Abstract
How are we to go about understanding the computations that underpin cognition? Here we set out a methodological framework that helps understand different approaches to solving this problem. We argue that a very powerful stratagem is to attempt to ‘reverse engineer’ the brain and that computational neuroscience plays a pivotal role in this programme. En passant, we also tackle the oft-asked and prior question of why we should build computational models of any kind. Our framework uses four levels of conceptual analysis: computation, algorithm, mechanism and biological substrate. As such it enables us to understand how (algorithmic) AI and connectionism may be recruited to help propel the reverse-engineering programme forward. The framework also incorporates the notion of different levels of structural description of the brain, and analysis of this issue gives rise to a novel proposal for capturing computations at multiple levels of description in a single model.








Similar content being viewed by others
Notes
In Marr’s original formulation of the computational framework, which appeared in an MIT technical report [35], a fourth level was described. However, this was dropped in the more popular account in Marr [34]. Independently, Gurney proposed a four level account in Ref. [15] which was subsequently developed in Ref. [19].
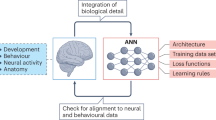
It is often argued that a ‘divine gift’ of a complete model of the brain would be useless. In the light of the above discussion, however, it would appear this is not true. It may be arduous to unravel the function of all aspects of the model/brain, but this task would certainly be easier than using biological experiments alone.
References
Bogacz R, Brown E, Moehlis J, Holmes P, Cohen JD. The physics of optimal decision making: a formal analysis of models of performance in two-alternative forced-choice tasks. Psychol Rev. 2006;113(4):700–65.
Bogacz R, Gurney K. The basal ganglia and cortex implement optimal decision making between alternative actions. Neural Comput. 2007;19(2):442–77.
Booch G. The accidental architecture. IEEE Software. 2006;23:9–11.
Chun MM, Nakayama K. On the functional role of implicit visual memory for the adaptive deployment of attention across scenes. Vis Cogn. 2000;7:65–81.
Churchland PS, Sejnowski TJ. The computational brain. Cambridge, MA: The MIT Press; 1992.
Cohen JD, Dunbar K, McClelland JL. On the control of automatic processes—a parallel distributed-processing account of the stroop effect. Psychol Rev. 1990;97(3):332–61.
Connor CE, Egeth HE, Yantis S. Visual attention: bottom-up versus top-down. Curr Biol. 2004;14(19):R850–2.
Dennett D. When philosophers encounter artificial intelligence. Daedalus. 1988;117:283–95. Reprinted in ‘Brain Children’ by D.C. Dennett, MIT Press, 1998.
De Schutter E. Reviewing multi-disciplinary papers: a challenge in neuroscience? Neuroinformatics. 2008;6(4):253–5.
Doya K. What are the computations of the cerebellum, the basal ganglia and the cerebral cortex? Neural Netw. 1999;12(7-8):961–74.
Dragalin VP, Tartakovsky AG, Veeravalli VV. Multihypothesis sequential probability ratio tests—part i: asymptotic optimality. IEEE Trans Inf Theory. 1999;45(7):2448–61.
Epstein JM. Why model? J Artif Soc Social Simul. 2008;11(4):12.
Findlay JM, Gilchrist ID. Active vision: the psychology of looking and seeing. Oxford, UK: Oxford University Press; 2003.
Girard B, Berthoz A. From brainstem to cortex: computational models of saccade generation circuitry. Prog Neurobiol. 2005;77(4):215–51.
Gurney KN. An introduction to neural networks. London, UK: UCL Press (Taylor and Francis group); 1997.
Gurney KN, Humphries M, Wood R, Prescott TJ, Redgrave P. Testing computational hypotheses of brain systems function: a case study with the basal ganglia. Network. 2004;15(4):263–90.
Gurney KN, Prescott TJ, Redgrave P. A computational model of action selection in the basal ganglia i: a new functional anatomy. Biol Cybern. 2001;84:401–10.
Gurney KN, Prescott TJ, Redgrave P. A computational model of action selection in the basal ganglia ii: analysis and simulation of behaviour. Biol Cybern. 2001;84:411–23.
Gurney KN, Prescott TJ, Wickens JR, Redgrave P. Computational models of the basal ganglia: from robots to membranes. Trends Neurosci. 2004;27(8):453–9.
Gurney KN, Wright MJ. A model for the spatial integration and differentiation of velocity signals. Vision Res. 1996;36(18):2939–55.
Hikosaka O, Nakamura K, Nakahara H. Basal ganglia orient eyes to reward. J Neurophysiol. 2006;95(2):567–84.
Hinton GE, Shallice T. Lesioning an attractor network: investigations of acquired dyslexia. Psychol Rev. 1991;98(1):74–95.
Humphries MD. High level modeling of dopamine mechanisms in striatal neurons (tech. rep.). Sheffield: Department of Psychology, University of Sheffield; 2003.
Humphries MD, Gurney KN. A pulsed neural network model of bursting in the basal ganglia. Neural Netw. 2001;14(6-7):845–63.
Humphries MD, Gurney KN. The role of intra-thalamic and thalamocortical circuits in action selection. Network. 2002;13(1):131–56.
Humphries MD, Gurney KN. Deep brain stimulation of the subthalamic nucleus causes paradoxical inhibition of output in a computational model of the “parkinsonian” basal ganglia. Society for Neuroscience Annual Meeting Session 622.9; 2007.
Humphries MD, Stewart RD, Gurney KN. A physiologically plausible model of action selection and oscillatory activity in the basal ganglia. J Neurosci. 2006;26(50):12921–42.
Hussain A, Gurney K, Abdullah R, Chambers J. Emergent common functional principles in control theory and the vertebrate brain: a case study with autonomous vehicle control. Icann 2008;(2):949–58.
Izhikevich. Dynamical systems in neuroscience: the geometry of excitability. Cambridge, MA: MIT Press; 2007.
Kitano H. Computational systems biology. Nature. 2002;420(6912):206–10.
Koch C. The biophysics of computation: information processing in single neurons. New York: Oxford University Press; 1999.
Lee TS, Yuille AL. Bayesian brain: probabilistic approaches to neural coding. In: Doya K, Ishi S, Pouget A, Rao RPN, editors. Cambridge, MA: MIT Press; 2007. p. 145–88.
Markram H. The blue brain project. Nat Rev Neurosci. 2006;7(2):153–60.
Marr D. Vision: a computational investigation into human representation and processing of visual information. New York: WH Freeeman and Co.; 1982.
Marr D, Poggio T. From understanding computation to understanding neural circuitry (tech. rep. no. AIM-357). MIT; 1976.
Mel BW, Ruderman DL, Archie KA. Translation-invariant orientation tuning in visual “complex” cells could derive from intradendritic computations. J Neurosci. 1998;18(11):4325–34.
Mink JW, Thach WT. Basal ganglia intrinsic circuits and their role in behavior. Curr Opin Neurobiol. 1993;3(6):950–7.
Minsky M. The society of mind. New York: Simon and Schuster; 1988.
Niv Y, Schoenbaum G. Dialogues on prediction errors. Trends Cogn Sci. 2008;12(7):265–72.
Prescott AJ, Gonzales FM, Gurney KN, Humphries M, Redgrave P. A robot model of the basal ganglia: behavior and intrinsic processing. Neural Netw 2005;19(1):31–61.
Putnam H. Artificial intelligence: much ado about not very much. Daedalus. 1988;117:269–81.
Rao RPN. Bayesian brain: probabilistic approaches to neural coding. In: Doya K, Ishi S, Pouget A, Rao RPN, editors. Cambridge, MA: MIT Press; 2007. p. 239–67.
Redgrave P. Basal ganglia. Scholarpedia 2007. http://www.scholarpedia.org/article/Basal_ganglia.
Redgrave P, Prescott TJ, Gurney KN. The basal ganglia: a vertebrate solution to the selection problem? Neuroscience. 1999;89:1009–23.
Schall JD. The neural selection and control of saccades by the frontal eye field. Philos Trans R Soc Lond B Biol Sci. 2002;357(1424):1073–82.
Schultz W, Dayan P, Montague PR. A neural substrate of prediction and reward. Science. 1997;275(5306):1593–9.
Sejnowski TJ, Koch C, Churchland PS. Computational neuroscience. Science. 1988;241(4871):1299–306.
Servan-Schreiber D, Printz H, Cohen JD. A network model of catecholamine effects: gain, signal-to-noise ratio, and behavior. Science. 1990;249(4971):892–5.
Smolensky P. On the proper treatment of connectionism. Behav Brain Sci. 1988;11:1–23.
Stafford T, Gurney KN. Biologically constrained action selection improves cognitive control in a model of the stroop task. Philos Trans R Soc Lond B Biol Sci. 2007;362(1485):1671–84.
Acknowledgements
This work was supported by EPSRC grant EP/C516303/1. I would like to acknowledge all the members of the Adaptive Behaviour Research Group, past and present, who contributed to the work presented here. In particular, I would like to thank Nathan Lepora for reading an early draft of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Gurney, K.N. Reverse Engineering the Vertebrate Brain: Methodological Principles for a Biologically Grounded Programme of Cognitive Modelling. Cogn Comput 1, 29–41 (2009). https://doi.org/10.1007/s12559-009-9010-2
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12559-009-9010-2