Abstract
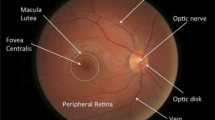
In today’s world blindness is a major concern in working population and diseases like glaucoma, diabetic retinopathy are main causes for this. Early and fast detection using automated software system can be a great help in this area. For that one major step is to detect and segment the optic disc (OD) in retinal fundus image. In this paper we have used U-Net based fully convolutional network to segment OD. U-Net is a very efficient architecture in image segmentation particularly in the area where availability of input images are very less. We have first trained U-Net from scratch on the extended Messidor dataset. It is then evaluated using three-fold cross validation on MESSIDOR image dataset. During the process we have removed false positives based on morphological operation and shape features. We have seen this method has outperformed existing techniques in OD segmentation on the images affected by diabetic retinopathy.
Supported by organization x.
You have full access to this open access chapter, Download conference paper PDF
Similar content being viewed by others
Keywords
1 Introduction
Throughout the world around 314 million people are suffering from Diabetic retinopathy, hypertensive retinopathy, glaucoma. These diseases gradually leads to vision loss of the patient which is a major area of concern in the developing counties [3]. Early identification and treatment can cure more than 85% visual impairments cases. In this field computer aided diagnosis system can make the process faster and assist ophthalmologists to cater more patients in less time.
There are different approaches followed in identification and segmentation of Optic Disc (OD). Welfer et al. [21] identified the OD boundary using morphological operations and watershed transformation technique. Aquino et al. [2] segmented the OD using morphological operations, edge detection method and circular Hough transformation technique. Morales et al. [17] applied inpainting as preprocessing for removing blood vessels and stochastic watershed transformation for determining the OD boundary. Xu et al. [22] applied active contour model (ACM) and proved better segmentation. Lowell et al. [13] applied a direction sensitive gradient-based technique to remove the vessel obstructions and deformable ACM for finding the OD boundary in low resulotion images. Chrastek et al. [6] applied distance map algorithm to remove the blood vessels and then segmented the OD by using sequence of methods like morphological operation, Hough transformation and ACM. The method presented by Joshi et al. [11] improved the robustness of ACM proposed by Chan and Vese [4] by taking care of the variations in the OD region. Morales et al. [17] detected the boundary of optic disc by the principal component analysis.
To overcome all these types of shortcomings deep learning based algorithms are playing an important role because of its ability to learn features during training time. The success of convolutional neural network in object segmentation [5, 7, 18] has motivated us to investigate the performance of fully convolutioal network for optic disc detection and segmentation. The major contributions of the present work are (i) initial segmentation of optic disc using U-Net based fully convolutional network and (ii) removal of false-positives based on anatomy-aware features.
In this paper, Sect. 1 has covered the existing works in this area. Then we have described the proposed segmentation framework in Sect. 2. Experimental results and comparison of the proposed method with the state-of-the-art techniques is provided in Sect. 3. Conclusion and future scope of work is stated in Sect. 4.
2 Segmentation Framework
In our proposed method (Fig. 1) U-net [19] based fully convolutional network has been used for initial segmentation after preprocessing of the input images. Then false positives have been reduced using anatomy-aware features. The U-Net architecture is used for initial segmentation as it provides better segmentation using few number of training images.
2.1 Preprocessing
First all the images are resized to \(512\times 512\) pixels. Then red channel image is threshold. Morphological opening, closing and erosion operations [8] with square structuring element are used to create a mask of circular retinal fundus region-of-interest, which allows focusing only on the foreground of retinal images.
2.2 Segmentation Using U-Net
Architecture of U-Net. The U-net [19] is a fully convolutional network which consists of convolution operation for down-sampling, max pooling, ReLU operation, concatenation and de-Convolution operation for up-sampling. The down-sampling path has 5 convolutional blocks and each block has two convolutional layers with a filter size of \(3\times 3\) and stride of 1. Max pooling with stride 2 is applied to the end of every blocks except the last block. The data is propagated through the network along all possible paths and generates the segmentation map at the end of the network. At the end input images of \(512\times 512\) size reduces to \(32\times 32\). The second part of the U-Net is the expansion layer which basically create the high resolution segmentation map. This part consists of a sequence of up-convolutions and concatenation with high-resolution features from contraction path. Therefore, the size of feature maps increases from \(32\times 32\) to \(512\times 512\). High-level information is represented at up-sampling blocks, and low-level features are transferred through skip connection.
Training of U-Net. First data augmentation techniques have been applied on the images of extended MESSIDOR database (MESSIDOR-II) [16] which is then used for training of U-Net from scratch. A stochastic gradient-based optimization [12] (ADAM) is applied to minimize the cross-entropy based cost function. The learning rate for the ADAM optimizer is set to 0.0001 and over-fitting is reduced by using dropout [10]. The weights of background and foreground are maintained as 1:10 and training were performed upto 60, 000 iterations.
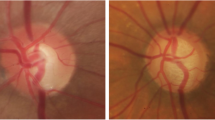
False Positive Removal. Morphological opening is applied to separate false positives from Optic disc. E.g., for Fig. 2(a), after initial segmentation Fig. 2(b) is showing some false positives caused by exudates. Compactness feature is used to eliminate false positive candidates from initial segmentation results which will create two objects. The object having bigger size is considered as optic disc Fig. 2(c).
3 Experimental Results and Discussions
3.1 Database Used for Evaluation of Segmentation Result
MESSIDOR. MESSIDOR [15] database contains 1200 colour retinal images, acquired using non-mydriatic camera, Topcon TRC NW6 with field-of-view set to 45\(^ \circ \). Binary mask of the optic disc of MESSIDOR dataset was provided by the experts of the University of Huelva [1].
3.2 Evaluation
The performance of optic disc detection is evaluated using Success Rate (SR) which represents the percentage of retinal images in a dataset where the centroid of optic disc is successfully localized within the boundary of the ground truth mask of optic disc. The performance of optic disc segmentation is evaluated in terms of Overlap Measure (OM) and Mean Absolute Distance (MAD) [20]. The OM represents the ratio of the intersecting area between the actual optic disc and segmented optic disc. MAD represents the mean of the shortest distances from the boundary of the actual optic disc to the boundary of the segmented optic disc.
3.3 Experimental Results
Quantitative Analysis. The evaluation of the proposed segmentation algorithm is performed on MESSIDOR datasets. During testing, we divided the images into three subsets. Out of three subsets, two are used for fine tuning of pre-trained network and remaining set is used for testing. Thus U-Net learns database specific features through transfer learning. The average value of SR, OM and MAD of the proposed framework and competing techniques are provided in Table 1. The OM of the proposed method is larger as compared to the competing techniques. Such improvement of OM is due to the application of fully convolutional network in initial segmentation.
There is only one failure case for optic disc detection for the MESSIDOR dataset. The MAD value of the proposed method is slightly better or comparable with the competing techniques. The high value of OM depict that the segmented mask of optic disc matches accurately with the ground truth mask. A comparison of segmentation performance in terms of percentage of test images included in various OM distributions, is provided in Table 2. The proposed method outperforms the competing techniques at all the four different OM levels such as \(\ge \) 0.7, 0.75, 0.85 and 0.9.
Qualitative Results. We have analysed the proposed framework for images of healthy subjects [Fig. 3(a)–(c)], the images with the presence of pathologies [Fig. 4(a)–(c)], and low contrast [Fig. 4(b)]. These qualitative results reveal that the proposed algorithm is capable of identifying and segmenting the optic disc in bad quality retinal images. Few images with poor segmentation results is shown in Fig. 5(a)–(c). The poor segmentation is due to non-uniform illumination during image acquisition.
3.4 Implementation
The U-Net architecture is implemented in Python using the PyTorch library in Linux environment using a 8 GB GPU (NVIDIA GeForce GTX 1070 with 8GB GDDR5 memory) on a system with Core-i7 processor and 32 GB RAM.
4 Conclusion
In the proposed method, fully convolutional network is trained by feeding thousands of varying grades of fundus images, where it is learns the best features on its own. Therefore, the proposed method outperforms the other competing techniques in most of the metrics measurements. The method is also successful in optic disc localization and segmentation, when tested on both dilated and non-dilated types of fundus images. The performance of this algorithm does not degrade while handling images containing strong distractors like yellowish exudates which prove the effectiveness and robustness of the proposed process. In future more research needs to be accomplished by testing on other independent datasets.
References
Expert system for early automated detection of DR by analysis of digital retinal images. http://www.uhu.es/retinopathy/muestr2as2.php
Aquino, A., Gegúndez-Arias, M.E., Marín, D.: Detecting the optic disc boundary in digital fundus images using morphological, edge detection, and feature extraction techniques. IEEE Trans. Med. Imaging 29(11), 1860–1869 (2010)
Bourne, R.R., et al.: Magnitude, temporal trends, and projections of the global prevalence of blindness and distance and near vision impairment: a systematic review and meta-analysis. Lancet Glob. Health 5(9), e888–e897 (2017)
Chan, T.F., Vese, L.A.: Active contours without edges. IEEE Trans. Image Process. 10(2), 266–277 (2001)
Chen, L.C., Papandreou, G., Kokkinos, I., Murphy, K., Yuille, A.L.: Deeplab: semantic image segmentation with deep convolutional nets, atrous convolution, and fully connected CRFS. arXiv preprint arXiv:1606.00915 (2016)
Chrstek, R., et al.: Med. Image Anal. Automated segmentation of the optic nerve head for diagnosis of glaucoma. 9(4), 297–314 (2005)
Cordts, M., et al.: The cityscapes dataset for semantic urban scene understanding. In: Proceedings of the IEEE Conference on Computer Vision and Pattern Recognition, pp. 3213–3223 (2016)
Gagnon, L., Lalonde, M., Beaulieu, M., Boucher, M.C.: Procedure to detect anatomical structures in optical fundus images. In: Medical Imaging 2001: Image Processing, vol. 4322, pp. 1218–1226. International Society for Optics and Photonics (2001)
Giachetti, A., Ballerini, L., Trucco, E.: Accurate and reliable segmentation of the optic disc in digital fundus images. J. Med. Imaging 1(2), 024001–024001 (2014)
Hinton, G.E., Srivastava, N., Krizhevsky, A., Sutskever, I., Salakhutdinov, R.R.: Improving neural networks by preventing co-adaptation of feature detectors. arXiv preprint arXiv:1207.0580 (2012)
Joshi, G.D., Sivaswamy, J., Krishnadas, S.: Optic disk and cup segmentation from monocular color retinal images for glaucoma assessment. IEEE Trans. Med. imaging 30(6), 1192–1205 (2011)
Kingma, D., Ba, J.: Adam: a method for stochastic optimization. arXiv preprint arXiv:1412.6980 (2014)
Lowell, J., et al.: Optic nerve head segmentation. IEEE Trans. Med. Imaging 23(2), 256–264 (2004)
Marin, D., Gegundez-Arias, M.E., Suero, A., Bravo, J.M.: Obtaining optic disc center and pixel region by automatic thresholding methods on morphologically processed fundus images. Comput. Methods Programs Biomed. 118(2), 173–185 (2015)
MESSIDOR: Messidor. digital retinal images, messidor techno-vision project, France. http://messidor.crihan.fr/download-en.php (download images section), May 2014
MESSIDOR-2: Messidor-2. digital retinal images, latim laboratory and the messidor program partners (2010). http://latim.univ-brest.fr/, http://messidor.crihan.fr/
Morales, S., Naranjo, V., Angulo, J., Alcañiz, M.: Automatic detection of optic disc based on pca and mathematical morphology. IEEE Trans. Med. Imaging 32(4), 786–796 (2013)
Papandreou, G., Kokkinos, I., Savalle, P.A.: Modeling local and global deformations in deep learning: epitomic convolution, multiple instance learning, and sliding window detection. In: 2015 IEEE Conference on Computer Vision and Pattern Recognition (CVPR), pp. 390–399. IEEE (2015)
Ronneberger, O., Fischer, P., Brox, T.: U-Net: convolutional networks for biomedical image segmentation. In: Navab, N., Hornegger, J., Wells, W.M., Frangi, A.F. (eds.) MICCAI 2015. LNCS, vol. 9351, pp. 234–241. Springer, Cham (2015). https://doi.org/10.1007/978-3-319-24574-4_28
Roychowdhury, S., Koozekanani, D.D., Kuchinka, S.N., Parhi, K.K.: Optic disc boundary and vessel origin segmentation of fundus images. IEEE J. Biomed. Health Inform. 20(6), 1562–1574 (2016)
Welfer, D., Scharcanski, J., Kitamura, C.M., Dal Pizzol, M.M., Ludwig, L.W., Marinho, D.R.: Segmentation of the optic disk in color eye fundus images using an adaptive morphological approach. Comput. Biol. Med. 40(2), 124–137 (2010)
Xu, C., Prince, J.L.: Snakes, shapes, and gradient vector flow. IEEE Trans. Image Process. 7(3), 359–369 (1998)
Yu, H., et al.: Fast localization and segmentation of optic disk in retinal images using directional matched filtering and level sets. IEEE Trans. Inf. Technol. Biomed. 16(4), 644–657 (2012)
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2018 Springer Nature Switzerland AG
About this paper
Cite this paper
Sadhukhan, S., Ghorai, G.K., Maiti, S., Karale, V.A., Sarkar, G., Dhara, A.K. (2018). Optic Disc Segmentation in Retinal Fundus Images Using Fully Convolutional Network and Removal of False-Positives Based on Shape Features. In: Stoyanov, D., et al. Deep Learning in Medical Image Analysis and Multimodal Learning for Clinical Decision Support. DLMIA ML-CDS 2018 2018. Lecture Notes in Computer Science(), vol 11045. Springer, Cham. https://doi.org/10.1007/978-3-030-00889-5_42
Download citation
DOI: https://doi.org/10.1007/978-3-030-00889-5_42
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-00888-8
Online ISBN: 978-3-030-00889-5
eBook Packages: Computer ScienceComputer Science (R0)