Abstract
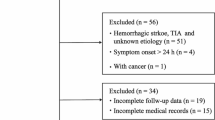
Stroke is an acute disorder of CNS being the leading factor of mortality and disability of the population. Dynamic assessment of trophic growth factors expression is a promising tool to predict the outcome of ischemic stroke. We investigated the concentration dynamics of the brain-derived neurotrophic factor (BDNF) and vascular endothelial growth factor (VEGF) in blood plasma of patients with acute ischemic stroke. 56 patients took part in the study. Venous blood was collected from all patients on the first, 7th and 21st day of their hospital stay. BDNF and VEGF plasma concentrations were measured using ELISA. Our study shows, that not single, but serial dynamic measures of BDNF plasma concentrations in the acute period of ischemic stroke have a prognostic significance. Increasing of the BDNF plasma concentration on day 7 in comparison to the concentration on day 1 was significantly associated with a better clinical outcome of acute ischemic stroke. Extremely high VEGF plasma concentrations (more than 260 pg/mL) on days 1 and 7 from the ischemic stroke onset were significantly associated with a worse clinical outcome on day 21 and a less favorable rehabilitation prognosis. Serial measurement of plasma concentrations of trophic growth factors in patients with ischemic stroke presents a rather simple, reliable and minimally invasive method of dynamic assessment of the clinical course of acute ischemic stroke and early outcome prediction.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kim, G., Kim, E.: The effects of antecedent exercise on motor function recovery and brain-derived neurotrophic factor expression after focal cerebral ischemia in rats. J. Phys. Ther. Sci. 25(5), 553–556 (2013)
Skvortsova, V., Evzelman, M.: Ischemic stroke, p. 404 (2006). (in Russian)
Emanueli, C., Schratzberger, P., Kirchmair, R., Madeddu, P.: Paracrine control of vascularization and neurogenesis by neurotrophins. Br. J. Pharmacol. 140(4), 614–619 (2003)
Huang, E.J., Reichardt, L.F.: Neurotrophins: roles in neuronal development and function. Annu. Rev. Neurosci. 24(1), 677–736 (2001)
Roux, P.P., Barker, P.A.: Neurotrophin signaling through the p75 neurotrophin receptor. Prog. Neurobiol. 67(3), 203–233 (2002)
Casoli, T., Giuli, C., Balietti, M., Giorgetti, B., Solazzi, M., Fattoretti, P.: Effect of cognitive training on the expression of brain-derived neurotrophic factor in lymphocytes of mild cognitive impairment patients. Rejuvenation Res. 17(2), 235–238 (2014)
Sartorius, A., et al.: Correlations and discrepancies between serum and brain tissue levels of neurotrophins after electroconvulsive treatment in rats. Pharmacopsychiatry 42(06), 270–276 (2009)
Gottlieb, M., Bonova, P., Danielisova, V., Nemethova, M., Burda, J., Cizkova, D.: Brain-derived neurotrophic factor blood levels in two models of transient brain ischemia in rats. Gen. Physiol. Biophys. 32, 139–142 (2013)
Wang, J., et al.: Low serum levels of brain-derived neurotrophic factor were associated with poor short-term functional outcome and mortality in acute ischemic stroke. Mol. Neurobiol. 54(9), 7335–7342 (2017). https://doi.org/10.1007/s12035-016-0236-1
Rodier, M., et al.: Relevance of post-stroke circulating BDNF levels as a prognostic biomarker of stroke outcome. Impact of rt-PA treatment. PLoS ONE 10(10), e0140668 (2015)
Berretta, A., Tzeng, Y.C., Clarkson, A.N.: Post-stroke recovery: the role of activity-dependent release of brain-derived neurotrophic factor. Expert. Rev. Neurother. 14(11), 1335–1344 (2014)
Madinier, A., et al.: Ipsilateral versus contralateral spontaneous post-stroke neuroplastic changes: involvement of BDNF? Neuroscience 231, 169–181 (2013)
Clarkson, A.N., Overman, J.J., Zhong, S., Mueller, R., Lynch, G., Carmichael, S.T.: AMPA receptor-induced local brain-derived neurotrophic factor signaling mediates motor recovery after stroke. J. Neurosci. 31(10), 3766–3775 (2011)
Schäbitz, W., et al.: Intravenous brain-derived neurotrophic factor reduces infarct size and counterregulates Bax and Bcl-2 expression after temporary focal cerebral ischemia. Stroke 31(9), 2212–2216 (2000)
Schäbitz, W.R., et al.: Intravenous brain-derived neurotrophic factor enhances poststroke sensorimotor recovery and stimulates neurogenesis. Stroke 38(7), 2165–2172 (2007)
Di Lazzaro, V., et al.: BDNF plasma levels in acute stroke. Neurosci. Lett. 422(2), 128–130 (2007)
Béjot, Y., Mossiat, C., Giroud, M., Prigent-Tessier, A., Marie, C.: Circulating and brain BDNF levels in stroke rats. Relevance to clinical studies. PLoS ONE 6(12), e29405 (2011)
Sun, J., Ke, Z., Yip, S.P., Hu, X.l., Zheng, X.x., Tong, K.y.: Gradually increased training intensity benefits rehabilitation outcome after stroke by BDNF upregulation and stress suppression. BioMed Res. Int. 2014, 8 (2014). https://doi.org/10.1155/2014/925762. Article ID 925762. PMCID: PMC4090448. PMID: 25045713
Senger, D.R., Galli, S.J., Dvorak, A.M., Perruzzi, C.A., Harvey, V.S., Dvorak, H.F.: Tumor cells secrete a vascular permeability factor that promotes accumulation of ascites fluid. Science 219(4587), 983–985 (1983)
Mărgăritescu, O., Pirici, D., Mărgăritescu, C.: VEGF expression in human brain tissue after acute ischemic stroke. Rom. J. Morphol. Embryol. 52(4), 1283–1292 (2011)
Zhang, Z.G., et al.: VEGF enhances angiogenesis and promotes blood-brain barrier leakage in the ischemic brain. J. Clin. Investig. 106(7), 829–838 (2000)
Gerber, H.P., et al.: Vascular endothelial growth factor regulates endothelial cell survival through the phosphatidylinositol 32-kinase/Akt signal transduction pathway requirement for Flk-1/KDR activation. J. Biol. Chem. 273(46), 30336–30343 (1998)
Pedram, A., Razandi, M., Levin, E.R.: Extracellular signal-regulated protein kinase/Jun kinase cross-talk underlies vascular endothelial cell growth factor-induced endothelial cell proliferation. J. Biol. Chem. 273(41), 26722–26728 (1998)
Hirashima, M.: Regulation of endothelial cell differentiation and arterial specification by VEGF and Notch signaling. Anat. Sci. Int. 84(3), 95–101 (2009). https://doi.org/10.1007/s12565-009-0026-1
Sentilhes, L., et al.: Vascular endothelial growth factor and its high-affinity receptor (VEGFR-2) are highly expressed in the human forebrain and cerebellum during development. J. Neuropathol. Exp. Neurol. 69(2), 111–128 (2010)
Svensson, B., et al.: Vascular endothelial growth factor protects cultured rat hippocampal neurons against hypoxic injury via an antiexcitotoxic, caspase-independent mechanism. J. Cereb. Blood Flow Metab. 22(10), 1170–1175 (2002)
Yang, J., Yao, Y., Chen, T., Zhang, T.: VEGF ameliorates cognitive impairment in in vivo and in vitro ischemia via improving neuronal viability and function. Neuromolecular Med. 16(2), 376–388 (2014). https://doi.org/10.1007/s12017-013-8284-4
Zhang, R., Zhang, Z., Zhang, L., Chopp, M.: Proliferation and differentiation of progenitor cells in the cortex and the subventricular zone in the adult rat after focal cerebral ischemia. Neuroscience 105(1), 33–41 (2001)
Liu, F., Ni, J.J., Huang, J.J., Kou, Z.W., Sun, F.Y.: VEGF overexpression enhances the accumulation of phospho-S292 MeCP2 in reactive astrocytes in the adult rat striatum following cerebral ischemia. Brain Res. 1599, 32–43 (2015)
Li, W.L., Fraser, J.L., Shan, P.Y., Zhu, J., Jiang, Y.J., Wei, L.: The role of VEGF/VEGFR2 signaling in peripheral stimulation-induced cerebral neurovascular regeneration after ischemic stroke in mice. Exp. Brain Res. 214(4), 503 (2011). https://doi.org/10.1007/s00221-011-2849-y
Clayton, J.A., Chalothorn, D., Faber, J.E.: Vascular endothelial growth factor-a specifies formation of native collaterals and regulates collateral growth in ischemia. Circ. Res. 103(9), 1027–1036 (2008)
Cao, L., et al.: VEGF links hippocampal activity with neurogenesis, learning and memory. Nat. Genet. 36(8), 827 (2004)
Sluimer, J.C., Daemen, M.J.: Novel concepts in atherogenesis: angiogenesis and hypoxia in atherosclerosis. J. Pathol. J. Pathol. Soc. Gt. Br. Irel. 218(1), 7–29 (2009)
van Bruggen, N., et al.: VEGF antagonism reduces edema formation and tissue damage after ischemia/reperfusion injury in the mouse brain. J. Clin. Investig. 104(11), 1613–1620 (1999)
Li, Y.N., Pan, R., Qin, X.J., Yang, W.L., Qi, Z., Liu, W., Liu, K.J.: Ischemic neurons activate astrocytes to disrupt endothelial barrier via increasing vegf expression. J. Neurochem. 129(1), 120–129 (2014)
Gunsilius, E., Petzer, A.L., Stockhammer, G., Kähler, C.M., Gastl, G.: Serial measurement of vascular endothelial growth factor and transforming growth factor-beta1 in serum of patients with acute ischemic stroke. Stroke 32(1), 275–278 (2001)
Slevin, M., Krupinski, J., Slowik, A., Kumar, P., Szczudlik, A., Gaffney, J.: Serial measurement of vascular endothelial growth factor and transforming growth factor-\(\beta \)1 in serum of patients with acute ischemic stroke. Stroke 31(8), 1863–1870 (2000)
Lee, S.C., Lee, K.Y., Kim, Y.J., Kim, S.H., Koh, S.H., Lee, Y.J.: Serum VEGF levels in acute ischaemic strokes are correlated with long-term prognosis. Eur. J. Neurol. 17(1), 45–51 (2010)
Gonchar, I., Prudyvus, I., Stepanova, J.: Expression of vascular endothelial growth factor in patients with acute ischemic stroke. Zhurnal nevrologii i psihiatrii 113(3), 25–29 (2013)
Gonchar, I., Stepanova, J.: Vascular endothelial growth factor in patients with non-cardioembolic brain infarction with mild atherosclerotic damage of cerebral arteries. Voennaya medicina 21(4), 36–39 (2011)
Adams Jr., H.P., et al.: Classification of subtype of acute ischemic stroke. Definitions for use in a multicenter clinical trial. TOAST. Trial of org 10172 in acute stroke treatment. Stroke 24(1), 35–41 (1993)
Navarro-Sobrino, M., et al.: A large screening of angiogenesis biomarkers and their association with neurological outcome after ischemic stroke. Atherosclerosis 216(1), 205–211 (2011)
Okazaki, H., Beppu, H., Mizutani, K., Okamoto, S., Sonoda, S.: Changes in serum growth factors in stroke rehabilitation patients and their relation to hemiparesis improvement. J. Stroke Cerebrovasc. Dis. 23(6), 1703–1708 (2014)
Fukunaga, K.: Introduction to Statistical Pattern Recognition. Academic Press, London (1990)
Gorban, A.N., Zinovyev, A.Y.: Fast and user-friendly non-linear principal manifold learning by method of elastic maps. In: 2015 IEEE International Conference on Data Science and Advanced Analytics, DSAA 2015, Campus des Cordeliers, Paris, France, 19–21 October 2015, pp. 1–9 (2015)
Acknowledgements
The work was supported by the State Assignment for Research issued by the Ministry of Public Health of the Russian Federation (2018–2020).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2020 Springer Nature Switzerland AG
About this paper
Cite this paper
Roslavtceva, V. et al. (2020). Blood Plasma Trophic Growth Factors Predict the Outcome in Patients with Acute Ischemic Stroke. In: Rojas, I., Valenzuela, O., Rojas, F., Herrera, L., Ortuño, F. (eds) Bioinformatics and Biomedical Engineering. IWBBIO 2020. Lecture Notes in Computer Science(), vol 12108. Springer, Cham. https://doi.org/10.1007/978-3-030-45385-5_3
Download citation
DOI: https://doi.org/10.1007/978-3-030-45385-5_3
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-030-45384-8
Online ISBN: 978-3-030-45385-5
eBook Packages: Computer ScienceComputer Science (R0)