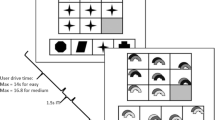
Abstract
Deductive and inductive reasoning are fundamental logical processes critical to the solution of common practical problems in daily life. We used functional magnetic resonance imaging (fMRI) to investigate the brain networks involved in Contradictory, Deductive, and Inductive judgments. The experimental paradigm was based on categorical propositions of the Aristotelian Square of Opposition (ASoO). In a full factorial design, identical sentences were combined into premise–conclusion pairs. Each sentence started with ‘every’ or ‘some’. The order of the two propositions in the pair created two types of logical operators (every→some: deductive, or some→every: inductive). The descriptive attributes of the category could be Contradictory or non-Contradictory. Imaging data was analyzed using Group Independent component analysis of fMRI Toolbox (GIFT). Connectivity of nodes within four intrinsic connectivity networks (ICNs) was sensitive to attribute manipulation (Contradiction): the anterior default mode network (aDMN), and the language and cerebellum networks were more involved in Contradictory than non-Contradictory statements, while the anterior salience network (aSN) showed the opposite pattern. Five networks were associated with logical operator manipulation. Stronger positive associations with Inductive than Deductive reasoning were observed in the dorsal and ventral parts of the aDMN, aSN, and orbitofrontal networks (OFN). A stronger negative association with deductive than inductive reasoning was observed in the executive control (ExCN) and dorsal attention (DAN) networks. Differences in the fractional amplitude of low‐frequency fluctuation of the BOLD signal in aDMN, ExCN, and OFN explained 67% of the variance of the behavioural cost of inductive relative to deductive reasoning. The results suggest that different ICNs support logical reasoning and conflict identification. Finally, the magnitude of the differences was positively correlated with behavioural cost.






Similar content being viewed by others
References
Canessa N, Gorini A, Cappa SF, Piattelli-Palmarini M, Danna M, Fazio F, Perani D. The effect of social content on deductive reasoning: an fMRI study. Hum Brain Mapp. 2005;26:30–43. https://doi.org/10.1002/hbm.20114.
Lassiter D, Goodman ND. How many kinds of reasoning? Inference, probability, and natural language semantics. Cognition. 2015;136:123–34. https://doi.org/10.1016/j.cognition.2014.10.016.
Heit E, Rotello CM. Relations between inductive reasoning and deductive reasoning. J Exp Psychol Learn Mem Cogn. 2010;36:805. https://doi.org/10.1037/a0018784.
Rips LJ. Two kinds of reasoning. Psychol Sci. 2001;12:129–34. https://doi.org/10.1111/1467-9280.00322.
Parsons T. The traditional square of opposition. In: Zalta EN, editor. The Stanford Encyclopedia of Philosophy (Fall 2021 Edition). California: Stanford University; 2017.
Goodman ND, Lassiter D. Probabilistic semantics and pragmatics uncertainty in language and thought. In: Lappin S, Fox C, editors. The Handbook of Contemporary Semantic Theory. Hoboken: John Wiley & Sons; 2015. https://doi.org/10.1002/9781118882139.ch21.
Oaksford M, Chater N. Bayesian rationality: the probabilistic approach to human reasoning. England: Oxford University Press; 2010. https://doi.org/10.1093/acprof:oso/9780198524496.001.0001.
Rotello CM, Heit E, Kelly LJ. Do modals identify better models? A comparison of signal detection and probabilistic models of inductive reasoning. Cogn Psychol. 2019;112:1–24. https://doi.org/10.1016/j.cogpsych.2019.03.004.
Goel V, Gold B, Kapur S, Houle S. Neuroanatomical correlates of human reasoning. J Cogn Neurosci. 1998;10:293–302. https://doi.org/10.1162/089892998562744.
Goel V, Gold B, Kapur S, Houle S. The seats of reason? An imaging study of deductive and inductive reasoning. NeuroReport. 1997;8:1305–10. https://doi.org/10.1097/00001756-199703240-00049.
Osherson D, Perani D, Cappa S, Schnur T, Grassi F, Fazio F. Distinct brain loci in deductive versus probabilistic reasoning. Neuropsychologia. 1998;36:369–76. https://doi.org/10.1016/S0028-3932(97)00099-7.
Parsons LM. New evidence for distinct right and left brain systems for deductive versus probabilistic reasoning. Cereb Cortex. 2001;11:954–65. https://doi.org/10.1093/cercor/11.10.954.
Goel V, Dolan RJ. Differential involvement of left prefrontal cortexin inductive and deductive reasoning. Cognition. 2004;93:109–21. https://doi.org/10.1016/j.cognition.2004.03.001.
Parsons LM, Osherson D. New evidence for distinct right and left brain systems for deductive versus probabilistic reasoning. Cereb Cortex. 2001;11:954–65. https://doi.org/10.1093/cercor/11.10.954.
Medaglia MT, Tecchio F, Seri S, Di Lorenzo G, Rossini PM, Porcaro C. Contradiction in universal and particular reasoning. Hum Brain Mapp. 2009;30:4187–97. https://doi.org/10.1002/hbm.20838.
Porcaro C, Medaglia MT, Thai NJ, Seri S, Rotshtein P, Tecchio F. Contradictory reasoning network: an EEG and fMRI study. PLoS ONE. 2014;9: e92835. https://doi.org/10.1371/journal.pone.0092835.
Goel V. Anatomy of deductive reasoning. Trends Cogn Sci. 2007;11:435–41. https://doi.org/10.1016/j.tics.2007.09.003.
Prado J, Chadha A, Booth JR. The brain network for deductive reasoning: a quantitative meta-analysis of 28 neuroimaging studies. J Cogn Neurosci. 2011;23:3483–97. https://doi.org/10.1162/jocn_a_00063.
Friston KJ. Functional and effective connectivity: a review. Brain Connect. 2011;1:13–36. https://doi.org/10.1089/brain.2011.0008.
Hermundstad AM, Bassett DS, Brown KS, Aminoff EM, Clewett D, Freeman S, Frithsen A, Johnson A, Tipper CM, Miller MB, Grafton ST, Carlson JM. Structural foundations of resting-state and task-based functional connectivity in the human brain. Proc Natl Acad Sci USA. 2013;110:6169–74. https://doi.org/10.1073/pnas.1219562110.
van den Heuvel M, Mandl R, Pol HH. Normalized cut group clustering of resting-state fMRI data. PLoS ONE. 2008;3: e2001. https://doi.org/10.1371/journal.pone.0002001.
Allen EA, Damaraju E, Plis SM, Erhardt EB, Eichele T, Calhoun VD. Tracking whole-brain connectivity dynamics in the resting state. Cereb Cortex. 2014;24:663–76. https://doi.org/10.1093/cercor/bhs352.
Buckner RL, Andrews-Hanna JR, Schacter DL. The brain’s default network: anatomy, function, and relevance to disease. Ann NY Acad Sci. 2008;1124:1–38. https://doi.org/10.1196/annals.1440.011.
Menon V, Uddin LQ. Saliency switching attention and control: a network model of insula function. Brain Struct Funct. 2010;214:655–67. https://doi.org/10.1007/s00429-010-0262-0.
Beckmann CF, DeLuca M, Devlin JT, Smith SM. Investigations into resting-state connectivity using independent component analysis. Philos Trans R Soc B Biol Sci. 2005;360:1001–13. https://doi.org/10.1098/rstb.2005.1634.
Cole MW, Bassett DS, Power JD, Braver TS, Petersen SE. Intrinsic and task-evoked network architectures of the human brain. Neuron. 2014;83:238–51. https://doi.org/10.1016/j.neuron.2014.05.014.
Gonzalez-Castillo J, Bandettini PA. Task-based dynamic functional connectivity: recent findings and open questions. Neuroimage. 2018;180:526–33. https://doi.org/10.1016/j.neuroimage.2017.08.006.
McKeown MJ, Makeig S, Brown GG, Jung TP, Kindermann SS, Bell AJ, Sejnowski TJ. Analysis of fMRI data by blind separation into independent spatial components. Hum Brain Mapp. 1998;6:160–88.
Worsley KJ, Friston KJ. Analysis of fMRI time-series revisited — again. Neuroimage. 1995;2:173–81. https://doi.org/10.1006/nimg.1995.1023.
Calhoun VD, Adali T, Pearlson GD, Pekar JJ. Spatial and temporal independent component analysis of functional MRI data containing a pair of task-related waveforms. Hum Brain Mapp. 2001;13:43–53. https://doi.org/10.1002/hbm.1024.
Calhoun VD, Adali T, Pearlson GD, Pekar JJ. A method for making group inferences from functional MRI data using independent component analysis. Hum Brain Mapp. 2001;14:140–51. https://doi.org/10.1002/hbm.1048.
Bell AJ, Sejnowski TJ. An information-maximization approach to blind separation and blind deconvolution. Neural Comput. 1995;7:1129–59. https://doi.org/10.1162/neco.1995.7.6.1129.
Li YO, Adali T, Calhoun VD. Estimating the number of independent components for functional magnetic resonance imaging data. Hum Brain Mapp. 2007;28:1251–66. https://doi.org/10.1002/hbm.20359.
Porcaro C, Di Renzo A, Tinelli E, Di Lorenzo G, Parisi V, Caramia F, Fiorelli M, Di Piero V, Pierelli F, Coppola G. Haemodynamic activity characterization of resting state networks by fractal analysis and thalamocortical morphofunctional integrity in chronic migraine. J Headache Pain. 2020;21:112. https://doi.org/10.1186/s10194-020-01181-8.
Porcaro C, Mayhew SD, Marino M, Mantini D, Bagshaw AP. Characterisation of haemodynamic activity in resting state networks by fractal analysis. Int J Neural Syst. 2020;30:2050061. https://doi.org/10.1142/S0129065720500616.
Porcaro C, Zappasodi F, Rossini PM, Tecchio F. Choice of multivariate autoregressive model order affecting real network functional connectivity estimate. Clin Neurophysiol. 2009;120:436–48. https://doi.org/10.1016/j.clinph.2008.11.011.
Genovese CR, Lazar NA, Nichols T. Thresholding of statistical maps in functional neuroimaging using the false discovery rate. Neuroimage. 2002;15:870–8. https://doi.org/10.1006/nimg.2001.1037.
Liu CH, Ma X, Wu X, Fan TT, Zhang Y, Zhou FC, Li LJ, Li F, Tie CL, Li SF, Zhang D, Zhou Z, Dong J, Wang YJ, Yao L, Wang CY. Resting-state brain activity in major depressive disorder patients and their siblings. J Affect Disord. 2013;149:299–306. https://doi.org/10.1016/j.jad.2013.02.002.
Johnson-Laird PN. Mental models and deduction. Trends Cogn Sci. 2001;5:434–42. https://doi.org/10.1016/S1364-6613(00)01751-4.
Braine MDS, O’Brien DP, Noveck IA, Samuels MC, Lea RB, Fisch SM, Yang Y. Further evidence for the theory: predicting intermediate and multiple conclusions in propositional logic inference problems. In: Braine MDS, O’Brien DP, Braine M, editors. Mental Logic. Wales: Taylor & Francis; 1998.
Oaksford M. Imaging deductive reasoning and the new paradigm. Front Hum Neurosci. 2015;9:101. https://doi.org/10.3389/fnhum.2015.00101.
Zhang S, Tsai SJ, Hu S, Xu J, Chao HH, Calhoun VD, Li CSR. Independent component analysis of functional networks for response inhibition: Inter-subject variation in stop signal reaction time. Hum Brain Mapp. 2015;36:3289–302. https://doi.org/10.1002/hbm.22819.
Atkinson RK, Derry SJ, Renkl A, Wortham D. Learning from examples: instructional principles from the worked examples research. Rev Educ Res. 2000;70:181–214. https://doi.org/10.3102/00346543070002181.
van Gog T, Rummel N. Example-based learning: integrating cognitive and social-cognitive research perspectives. Educ Psychol Rev. 2010;22:155–74. https://doi.org/10.1007/s10648-010-9134-7.
Calhoun VD, Adali T, Hansen LK. 2003. ICA of functional MRI data: an overview. In: Proceedings of the International Workshop on Independent Component Analysis and Blind Signal Separation. Centre County: Pennsylvania State University; 2003.
Price CJ, Moore CJ, Friston KJ. Subtractions conjunctions and interactions in experimental design of activation studies. Hum Brain Mapp. 1997;5:264–72. https://pubmed.ncbi.nlm.nih.gov/20408227/.
Heit E. Brain imaging, forward inference, and theories of reasoning. Front Hum Neurosci. 2015;8:1056. https://doi.org/10.3389/fnhum.2014.01056.
Henson R. Forward inference using functional neuroimaging: dissociations versus associations. Trends Cogn Sci. 2006;10:64–9. https://doi.org/10.1016/j.tics.2005.12.005.
Henson R. What can functional neuroimaging tell the experimental psychologist? Q J Exp Psychol Sect A Hum Exp Psychol. 2005;58:193–233. https://doi.org/10.1080/02724980443000502.
Author information
Authors and Affiliations
Contributions
Study conception and design were performed by Camillo Porcaro and Maria Teresa Medaglia. Material preparation and data collection were performed by Camillo Porcaro and Maria Teresa Medaglia. Data analysis was performed by Camillo Porcaro and Silvia Angela Mansi. The first draft of the manuscript was written by Camillo Porcaro, Silvia Angela Mansi, Maria Teresa Medaglia, and Pia Rotshtein. All authors commented on previous versions of the manuscript. All authors read and approved the final manuscript.
Corresponding author
Ethics declarations
Ethical Approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Informed Consent
After full disclosure of the study purpose, all the participants gave their informed written consent.
Conflict of Interest
The authors declare no competing interests.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Silvia Angela Mansi and Medaglia Maria Teresa equally contributed to the manuscript.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Mansi, S.A., Teresa, M.M., Seri, S. et al. Frontal Intrinsic Connectivity Networks Support Contradiction Identification During Inductive and Deductive Reasoning. Cogn Comput 14, 677–692 (2022). https://doi.org/10.1007/s12559-021-09982-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s12559-021-09982-y