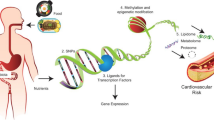
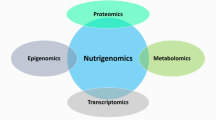
Abstract
TP53 is one of the tumor suppressor genes found to be highly correlated with human tumor development, this gene can sense stress or damage to cells and prevent cell division or trigger cell death, thereby preventing the proliferation of damaged cells. The P53 protein encoded by TP53 has an anti-tumor effect and is known as the “guardian of the genome”. The mutation of TP53 gene eliminates a key cell safety mechanism, making it the trigger of cancer. In this paper, we first described the relationship between TP53 gene and tumors, and discussed the application of TP53 gene in tumor prediction and treatment, then we developed a TP53 genetic testing service to evaluate the cancer suppression ability of tumors, so that to help individuals to establish a scientific and reasonable lifestyle in a timely manner, and better grasp the initiative of health. Further, we describe the interaction between TP53 gene and nutrition and its impact on the occurrence and development of cancer. Finally, based on the analysis of the genetic testing results and food frequency questionnaires, we developed a personalized nutrition service to reduce the risk of developing the diseases with high genetic risk score.
Access this chapter
Tax calculation will be finalised at checkout
Purchases are for personal use only
Similar content being viewed by others
References
Kaur, R.P., Vasudeva, K., Kumar, R., Munshi, A.: Role of p53 gene in breast cancer: focus on mutation spectrum and therapeutic strategies. Curr. Pharm. Des. 24(30), 3566–3575 (2018)
DeLeo, A.B., Jay, G., Appella, E., et al.: Detection of a transformation-related antigen in chemically induced sarcomas and other transformed cells of the mouse. Proc. Natl. Acad. Sci. U.S.A. 76(5), 2420–4 (1979)
Levine, A.J., Oren, M.: The first 30 years of p53: growing ever more complex. Nat. Rev. Cancer 9(10), 749–58 (2009)
Finlay, C.A., Hinds, P.W., Levine, A.J.: The p53 proto-oncogene can act as a suppressor of transformation. Cell 57, 1083–1093 (1989)
Baugh, E.H., Ke, H., Levine, A.J., et al.: Why are there hotspot mutations in the TP53 gene in human cancers? Cell Death Differ. 25(1), 154–160 (2018)
Lane, D.P.: p53, guardian of the genome. Nature 358(6381), 15–6 (1992)
Donehower, L.A., Soussi, T., Korkut, A., et al.: Integrated analysis of TP53 gene and pathway alterations in the cancer genome atlas. Cell Rep. 28(5), 1370-1384.e5 (2019)
Schneider, K., Zelley, K., Nichols, K.E., Garber, J.: Li-Fraumeni syndrome. In: Adam, M.P., et al. (eds.) GeneReviews [Internet]. University of Washington, Seattle, Seattle (WA) (1999). Accessed 21 Nov 2019
Olivier, M., Hollstein, M., Hainaut, P.: TP53 mutations in human cancers: origins, consequences, and clinical use. Cold Spring Harb. Perspect. Biol. 2(1), a001008 (2010)
Petitjean, A., Mathe, E., Kato, S., et al.: Impact of mutant p53 functional properties on TP53 mutation patterns and tumor phenotype: lessons from recent developments in the IARC TP53 database. Hum. Mutat. 28(6), 622–9 (2007)
Wang, Y., Helland, A., Holm, R., et al.: TP53 mutations in early-stage ovarian carcinoma, relation to long-term survival. Br. J. Cancer 90, 678–685 (2004)
Langerod, A., Zhao, H., Borgan, O., et al.: TP53 mutation status and gene expression profiles are powerful prognostic markers of breast cancer. Breast Cancer Res. 9, R30 (2007)
Li, F.P., Fraumeni, J.F.J., et al.: A cancer family syndrome in twenty-four kindreds. Cancer Res. 48, 5358–5362 (1988)
Olivier, M., Goldgar, D.E., Sodha, N., et al.: Li-Fraumeni and related syndromes: correlation between tumor type, family structure, and TP53 genotype. Cancer Res. 63, 6643–6650 (2003)
Lalloo, F., Varley, J., Moran, A., et al.: BRCA1, BRCA2 and TP53 mutations in very early-onset breast cancer with associated risks to relatives. Eur. J. Cancer 42, 1143–1150 (2006)
Gonzalez, K.D., Noltner, K.A., Buzin, C.H., et al.: Beyond Li Fraumeni syndrome: clinical characteristics of families with p53 germline mutations. J. Clin. Oncol. 27, 1250–1256 (2009)
Shi, H., Tan, S.J., Zhong, H., et al.: Winter temperature and UV are tightly linked to genetic changes in the p53 tumor suppressor pathway in Eastern Asia. Am. J. Hum. Genet. 84, 534–541 (2009)
Mohammed Basabaeen, A.A., Abdelgader, E.A., Babekir, E.A., et al.: TP53 Gene 72 Arg/Pro (rs1042522) single nucleotide polymorphism contribute to increase the risk of B-Chronic lymphocytic leukemia in the Sudanese population. Asian Pac. J. Cancer Prev. 20(5), 1579–1585 (2019)
Zhang, L., Wang, Y., Qin, Z., et al.: TP53 codon 72 Polymorphism and bladder cancer risk: a meta-analysis and emphasis on the role of tumor or smoking status. J. Cancer 9(19), 3522–3531 (2018)
Lu, Y., et al.: Association of p53 codon 72 polymorphism with prostate cancer: an update meta-analysis. Tumor Biol. 35(5), 3997–4005 (2014). https://doi.org/10.1007/s13277-014-1657-y
Wang, X., Liu, Z.: Systematic meta-analysis of genetic variants associated with osteosarcoma susceptibility. Medicine (Baltimore) 97(38), e12525 (2018)
Naccarati, A., Pardini, B., Polakova, V., et al.: Genotype and haplotype analysis of TP53 gene and the risk of pancreatic cancer: an association study in the Czech Republic. Carcinogenesis 31(4), 666–70 (2010)
Cao, J., Chen, Z., Tian, C., et al.: A shared susceptibility locus in the p53 gene for both gastric and esophageal cancers in a northwestern Chinese population. Genet. Test. Mol. Biomarkers 24(12), 804–811 (2020)
Li, Y., Chang, S.C., Niu, R., et al.: TP53 genetic polymorphisms, interactions with lifestyle factors and lung cancer risk: a case control study in a Chinese population. BMC Cancer 13, 607 (2013)
Egan, K.M., Nabors, L.B., Olson, J.J., et al.: Rare TP53 genetic variant associated with glioma risk and outcome. J. Med. Genet. 49(7), 420–1 (2012)
Guan, X., Wang, L.E., Liu, Z., et al.: Association between a rare novel TP53 variant (rs78378222) and melanoma, squamous cell carcinoma of head and neck and lung cancer susceptibility in non-Hispanic Whites. J. Cell Mol. Med. 17(7), 873–8 (2013)
Schildkraut, J.M., Iversen, E.S., Wilson, M.A., et al.: Association between DNA damage response and repair genes and risk of invasive serous ovarian cancer. PLoS ONE 5(4), e10061 (2010)
Wang, S., Zhang, K., Tang, L., et al.: Association between single-nucleotide polymorphisms in breast cancer susceptibility genes and clinicopathological characteristics. Clin. Epidemiol. 13, 103–112 (2021)
Zhao, Z., Wan, J., Guo, M., et al.: Expression and prognostic significance of m6A-related genes in TP53-mutant non-small-cell lung cancer. J. Clin. Lab. Anal. 36(1), e24118 (2022)
Seemann, S., Maurici, D., Olivier, M., et al.: The tumor suppressor gene TP53: implications for cancer management and therapy. Crit. Rev. Clin. Lab. Sci. 41(5–6), 551–83 (2004)
Igo, R.P., Jr., Kinzy, T.G., Cooke Bailey, J.N.: Genetic risk scores. Curr. Protoc. Hum. Genet. 104(1), e95 (2019)
Ruan, Y., Lin, Y., Feng, Y., et al.: Improving polygenic prediction in ancestrally diverse populations. Nat. Genet. 54, 57–3580 (2022)
Perri, F., Pisconti, S., Scarpati, G.D.V.: p53 mutations and cancer: a tight linkage. Ann. Transl. Med. 4(24), 522 (2016)
Levine, A.J.: p53: 800 million years of evolution and 40 years of discovery. Nat. Rev. Cancer 20, 471–480 (2020)
Xiong, Y., Xu, S., Fu, B., et al.: Vitamin C-induced competitive binding of HIF-1\(\alpha \) and p53 to ubiquitin E3 ligase CBL contributes to anti-breast cancer progression through p53 deacetylation. Food Chem. Toxicol. 168, 113321 (2022)
Kim, J., Lee, S.D., Chang, B., et al.: Enhanced antitumor activity of vitamin C via p53 in cancer cells. Free Radic. Biol. Med. 53(8), 1607–15 (2012)
Li, M., Li, L., Zhang, L., et al.: 1,25-Dihydroxyvitamin D3 suppresses gastric cancer cell growth through VDR- and mutant p53-mediated induction of p21. Life Sci. 15(179), 88–97 (2017)
Reichrath, J., Reichrath, S., Vogt, T., Romer, K.: Crosstalk between Vitamin D and p53 signaling in cancer: an update. Adv. Exp. Med. Biol. 1268, 307–318 (2020)
Stambolsky, P., Tabach, Y., Fontemaggi, G.: Modulation of the vitamin D3 response by cancer-associated mutant p53. Cancer Cell 17(3), 273–85 (2010)
Liu, T., Yang, H., Mou, Y., Zhang, H.: Correlation of changes in HIF-1\(\alpha \) and p53 expressions with vitamin B3 deficiency in skin cancer patients. G. Ital. Dermatol. Venereol. 154(5), 513–518 (2019)
Affret, A., El Fatouhi, D., Dow, C., et al.: Relative validity and reproducibility of a new 44-item diet and food frequency questionnaire among adults: online assessment. J. Med. Internet Res. 20(7), e227 (2018)
Acknowledgment
This research project is supported by Science Foundation of Beijing Language and Culture University (supported by “the Fundamental Research Funds for the Central Universities”) (Approval number: 23YJ080003).
Author information
Authors and Affiliations
Corresponding author
Editor information
Editors and Affiliations
Rights and permissions
Copyright information
© 2024 The Author(s), under exclusive license to Springer Nature Switzerland AG
About this paper
Cite this paper
Yang, J. (2024). TP53 Genetic Testing and Personalized Nutrition Service. In: Florez, H., Leon, M. (eds) Applied Informatics. ICAI 2023. Communications in Computer and Information Science, vol 1874. Springer, Cham. https://doi.org/10.1007/978-3-031-46813-1_23
Download citation
DOI: https://doi.org/10.1007/978-3-031-46813-1_23
Published:
Publisher Name: Springer, Cham
Print ISBN: 978-3-031-46812-4
Online ISBN: 978-3-031-46813-1
eBook Packages: Computer ScienceComputer Science (R0)