach Learn Res 18:559–563
Chawla NV, Bowyer KW, Hall LO, Kegelmeyer WP (2002) SMOTE: synthetic minority over-sampling technique. J Artif Intell Res 16:321–357. https://doi.org/10.1613/jair.953
Savitzky A, Golay MJE (1964) Smoothing and differentiation of data by simplified least-squares procedures. Anal Chem 36:1627–1639. https://doi.org/10.1021/ac60214a047
Boughorbel S, Jarray F, El-Anbari M (2017) Optimal classifier for imbalanced data using Matthews Correlation Coefficient metric. PLoS ONE 12:e0177678. https://doi.org/10.1371/journal.pone.0177678
Lu G, Fei B (2014) Medical hyperspectral imaging: a review. J Biomed Opt 19:24
Baskar R, Lee KA, Yeo R, Yeoh K-W (2012) Cancer and radiation therapy: current advances and future directions. Int J Med Sci 9:193–199. https://doi.org/10.7150/ijms.3635
Ricci MS (2006) Chemotherapeutic approaches for targeting cell death pathways. Oncologist 11:342–357. https://doi.org/10.1634/theoncologist.11-4-342
Bashkatov AN, Genina EA, Kochubey VI, Tuchin VV (2005) Optical properties of human skin, subcutaneous and mucous tissues in the wavelength range from 400 to 2000 nm. J Phys Appl Phys 38:2543–2555. https://doi.org/10.1088/0022-3727/38/15/004
Funding
This study was funded by the Federal Ministry of Education and Research 13GW0248B.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards. Experimental hyperspectral measurements from patients for the evaluation of the new technology have obtained the ethics approval by the Ethics Committee of the University Hospital of Leipzig under 026/18-ek.
Informed consent
Informed consent was obtained from all individual participants included in the study.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Maktabi, M., Köhler, H., Ivanova, M. et al. Tissue classification of oncologic esophageal resectates based on hyperspectral data. Int J CARS 14, 1651–1661 (2019). https://doi.org/10.1007/s11548-019-02016-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-019-02016-x