Abstract
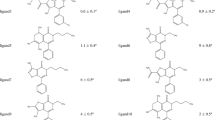
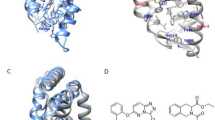
The knowledge of the free energy of binding of small molecules to a macromolecular target is crucial in drug design as is the ability to predict the functional consequences of binding. We highlight how a molecular dynamics (MD)-based approach can be used to predict the free energy of small molecules, and to provide priorities for the synthesis and the validation via in vitro tests. Here, we study the dynamics and energetics of the nuclear receptor REV-ERBα with its co-repressor NCoR and 35 novel agonists. Our in silico approach combines molecular docking, molecular dynamics (MD), solvent-accessible surface area (SASA) and molecular mechanics poisson boltzmann surface area (MMPBSA) calculations. While docking yielded initial hints on the binding modes, their stability was assessed by MD. The SASA calculations revealed that the presence of the ligand led to a higher exposure of hydrophobic REV-ERB residues for NCoR recruitment. MMPBSA was very successful in ranking ligands by potency in a retrospective and prospective manner. Particularly, the prospective MMPBSA ranking-based validations for four compounds, three predicted to be active and one weakly active, were confirmed experimentally.










Similar content being viewed by others
Abbreviations
- FRET:
-
Fluorescence resonance energy transfer
- M2H:
-
Mammalian-2-hybrid assay
- MD:
-
Molecular dynamics
- MMPBSA:
-
Molecular mechanics poisson boltzmann surface area calculations
- NR:
-
Nuclear receptor
- RMSD:
-
Root-mean-square deviation
- SASA:
-
Solvent-accessible surface area
References
Akashi M, Takumi T (2005) Nat Struct Mol Biol 12:441
Preitner N et al (2002) Cell 110:251
Sato TK et al (2004) Neuron 43:527
Yin L, Lazar MA (2005) Mol Endocrinol 19:1452
Triqueneaux G et al (2004) J Mol Endocrinol 33:585
Laposky AD, Bass J, Kohsaka A, Turek FW (2008) FEBS Lett 582:142
Cho H et al (2012) Nature 485:123
Delezie J et al (2012) Faseb J 26:3321
Xu HE et al (2002) Nature 415:813
Wang L et al (2006) J Biol Chem 281:37773
Madauss KP et al (2007) Mol Endocrinol 21:1066
Woo EJ et al (2007) J Mol Biol 373:735
Horlein AJ et al (1995) Nature 377:397
Chen JD, Evans RM (1995) Nature 377:454
Phelan CA et al (2010) Nat Struct Mol Biol 17:808
Hu X, Li Y, Lazar MA (2001) Mol Cell Biol 21:1747
Vaissière A et al (2015) Protein Sci 24:1129
Grant D et al (2010) ACS Chem Biol 5:925
Westermaier Y, Hubbard RE (2013) In: Schneider G (ed) De novo molecular design. Wiley, Weinheim
De Vivo M, Masetti M, Bottegoni G, Cavalli A (2016) J Med Chem 59:4035
Stahl M, Rarey M (2001) J Med Chem 44:1035
Bursulaya BD, Totrov M, Abagyan R, C. L. Brooks (2003) 3rd, J Comput Aided Mol Des 17:755
Kellenberger E, Rodrigo J, Muller P, Rognan D (2004) Proteins 57:225
Warren GL et al (2006) J Med Chem 49:5912
Chipot C, Pande VS, Mark AE, Simonson T (2007) In: Chipot C, Pohorille C (eds) Free energy calculations: theory and applications in chemistry and biology. Springer, Berlin and Heidelberg, 461–506
Jones G, Willett P, Glen RC, Leach AR, Taylor R (1997) J Mol Biol 267:727
Jones G, Willett P, Glen RC (1995) J Mol Biol 245:43
Verdonk ML, Cole JC, Hartshorn MJ, Murray CW, Taylor RD (2003) Proteins 52:609
In MOE (2014) Chemical Computing Group Inc., Montréal, QC, Québec
Frisch MJ, Trucks GW, Schlegel HB, Scuseria GE, Robb MA, Cheeseman JR, Scalmani G, Barone V, Mennucci B, Petersson GA, Nakatsuji H, Caricato M, Li X, Hratchian HP, Izmaylov AF, Bloino J, Zheng G, Sonnenberg JL, Hada M, Ehara M, Toyota K, Fukuda R, Hasegawa J, Ishida M, Nakajima T, Honda Y, Kitao O, Nakai H, Vreven T, Montgomery JA Jr, Peralta JE, Ogliaro F, Bearpark M, Heyd JJ, Brothers E, Kudin KN, Staroverov VN, Kobayashi R, Normand J, Raghavachari K, Rendell A, Burant JC, Iyengar SS, Tomasi J, Cossi M, Rega N, Millam JM, Klene M, Knox JE, Cross JB, Bakken V, Adamo C, Jaramillo J, Gomperts R, Stratmann RE, Yazyev O, Austin AJ, Cammi R, Pomelli C, Ochterski JW, Martin RL, Morokuma K, Zakrzewski VG, Voth GA, Salvador P, Dannenberg JJ, Dapprich S, Daniels AD, Farkas Ö, Foresman JB, Ortiz JV, Cioslowski J, Fox DJ (2009) Gaussian, Inc., Wallingfort, CT
Case DA et al (2010) University of California, San Francisco
Lindorff-Larsen K et al (2010) Proteins 78:1950
Case DA et al (2012) (University of California, San Francisco
Ruiz-Carmona S, Barril X (2013) Small Molecule Dihedrals Parametrization. Zenodo. http://doi.org/10.5281/zenodo.400235
Álvarez-García D, Barril X (2014) J Med Chem 57:8530
Álvarez-García D, Barril X (2014) Computational Drug Design Group. University of Barcelona, Barcelona
Darden T, York D, Pedersen L (1993) J Chem Phys 98:10089–10092
Roe DR, Cheatham TE III (2013) J Chem Theor Comput 9:3084
In PyMOL (2014) Molecular Graphics System. Schrödinger, LLC
Humphrey W, Dalke A, Schulten K (1996) J Mol Graph 14:33
In VMD 1.9.1. (2014)
Lee B, Richards FM (1971) J Mol Biol 55:379
Kollman PA et al (2000) Acc Chem Res 33:889
Srinivasan J, Cheatham TE, Cieplak P, Kollman PA, Case DA (1998) J Am Chem Soc 120:9401
Wang J, Morin P, Wang W, Kollman PA (2001) J Am Chem Soc 123:5221
Gohlke H, Case DA (2004) J Comput Chem 25:238
Kuhn B, Kollman PA (2000) J Med Chem 43:3786
Rastelli G, Del Rio A, Degliesposti G, Sgobba M (2010) J Comput Chem 31:797
Sitkoff D, Sharp K, Honing B (1994) J Phys Chem 98:1978–1988
Acknowledgements
Y.W., S.R.-C. and X. B. thank Servier for financial support.
Author information
Authors and Affiliations
Corresponding author
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Westermaier, Y., Ruiz-Carmona, S., Theret, I. et al. Binding mode prediction and MD/MMPBSA-based free energy ranking for agonists of REV-ERBα/NCoR. J Comput Aided Mol Des 31, 755–775 (2017). https://doi.org/10.1007/s10822-017-0040-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-017-0040-7