Abstract
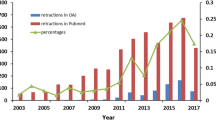
Among the many forms of research misconduct, publishing fraudulent data is considered to be serious where the confidence and validity of the research is detrimentally undermined. In this study, the trend of 303 retracted publications from 44 authors (with more than three retracted publications each) was analysed. The results showed that only 6.60% of the retracted publications were single-authored and the discovery of fraudulent publications had reduced from 52.24 months (those published before the year 2000) to 33.23 months (those published on the year 2000 and onwards). It appears that with the widely accessible public databases like PubMed, fraudulent publications can be detected more easily. The different approaches adopted by authors who had previous publications retracted are also discussed herein.
Similar content being viewed by others
References
Brach, M. A. (1998). Scapegoat for fraud in Germany? Nature, 392, 431.
Brown, H. (2007). How impact factor changed medical publishing and science. BMJ, 334, 561–564.
Darsee, J. R. (1983). A retraction of two papers on cardiomyopathy. New England Journal of Medicine, 308, 1419.
Franzen, Martina, Rodder, Simone, & Weingart, Peter. (2007). Fraud: Causes and culprits as perceived by science and the media. EMBO Reports, 8, 3–7.
Fuchs, S., & Westervelt, S. D. (1996). Fraud and trust in science. Perspectives in Biology and Medicine, 39, 248–269.
Giles, J. (2005). Taking on the cheats. Nature, 435, 258–259.
Graf, C., Wager, E., Bowman, A., Fiack, S., Scott-Lichter, D., & Robinson, A. (2007). Best practice guidelines on publication ethics: A publisher’s perspective. International Journal of Clinical Practice, 61(Suppl 152), 1–26.
Hwang, W. S., Roh, S. I., Lee, B. C., Kang, S. K., Kwon, D. K., Kim, et al. (2005). Patient-specific embryonic stem cells derived from human SCNT blastocysts. Science, 308, 1777–1783. Retraction in: Kennedy, D. (2006). Science, 311, 335.
Hwang, W. S., Ryu, Y. J., Park, J. H., Park, E. S., Lee, E. G., Koo, J. M., et al. (2004). Evidence of a pluripotent human embryonic stem cell line derived from a cloned blastocyst. Science, 303, 1669–1674. Retraction in: Kennedy, D. (2006). Science, 311, 335.
Kakuk, P. (2009). The legacy of the Hwang case: Research misconduct in biosciences. Science and Engineering Ethics. doi:10.1007/s11948-009-9121-x.
Martinson, B. C., Anderson, M. S., & de Vries, R. (2005). Scientists behaving badly. Nature, 435, 737–738.
Plemmons, D. K., Brody, S. A., & Kalichman, M. W. (2006). Student perceptions of the effectiveness of education in the responsible conduct of research. Science and Engineering Ethics, 12, 571–582.
Powell, S. T., Allison, M. A., & Kalichman, M. W. (2007). Effectiveness of a responsible conduct of research course: A preliminary study. Science and Engineering Ethics, 13, 249–264.
Reich, E. S. (2009). The rise and fall of a physics fraudster. Physics World, 22, 24.
Smith, R. (2005). Investigating the previous studies of a fraudulent author. BMJ, 331, 288–291.
Sox, H. C., & Rennie, D. (2006). Research misconduct, retraction, and cleansing the medical literature: Lessons from the Poehlman case. Annals of Internal Medicine, 144, 609–613.
Williams, G., & Hobbs, R. (2007). Should we ditch impact factors? BMJ, 334, 568–569.
Acknowledgments
All the bibliographical data used were obtained from the PubMed database (United States National Library of Medicine, Bethesda, MD).
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Foo, J.Y.A. A Retrospective Analysis of the Trend of Retracted Publications in the Field of Biomedical and Life Sciences. Sci Eng Ethics 17, 459–468 (2011). https://doi.org/10.1007/s11948-010-9212-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11948-010-9212-8