Abstract
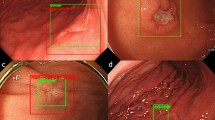
With the improvement of people's requirements for the accuracy of disease detection, increasing researchers apply image recognition to the detection and diagnosis of gastrointestinal diseases. In addition, the evaluation of detection and diagnostic accuracy is also an important factor in the diagnosis and treatment of gastrointestinal diseases. Referring to the QoS and QoE evaluation scheme in 5G network communication, this paper proposes a secondary quality experience evaluation model for gastrointestinal diseases. Firstly, three mandatory detection implementation nodes of 5G network communication are created in the image recognition model, and the first node detects the detected points (images of intestinal diseases) in 5G network communication in the form of interactive information. Then, take the above detection results as the first level to obtain the quality detection strategy results, and the second node uses the policy detection server to evaluate the measurement results as the results of the second level quality evaluation strategy. Finally, the third node is used to evaluate the results of the secondary quality evaluation, and the evaluation results of gastrointestinal disease detection are returned to the output of the whole model. In this paper, 200 CT images of the gastrointestinal tract in the CT colonography dataset were used to evaluate the performance of the model. The study shows that after 200 gastrointestinal CT images are imported into the model, 173 samples have a score of more than 85% for the output of other nodes at the end of the enforcement node, accounting for 86.5% of the sample size. After 24 samples were evaluated by the second level, the output result was the first level scoring error, accounting for 12% of the sample size. Three samples were judged as abnormal images by the first level enforcement node, accounting for 1.5% of the sample size. From the results, the secondary quality experience evaluation model can improve the image recognition accuracy of gastrointestinal disease detection and diagnosis from 86.5% to 98.5%, which greatly improves the accuracy of image recognition.












Similar content being viewed by others
Data availability
Enquiries about data availability should be directed to the authors.
References
Deepak KJ, Masoumeh Z, Rachna J, Abhishek K, Shivam B (2020) GAN-Poser: an improvised bidirectional GAN model for human motion prediction. Neural Comput Appl 32(18):14579–14591
Deepak KJ, Sumarga KST, Neelakandan S, Prakash M, Lakshmaiya N (2022) Metaheuristic optimization-based resource allocation technique for cybertwin-driven 6G on IoE environment. IEEE Trans Ind Inform 18(7):4884–4892
El-Deeb OS, Atef MM, Hafez YM (2019) The interplay betweenmicrobiota-dependent metabolite trimethylamine N-oxide, transforming growth factor SMAD signaling and inflamma some activation in chronic kidney disease patients:a new mechan is ticperspective. J Cell Biochem 120(9):14476–14485
Falony G, Vieira-Silva S, Raes J (2019) Microbiology meets big data:the case of gut microbiota–derived trimethylamine. AnnuRev Microbiol 69(1):305–321
Fennema D, Phillips IR, Shephard EA (2019) Trimethylamine andtrimethylamine N-oxide, a flavin-containing monooxygenase 3(FMO3)-mediated host-microbiome metabolic axis implicatedin health and disease. Drug Metab Dispos 44(11):1839–1850
Geovanini GR, Libby P (2018) Atherosclerosis and inflammation: overview and updates. Clin Sci (lond) 132(12):1243–1252
Hoyles L, Jiménez-Pranteda ML, Chilloux J et al (2018) Metabolicretroconversion of trimethylamine N-oxide and the gutmicrobiota. Microbiome 6(1):73–79
Landfald B, Valeur J, Berstad A et al (2017) Microbialtrimethylamine-N-oxide as a disease marker: somethingfishy? Microb Ecol Health Dis 28(1):132–139
Levin A, Tonelli M, Bonventre J et al (2017) Global kidney health2017 and beyond:a roadmap for closing gaps in care, research, and policy. Lancet 390(10105):1888–1917
Li TJ, Chen YL, Gua CJ et al (2017) Elevated circulatingtrimethylamine N-oxide levels contribute to endothelialdysfunction in aged rats through vascular inflammation andoxidative stress. Front Physiol 8(4):350–351
Li D, Ke Y, Zhan R et al (2018) Trimethylamine-N-oxide promotesbrain aging and cognitive impairment in mice. Aging Cell 17(4):68–72
Liu J, Zhao M, Zhou J et al (2019) Simultaneous targeted analysis oftrimethylamine-N-oxide, choline, betaine, and carnitine by highperformance liquid chromatography tandem massspectrometry. J Chromatogr B Analyt Technol Biomed LifeSci 1035(4):42–48
Missailidis C, Hällqvist J, Qureshi AR et al (2019) Serumtrimethylamine-N-oxide is strongly related to renal function andpredicts outcome in chronic kidney disease. PLoS ONE 11(1):38–39
Pelletier CC, Croyal M, Ene L et al (2019) Elevation oftrimethylamine-N-oxide in chronic kidney disease:contribution ofdecreased glomerular filtration rate. Toxins (basel) 11(11):35–36
Shih DM, Wang Z, Lee R et al (2019) Flavin containingmonooxygenase 3 exerts broad effects on glucose and lipidmetabolism and atherosclerosis. J Lipid Res 56(1):22–37
Taesuwan S, Cho CE, Malysheva OV et al (2017) The metabolic fateof isotopically labeled trimethylamine-N-oxide (TMAO) inhumans. J Nutr Biochem 45(5):77–82
Tang WH, Wang ZN, Kennedy DJ et al (2019) Gut microbiota-dependent trimethylamine N-oxide (TMAO) pathway contributesto both development of renal insufficiency and mortality risk inchronic kidney disease. Circ Res 116(3):448–455
Thompson S, James M, Wiebe N et al (2019) Cause of death inpatients with reduced kidney function. J Am SocNephrol 26(10):2504–2511
Wang Z, Han Z, Tao J et al (2017) Role of endothelial-to-mesenchymal transition induced by TGF-β1 in transplant kidneyinterstitial fibrosis. J Cell Mol Med 21(10):2359–2369
Wang Z, Bergeron N, Levison BS et al (2019) Impact of chronicdietary red meat, white meat, or non-meat protein ontrimethylamine N-oxide metabolism and renal excretion in healthymen and women. Eur Heart J 40(7):583–594
Watanabe H, Miyamoto Y, Honda D et al (2019) p-Cresyl sulfatecauses renal tubular cell damage by inducing oxidative stress byactivation of NADPH oxidase. Kidney Int 83(4):582–592
Xu KY, Xia GH, Lu JQ et al (2017) Impaired renal function anddysbiosis of gut microbiota contribute to increased trimethylamine-N-oxide in chronic kidney disease patients. Sci Rep 7(1):1445–1448
Yancey PH, Speers-Roesch B, Atchinson S et al (2018) Osmolyteadjustments as a pressure adaptation in deep-sea chondrichthyan1 Acad J Chin PLA Med Sch: an intraspecific test in arctic skates (Amblyrajahyperborea) along a depth gradient. Physiol BiochemZool 91(2):788–796
Yang J, Lim SY, Ko YS et al (2019) Intestinal barrier disruption anddysregulated mucosal immunity contribute to kidney fibrosis inchronic kidney disease. Nephrol Dial Transplant 34(3):419–428
Zeisel SH, Warrier M (2017) Trimethylamine N-oxide, themicrobiome, and heart and kidney disease. Annu RevNutr 37(4):157–181
Funding
There is no funding for this study.
Author information
Authors and Affiliations
Contributions
WZ: Experiment designing, editing. YG: Experiment process implementing, writing. SS: Data collecting. FG: Data analysis.
Corresponding author
Ethics declarations
Conflict of interest
There is no potential conflict of interest in this study.
Ethical approval
This paper does not contain any studies with human participants or animals performed by any of the authors.
Informed consent
This paper does not contain any studies with human participants performed by any of the authors, so there's no informed consent involved.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Zhang, W., Gao, Y., Song, S. et al. Image recognition for gastrointestinal disease detection and diagnosis in QoS and QoE evaluation of 5G network communication. Soft Comput 26, 13799–13813 (2022). https://doi.org/10.1007/s00500-022-07368-2
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s00500-022-07368-2