Abstract
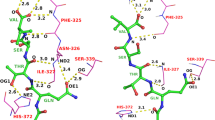
This paper describes the application of PRO−LEADS to the flexible docking of ligands into crystallographically derived enzyme structures that are assumed to be rigid. PRO−LEADS uses a Tabu search methodology to perform the flexible search and an empirically derived estimate of the binding affinity to drive the docking process. The paper tests the extent to which the assumption of a rigid enzyme compromises the accuracy of the results. All-pairs docking experiments are performed for three enzymes (thrombin, thermolysin and influenza virus neuraminidase) based on six or more ligand-enzyme crystal structures for each enzyme. In 76% of the cases, PRO−LEADS can successfully identify the correct ligand conformation as the lowest energy configuration when the enzyme structure is derived from that ligand's crystal structure, but the methodology only docks 49% of the cases successfully when the ligand is docked against enzyme crystal structures derived from other ligands. Small movements in the enzyme structure lead to an under-prediction in the energy of the correct binding mode by up to 14 kJ/mol and in some cases this under-prediction can lead to the native mode not being recognised as the lowest energy solution. The type of movements responsible for mis-docking are: the movement of sidechains as a result of changes in Cα position; the movement of sidechains without changes in Cα position; the movement of flexible portions of main chains to facilitate the formation of hydrogen bonds; and the movement of metal atoms bound to the enzyme active site. The work illustrates that the assumption of a rigid active site can lead to errors in identification of the correct binding mode and the assessment of binding affinity, even for enzymes which show relatively small shift in atomic positions from one ligand to the next. A good docking code, such as PRO−LEADS, can usually dock successfully if there is induced fit in relatively rigid enzymes but there remains the need to develop improved strategies for dealing with enzyme flexibility. The work implies that treatments of enzyme flexibility which focus only on sidechain rotations will not deal with the critical shifts responsible for mis-docking of ligands in thrombin, thermolysin and neuraminidase. The paper demonstrates the utility of all pairs docking experiments as a method of assessing the effectiveness of docking methodologies in dealing with enzyme flexibility.
Similar content being viewed by others
References
Blaney, J.M. and Dixon, J.S., Perspect. Drug Discov. Design, 1 (1993) 301.
Jones, G. and Willett, P., Curr. Opin. Biotech., 6 (1995) 652.
Lybrand, T.P., Curr. Opin. Struct. Biol., 5 (1995) 224.
Rosenfeld, R., Vajda, S. and Delisi, C., Ann. Rev. Biophys. Biomol. Struct., 24 (1995) 677.
Gschwend, D.A., Good, A.C. and Kuntz, I.D., J. Mol. Recognition, 9 (1996) 175.
Lengauer, T. and Rarey, M., Curr. Opin. Struct. Biol., 6 (1996) 402.
Kuntz, I.D., Blaney, J.M., Oatley, S.J., Langridge, R. and Ferrin, T.E., J. Mol. Biol., 161 (1982) 269.
Miller, M.D., Kearsley, S.K., Underwood, D.J. and Sheridan, R.P., J. Comput.-Aided Mol. Design, 8 (1994) 153.
Welch, W., Ruppert, J. and Jain, A.N., Chem. Biol., 3 (1996) 449.
Goodsell, D.S. and Olson, A.J., Proteins Struct. Funct. Genet., 8 (1990) 195.
Morris, G.M., Goodsell, D.S., Huey, R. and Olson, A.J., J. Comput.-Aided Mol. Design, 10 (1996) 293.
Gehlhaar, D.K., Verkhivker, G.M., Rejto, P.A., Sherman, C.J., Fogel, D.B., Fogel, L.J. and Freer, S.T., Chem. Biol., 2 (1995) 317.
Verkhivker, G.M., Rejto, P.A., Gehlhaar, D.K. and Freer, S.T., Proteins Struct. Funct. Genet., 250 (1996) 342.
Jones, G., Willett, P. and Glen, R.C., J. Mol. Biol., 245 (1995) 43.
Jones, G., Willett, P., Glen, R.C., Leach, A.R. and Taylor, R., J. Mol. Biol., 267 (1997) 727.
Judson, R.S., Tan, Y.T., Mori, E., Melius, C., Jaeger, E.P., Treasurywala, A.M. and Mathiowetz, A., J. Comput. Chem., 16 (1995) 1405.
Oshiro, C.M., Kuntz, I.D. and Dixon, J.S., J. Comput.-Aided Mol. Design, 9 (1995) 113.
Clark, K.P. and Ajay, J. Comput. Chem., 16 (1995) 1210.
Yamada, M. and Itai, A., Chem. Pharm. Bull., 41 (1993) 1200.
McMartin, C. and Bohacek, R.S., J. Comput.-Aided Mol. Design, 11 (1997) 333.
Leach, A.R. and Kuntz, I.D., J. Comput. Chem., 13 (1992) 730.
Rarey, M., Kramer, B., Lengauer, T. and Klebe, G., J. Mol. Biol., 261 (1996) 470.
Rarey, M., Kramer, B., Lengauer, T. and Klebe, G., J. Comput.-Aided Mol. Design, 11 (1997) 369.
Westhead, D.R., Clark, D.E. and Murray, C.W., J. Comput.-Aided Mol. Design, 11 (1997) 209.
Glover, F. and Laguna, M., In Reeves, C.R. (Ed.), Modern Heuristic Techniques for Combinatorial Problems, Blackwell Scientific Publications, Oxford, 1993, pp. 70–150.
Eldridge, M.D., Murray, C.W., Auton, T.R., Paolini, G.V. and Mee, R.P., J. Comput.-Aided Mol. Design, 11 (1997) 425.
Böhm, H.-J., J. Comput.-Aided Mol. Design, 8 (1994) 243.
Jain, A.J., J. Comput.-Aided Mol. Design, 10 (1996) 427.
Bernstein, F.C., Koetzle, T.F., Williams, G.J.B., Meyer, E.F., Brice, M.D., Rodgers, J.R., Kennard, O., Shimanouchi, T. and Tasumi, M., J. Mol. Biol., 112 (1977) 535.
Baxter, C.A., Murray, C.W., Clark, D.E., Westhead, D.R. and Eldridge, M.D., Proteins Struct. Funct. Genet., 33 (1998) 367.
Molecular Simulations Inc., San Diego, CA. InsightII/Discover, v95.0, 1996.
Leach, A.R., J. Mol. Biol., 235 (1994) 345.
GOLD; http://www.ccdc.cam.ac.uk/prods/gold/index.html
Knegtel, R.A., Kuntz, I.D. and Oshiro, C.M., J. Mol. Biol., 266 (1997) 424.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Murray, C.W., Baxter, C.A. & Frenkel, A.D. The sensitivity of the results of molecular docking to induced fit effects: Application to thrombin, thermolysin and neuraminidase. J Comput Aided Mol Des 13, 547–562 (1999). https://doi.org/10.1023/A:1008015827877
Issue Date:
DOI: https://doi.org/10.1023/A:1008015827877