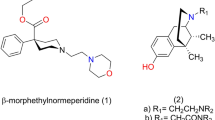
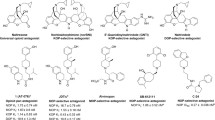
Summary
Extensive conformational calculations were performed on the potent opiate analgesics etorphine, PET, R30490 and etonitazene to determine all of their many low-energy conformations. The results were used to characterize four possible models for binding of a simple pharmacophore, comprising two phenyl rings plus a protonated nitrogen, to opiate analgesic receptors. These four models may define the necessary three-dimensional features leading to particular opiate actions. The model favoured for μ receptor activity can accommodate a protonated nitrogen, an aromatic ring (which may be substituted with an electronegative group) and a second lipophilic group. These structural features must be presented in a precise three-dimensional arrangement. It appears likely that a hydrophilic substituent in a certain region of the analgesic pharmacophore may also interact with the receptor as a secondary binding group.
Similar content being viewed by others
References
Andrews, P.R. and Lloyd, E.J., J. Pharm. Pharmacol., 35 (1983) 516–518.
Lloyd, E.J. and Andrews, P.R., J. Med. Chem., 29 (1986) 453–462.
Andrews, P.R., Craik, D.J. and Munro, S.L., Quant. Struct.-Act Relat. (in press).
Andrews, P.R., Lloyd, E.J., Martin, J.L. and Munro, S.L.A., J. Mol. Graph. 4 (1986) 41–45.
Beckett, A.H. and Casy, A.F., J. Pharm. Pharmacol., 6 (1954) 986–1001.
Portoghese, P.S., J. Med. Chem., 8, (1965) 609–616.
Portoghese, P.S., J. Pharm. Sci. 55 (1966) 865–887.
Feinberg, A.P., Creese, I. and Snyder, S.H., Proc. Natl. Acad. Sci. U.S.A., 73 (1976) 4215–4219.
Humblet, C. and Marshall, G.R., Drug Dev. Res., 1 (1981) 409–434.
Gorin, F.A. and Marshall, G.R., Proc. Natl. Acad. Sci. U.S.A., 74 (1977) 5179–5183.
Clarke, F.H., Jaggi, H. and Lovell, R.A., J. Med. Chem., 21 (1978) 600–606.
Loew, G.H. and Burt, S.K., Proc. Natl. Acad. Sci. U.S.A., 75 (1978) 7–11.
Horn, A.S. and Rodgers, J.R., Nature (London), 260 (1976) 795–797.
Horn, A.S. and Rodgers, J.R., J. Pharm. Pharmacol., 29 (1977) 257–265.
Koch, M.H.J., Acta Cryst., B29 (1973) 379–382.
Allinger, N.L., Quantum Chemistry Program Exchange, Program 318 (1976).
Allinger, N.L. and Yuh, Y.H., Quantum Chemistry Program Exchange, Program 395 (1980).
Klyne, W. and Prelog, V., Experientia, 16 (1960) 521–523.
Palmer, J.A.B., Aust. Comp. J., 2 (1970) 27–31.
Sutton, L.E. (Ed.), Tables of Interatomic Distances and Configurations in Molecules and Ions, Chemical Society Special Publications, No. 11, 1958, No. 18, 1965.
Olson, W.K., Biopolymers, 15 (1976) 859–878.
Taylor, R., Kennard, O. and Versichel, W., Acta Cryst., B40 (1984) 280–288.
Schulz, G.E. and Schirmer, R.E., Principles of Protein Structure, Springer-Verlag, New York, 1979, pp. 27–45.
Koch, M.H.J., De Ranter, C.J., Rolies, M. and Dideberg, O., Acta Cryst., B32 (1976) 2529–2531.
Allinger, N.L., Quantum Chemistry Program Exchange Bulletin, 3 (1983) 32–33.
Karlsson, S., Liljefors, T. and Sandstrom, J., Acta Chem. Scand., B31 (1977) 399–406.
Casy, A.F., Hassan, M.M.A., Simmonds, A.B. and Staniforth, D., J. Pharm. Pharmacol., 21 (1969) 434–440.
Riley, T.N. and Bagley, J.R., J. Med. Chem. 22 (1979) 1167–1171.
Borne, R.F., Law, S-J., Kapeghian, J.C. and Masten, L.W., J. Pharm. Sci., 69 (1980) 1104–1106.
De Ranter, C., Peeters, O. and Gelders, Y., Arch. Int. Physiol. Biochim. 87 (1979) 1031–1033.
Berger, J.G., Davidson, F. and Langford, G.E., J. Med. Chem. 20 (1977) 600–602.
Klein, W., Back, W. and Mutschler, E., Arch. Pharm. (Weinheim. F.R.G.), 308 (1975) 910–916.
Borne R.F., Fifer E.K. and Waters, I.W., J. Med. Chem., 27 (1984) 1271–1275.
Van den Hende, J.H. and Nelson, N.R., J. Am. Chem. Soc., 89 (1967) 2901–2905.
Gylbert, L, Acta Cryst., B, 29 (1973) 1630.
Loew, G.H. and Berkowitz, D.S., J. Med. Chem. 22 (1979) 603–607.
Hutchins, C.W., Cooper, G.K., Purro, S. and Rapoport, H., J. Med. Chem. 24 (1981) 773–775.
Hutchins, C.W. and Rapoport, H., J. Med. Chem. 27 (1984) 521–527.
Humblet, C., Evrard, G. and Durant, F., Acta Cryst., B34 (1978) 3828–3829.
Lednicer, D., Von Voigtlander, P.F. and Emmert, D.E., J. Med. Chem. 24 (1981) 404–408.
Lednicer, D. and Von Voigtlander, P.F., J. Med. Chem., 22 (1979) 1157–1158.
Froimowitz, M., Salva, P., Hite, G.J., Gianutsos, G.G., Suzdak, P. and Heyman, R., J. Comput. Chem., 5 (1984) 291.
Van Bever, W.F.M., Niemegeers, C.J.E., Schellekens, K.H.L. and Janssen, P.A.J., Arzneim.-Forsch./Drug Res., 26 (1976) 1548–1551.
Martin, J.L., Conformational Analysis of Opioid Analgesics, M. Pharm. thesis, Victorian College of Pharmacy Ltd., 1985.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Martin, J., Andrews, P. Conformation-activity relationships of opiate analgesics. J Computer-Aided Mol Des 1, 53–72 (1987). https://doi.org/10.1007/BF01680557
Received:
Accepted:
Issue Date:
DOI: https://doi.org/10.1007/BF01680557