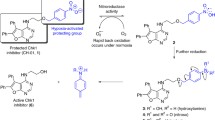
Abstract
Cyclophosphamides have been in clinical use as anti-cancer drugs for a long time and much research has been directed towards reducing their side effects. Here we have performed a theoretical investigation into the possibility of designing bioreductive analogues of cyclophosphamides. Our calculations have employed semiempirical molecular orbital AM1-SM2 and PM3-SM3 calculations, as implemented in MOPAC 93, which include a modified Born method for the treatment of solvation. We have investigated the effect of bioreductive activation on the β-elimination reaction that is central to the activation of cyclophosphamides. The approach was tested on two known bioreductive agents, including CB1954, and gave results in agreement with experiment. Non-local density functional calculations on CB1954 and its metabolites, including the radical anion, were in agreement with the semiempirical calculations. The calculations have identified a number of potentially novel bioreductive cyclophosphamides. In particular, our calculations identified compounds in which the initial one-electron reduction was not activating. Such compounds are likely to be more effective bioreductive agents, as the β-elimination will not compete under oxic conditions with the important re-oxidation required for the protection of oxic tissue.
Similar content being viewed by others
References
a. Sladek, N.E., Pharmacol. Ther., 37 (1988) 301.
Friedman, O.M., Myles, A. and Colvin, M., Adv. Cancer Chemother., 1 (1979) 143.
Borch, R.F. and Canute, G.W., J.Med. Chem., 34 (1991) 3044.
Moon, K.Y., Shirota, F.N., Baturay, N. and Kwon, C.H., J. Med. Chem., 38 (1995) 848.
Firestone, A., Mulcahy, R.T. and Borch, R.F., J. Med. Chem., 34 (1991) 2933.
Wilson, W.R., In: Waring, M.J. and Ponder, B.A.J. (Eds), Cancer Biology and Medicine, Vol. 3, Kluwer Academic Publishers, Lancaster, 1992, pp. 87-131.
Denny, W.A. and Wilson, W.R., Cancer Metastasis Rev., 12 (1993) 135.
Adams, G.E. and Stratford, I.J., In: Peckham, M.J., Pinedo, B. and Veronesi, U. (Eds.), Hypoxia-Selective Bioreductive Drugs, Oxford Textbook of Oncology, Vol. 1, Oxford University Press, Oxford, 1995, Section 4.28, p. 785.
Ware, D.C., Palmer, B.D., Wilson, W.R. and Denny, W.A., J. Med. Chem., 36 (1993) 1839.
Wilson, W.R., Moselen, J.W., Cliffe, S., Denny, W.A. and Ware, D.C., Int. J. Radiation Oncol. Biol. Phys., 29 (1994) 323.
Wu, J.H. and Reynolds, C.A., Anticancer Drugs Design, 11 (1996) 415.
Connors, T.A., Cox, P.J., Farmer, P.B., Foster, A.B. and Jarman, M., Biochem. Pharmac., 23 (1974) 115.
Borch, R.F., Hoye, T.R. and Swanson, T.A., J. Med. Chem., 27 (1984) 490.
Zon, G., Ludeman, S.M. and Bandt, J.A., J. Med. Chem., 27 (1984) 466.
Chang, T.K.H. and Waxman, D.J., Cancer Res., 53 (1993) 5629.
Cox, P.J., Biochem. Pharmacol., 28 (1979) 2045.
Andersson, B.S., Acta Oncologica, 34 (1995) 247.
Sladek, N.E., Cancer Res., 45 (1985) 1549.
Zon, G., Prog. Med. Chem., 19 (1982) 205.
Borch, R.F. and Valente, R.R., J. Med. Chem., 34 (1991) 3052.
Kaizer, H. and Stuart, R.K., Blood, 65 (1985) 1504.
Niemeyer, U., Engel, J. and Weigert, W., Invest. New Drugs, 2 (1984) 133.
Mulcahy, R.T., Gipp, J.J., Schmidt, J.P., Joswig, C. and Borch, R.F., J. Med. Chem., 37 (1994) 1610.
Wei, M.X., Tamiya, T., Rhee, R.J., Breakefield, X.O. and Chiocca, E.A., Clin. Cancer Res., 1 (1995) 1171.
Stratford, I.J., Williamson, C. and Adams, G.E., Radiat. Res., 88 (1981) 502.
Jarman, M. and Melzack, D.H., J. Biochem. Pharmacol., 25 (1976) 2475.
Dewar, M.J.S., Zoebisch, E.G., Healy, E.F. and Stewart, J.J.P., J. Am. Chem. Soc., 107 (1985) 3902.
Andres, J., Moliner, V., Krechl, J. and Silla, E., J. Phys. Chem., 98 (1994) 3664, and references therein.
Andzelm, J. and Wimmer, E.J., Chem. Phys., 96 (1992) 1280.
Becke, A.D., Phys. Rev. A, 38 (1988) 3098.
Lee, C., Yang, W. and Parr, R.G., Phys. Rev. B, 37 (1988) 785.
AMSOL version 3.0: Hawkins, G.D., Giesen, D.J., Chambers, G.C., Rossi, I., Storer, J.W., Rinaldi, D., Liotard, D.A., Cramer, C.J. and Truhlar, D.G., University of Minnesota, Minneapolis, MN.
Leach, A.R., Dolata, D. and Prout, K., J. Comput.-Aided Mol. Design, 4 (1990) 271.
Morley, S.D., Abraham, R.J., Haworth, I.S., Jackson, D.E., Saunders, M.R. and Vinter, J.G., J. Comput.-Aided Mol. Design, 5 (1991) 475.
Brown, J.M. and Zeman, E.M., Proc. Am. Ass. Cancer Res., 28 (1987) 1227.
Laderoute, K., Wardman, P. and Rauth, A.M., Biochem. Pharmacol., 37 (1988) 1487.
Carter, D.B. and Phillips, A.F., Nature (London), 174 (1954) 121.
Riley, R.J. and Workman, P., Biochem. Phamacol., 43 (1992) 1657.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Wu, J.H., Reynolds, C.A. Cyclophosphamides as hypoxia-activated diffusible cytotoxins: A theoretical study. J Comput Aided Mol Des 14, 307–316 (2000). https://doi.org/10.1023/A:1008128023564
Issue Date:
DOI: https://doi.org/10.1023/A:1008128023564