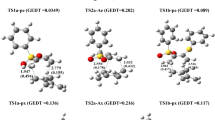
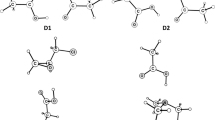
Abstract
Electrophilic olefins can react with the S–H moiety of cysteine side chains. The formation of a covalent adduct through this mechanism can result in the inhibition of an enzyme. The reactivity of an olefin towards cysteine depends on its functional groups. In this study, 325 reactions of thiol-Michael-type additions to olefins were modeled using density functional theory. All combinations of ethenes with hydrogen, methyl ester, amide, and cyano substituents were included. An automated workflow was developed to perform the construction, conformation search, minimization, and calculation of molecular properties for the reactant, carbanion intermediate, and thioether products for a model reaction of the addition of methanethiol to the electrophile. Known cysteine-reactive electrophiles present in the database were predicted to react exergonically with methanethiol through a carbanion with a stability in the 30–40 kcal mol−1 range. 13 other compounds in our database that are also present in the PubChem database have similar properties. Natural bond orbital parameters were computed and regression analysis was used to determine the relationship between properties of the olefin electronic structure and the product and intermediate stability. The stability of the intermediates is very sensitive to electronic effects on the carbon where the anionic charge is centered. The stability of the products is more sensitive to steric factors.








Similar content being viewed by others
References
Enoch SJ, Ellison CM, Schultz TW, Cronin MTD (2011) Crit Rev Toxicol 41(9):783
Potashman MH, Duggan ME (2009) J Med Chem 52(5):1231
Gersch M, Kreuzer J, Sieber SA (2012) Nat Prod Rep 29(6):659
Miller RM, Taunton J (2014) Chapter four—targeting protein kinases with selective and semi-promiscuous covalent inhibitors. In: Kevan MS (ed) Methods in enzymology, vol 548. Academic Press, New York, p 93
Rabindran SK, Discafani CM, Rosfjord EC, Baxter M, Floyd MB, Golas J, Hallett WA, Johnson BD, Nilakantan R, Overbeek E, Reich MF, Shen R, Shi X, Tsou H-R, Wang Y-F, Wissner A (2004) Cancer Res 64(11):3958
Zhou W, Hur W, McDermott U, Dutt A, Xian W, Ficarro SB, Zhang J, Sharma SV, Brugge J, Meyerson M, Settleman J, Gray NS (2010) Chem Biol 17(3):285–295. doi:10.1016/j.chembiol.2010.02.007
Pan Z, Scheerens H, Li S-J, Schultz BE, Sprengeler PA, Burrill LC, Mendonca RV, Sweeney MD, Scott KCK, Grothaus PG, Jeffery DA, Spoerke JM, Honigberg LA, Young PR, Dalrymple SA, Palmer JT (2007) Chem Med Chem 2(1):58
Ettari R, Micale N, Schirmeister T, Gelhaus C, Leippe M, Nizi E, Di Francesco ME, Grasso S, Zappalà M (2009) J Med Chem 52(7):2157
Serafimova IM, Pufall MA, Krishnan S, Duda K, Cohen MS, Maglathlin RL, McFarland JM, Miller RM, Frödin M, Taunton J (2012) Nat Chem Biol 8(5):471
Mulliner D, Wondrousch D, Schuurmann G (2011) Org Biomol Chem 9(24):8400
Hoyle CE, Bowman CN (2010) Angew Chem Int Ed 49(9):1540
Lutolf MP, Tirelli N, Cerritelli S, Cavalli L, Hubbell JA (2001) Bioconj Chem 12(6):1051
Nair DP, Podgórski M, Chatani S, Gong T, Xi W, Fenoli CR, Bowman CN (2014) Chem Mater 26(1):724
Krishnan S, Miller RM, Tian B, Mullins RD, Jacobson MP, Taunton J (2014) J Am Chem Soc 136(36):12624
London N, Miller RM, Krishnan S, Uchida K, Irwin JJ, Eidam O, Gibold L, Cimermančič P, Bonnet R, Shoichet BK, Taunton J (2014) Nat Chem Biol 10(12):1066
Schwöbel JAH, Wondrousch D, Koleva YK, Madden JC, Cronin MTD, Schüürmann G (2010) Chem Res Toxicol 23(10):1576
Krenske EH, Petter RC, Zhu Z, Houk KN (2011) J Org Chem 76(12):5074
Capoferri L, Lodola A, Rivara S, Mor M (2015) J Chem Inf Model 55(3):589
Irwin JJ, Shoichet BK, Mysinger MM, Huang N, Colizzi F, Wassam P, Cao Y (2009) J Med Chem 52(18):5712
Del Rio A, Sgobba M, Parenti M, Degliesposti G, Forestiero R, Percivalle C, Conte P, Freccero M, Rastelli G (2010) J Comput Aided Mol Des 24(3):183
Ouyang X, Zhou S, Su CTT, Ge Z, Li R, Kwoh CK (2013) J Comput Chem 34(4):326
Smith JM, Jami Alahmadi Y, Rowley CN (2013) J Chem Theory Comput 9(11):4860
Hori K, Higuchi S, Kamimura A (1990) J Org Chem 55(23):5900
Bolton EE, Wang Y, Thiessen PA, Bryant SH (2008) Chapter 12—PubChem: integrated platform of small molecules and biological activities. In: Ralph AW, David CS (eds) Annual reports in computational chemistry, vol 4. Elsevier, p 217
Schmidt TJ, Ak M, Mrowietz U (2007) Bioorg Med Chem 15(1):333
Edwards PM (1975) Br J Ind Med 32(1):31
Guengerich FP, Geiger LE, Hogy LL, Wright PL (1981) Cancer Res 41(12 Part 1):4925
Schwartz PA, Kuzmic P, Solowiej J, Bergqvist S, Bolanos B, Almaden C, Nagata A, Ryan K, Feng J, Dalvie D, Kath JC, Xu M, Wani R, Murray BW (2014) Proc Natl Acad Sci 111(1):173
Parr RG, Szentpály L, Liu S (1999) J Am Chem Soc 121(9):1922
Taylor JB, Kennewell PD (1981) Introductory medicinal chemistry. Ellis Horwood Limited, Chichester
Weininger D (1988) J Chem Inf Comput Sci 28(1):31
O’Boyle N, Banck M, James C, Morley C, Vandermeersch T, Hutchison G (2011) J Cheminform 3(1):33
Wang J, Wang W, Kollman PA, Case DA (2006) J Mol Graph Model 25(2):247
Wang J, Wolf RM, Caldwell JW, Kollman PA, Case DA (2004) J Comput Chem 25(9):1157
Earl DJ, Deem MW (2005) Phys Chem Chem Phys 7(23):3910
Brooks BR, Brooks CL, Mackerell AD, Nilsson L, Petrella RJ, Roux B, Won Y, Archontis G, Bartels C, Boresch S, Caflisch A, Caves L, Cui Q, Dinner AR, Feig M, Fischer S, Gao J, Hodoscek M, Im W, Kuczera K, Lazaridis T, Ma J, Ovchinnikov V, Paci E, Pastor RW, Post CB, Pu JZ, Schaefer M, Tidor B, Venable RM, Woodcock HL, Wu X, Yang W, York DM, Karplus M (2009) J Comput Chem 30(10):1545
Rowley CN CHARMM Conformation Search. https://github.com/RowleyGroup/charmm-conformation. Accessed 15 May 2015
Dewar MJS, Thiel W (1977) J Am Chem Soc 99(15):4899
TURBOMOLE V6.6 2014, a development of University of Karlsruhe and Forschungszentrum Karlsruhe GmbH, 1989–2007, TURBOMOLE GmbH, since 2007. http://www.turbomole.com
Adamo C, Barone V (1999) J Chem Phys 110(13):6158
Schäfer A, Huber C, Ahlrichs R (1994) J Chem Phys 100(8):5829
Chai J-D, Head-Gordon M (2008) J Chem Phys 128(8):084106
Spartan ‘10 (2010) Wavefunction Inc., Irvine, CA
Lamoureux G, Roux B (2006) J Phys Chem B 110(7):3308
Kreevoy MM, Eichinger BE, Stary FE, Katz EA, Sellstedt JH (1964) J Org Chem 29(6):1641
Dunning TH (1989) J Chem Phys 90(2):1007
Woon DE, Dunning TH (1993) J Chem Phys 98(2):1358
Tomasi J, Mennucci B, Cammi R (2005) Chem Rev 105(8):2999
Reed AE, Weinstock RB, Weinhold F (1985) J Chem Phys 83(2):735
Glendening ED, Reed AE, Carpenter JE, Weinhold F (2003) NBO Version 3.1. Gaussian Inc., Pittsburg, PA. http://www.gaussian.com/g_tech/g_ur/m_citation.htm
Mennucci B (2012) Wiley Interdiscip Rev Comput Mol Sci 2(3):386
Acknowledgments
We thank NSERC of Canada for funding through a Discovery Grant (Application No. 418505-2012) and an Undergraduate Student Research Award for JMS. JMS thanks the Dean of Science of Memorial University for a travel grant. Computational resources were provided by the Compute Canada Consortium (CCI: djk-615-ac). We thank Archita Adluri for proofreading the manuscript. We thank the reviewers for meticulous examination of the manuscript.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Smith, J.M., Rowley, C.N. Automated computational screening of the thiol reactivity of substituted alkenes. J Comput Aided Mol Des 29, 725–735 (2015). https://doi.org/10.1007/s10822-015-9857-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-015-9857-0