Abstract
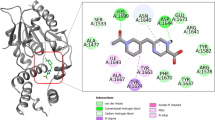
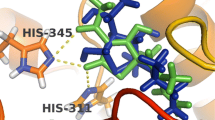
Tuberculosis (TB) remains a major threat to human health. This due to the fact that current drug treatments are less than optimal and the increasing occurrence of multi drug-resistant strains of etiological agent, Mycobacterium tuberculosis (Mt). Given the wide-spread significance of this disease, we have undertaken a design and evaluation program to discover new anti-TB drug leads. Here, we focused on ketol-acid reductoisomerase (KARI), the second enzyme in the branched-chain amino acid biosynthesis pathway. Importantly, this enzyme is present in bacteria but not in humans, making it an attractive proposition for drug discovery. In the present work, we used molecular docking to identify seventeen potential inhibitors of KARI using an in-house database. Compounds were selected based on docking scores, which were assigned as the result of favourable interactions between the compound and the active site of KARI. The inhibitory constant values for two leads, compounds 14 and 16 are 3.71 and 3.06 µM respectively. To assess the mode of binding, 100 ns molecular dynamics simulations for these two compounds in association with Mt KARI were performed and showed that the complex was stable with an average root mean square deviation of less than 3.5 Å for all atoms. Furthermore, compound 16 showed a minimum inhibitory concentration of 2.06 ± 0.91 µM and a 1.9 fold logarithmic reduction in the growth of Mt in an infected macrophage model. The two compounds exhibited low toxicity against RAW 264.7 cell lines. Thus, both compounds are promising candidates for development as an anti-TB drug leads.










Similar content being viewed by others
References
WHO (2017) WHO global tuberculosis report 2017. World Health Organization, Geneva
Zumla A, Raviglione M, Hafner R, Von Reyn CF (2013) Tuberculosis. N Engl J Med 368:745–755
Jain A, Mondal R (2008) Extensively drug-resistant tuberculosis: current challenges and threats. FEMS Immunol Med Microbiol 53:145–150
Rawat DS (2013) Antituberculosis drug research: a critical overview. Med Res Rev 33:693–764
Griffin JE, Gawronski JD, DeJesus MA, Ioerger TR, Akerley BJ, Sassetti CM (2011) High-resolution phenotypic profiling defines genes essential for mycobacterial growth and cholesterol catabolism. PLoS Pathog. https://doi.org/10.1371/journal.ppat.1002251
Wang D, Zhu X, Cui C (2013) Discovery of novel acetohydroxyacid synthase inhibitors as active agents against Mycobacterium tuberculosis by virtual screening and bioassay. J Chem Inf Model 53:343–353
Choi KJ, Yu YG, Hahn HG, Choi JD, Yoon MY (2005) Characterization of acetohydroxyacid synthase from Mycobacterium tuberculosis and the identification of its new inhibitor from the screening of a chemical library. FEBS Lett 579:4903–4910
Chunduru SK, Mrachko GT, Calvo KC (1989) Mechanism of ketol acid reductoisomerase—steady-state analysis and metal ion requirement. Biochemistry 28:486–493
Tadrowski S, Pedroso MM, Sieber V, Larrabee JA, Guddat LW, Schenk G (2016) Metal ions play an essential catalytic role in the mechanism of ketol-acid reductoisomerase. Chem-A Eur J 22:7427–7436
Grandoni JA, Marta PT, Schloss JV (1998) Inhibitors of branched-chain amino acid biosynthesis as potential antituberculosis agents. J Antimicrob Chemother 42:475–482
Lv Y, Kandale A, Wun SJ et al (2016) Crystal structure of Mycobacterium tuberculosis ketol-acid reductoisomerase at 1.0 Å resolution—a potential target for anti-tuberculosis drug discovery. FEBS J 283:1184–1196
Friesner RA, Banks JL, Murphy RB et al (2004) Glide: a new approach for rapid, accurate docking and scoring. 1. Method and assessment of docking accuracy. J Med Chem 47:1739–1749
Beesetti H, Tyagi P, Medapi B et al (2018) A quinoline compound inhibits the replication of dengue virus serotypes 1–4 in Vero cells. Antivir Ther. https://doi.org/10.3851/IMP3231
Friesner RA, Murphy RB, Repasky MP et al (2006) Extra precision glide: docking and scoring incorporating a model of hydrophobic enclosure for protein-ligand complexes. J Med Chem 49:6177–6196
Tripathi SK, Muttineni R, Singh SK (2013) Extra precision docking, free energy calculation and molecular dynamics simulation studies of CDK2 inhibitors. J Theor Biol 334:87–100
Katari SK, Natarajan P, Swargam S, Kanipakam H, Pasala C, Umamaheswari A (2016) Inhibitor design against JNK1 through e-pharmacophore modeling docking and molecular dynamics simulations. J Recept Signal Transduct 36:558–571
Martyna GJ, Tobias DJ, Klein ML (1994) Constant pressure molecular dynamics algorithms. J Chem Phys 101:4177–4189
Mobley DL, Dill KA (2009) Binding of small-molecule ligands to proteins: ‘what you see’ is not always ‘what you get’. Structure 17:489–498
Lyne PD, Lamb ML, Saeh JC (2006) Accurate prediction of the relative potencies of members of a series of kinase inhibitors using molecular docking and MM-GBSA scoring. J Med Chem 49:4805–4808
Collins LA, Franzblau SG (1997) Microplate Alamar blue assay versus BACTEC 460 system for high-throughput screening of compounds against Mycobacterium tuberculosis and Mycobacterium avium. Antimicrob Agents Chemother 41:1004–1009
Betts JC, Lukey PT, Robb LC, McAdam RA, Duncan K (2002) Evaluation of a nutrient starvation model of Mycobacterium tuberculosis persistence by gene and protein expression profiling. Mol Microbiol 43:717–731
Malapati P, Krishna VS, Nallangi R, Srilakshmi RR, Sriram D (2018) Identification and development of benzoxazole derivatives as novel bacterial glutamate racemase inhibitors. Eur J Med Chem 145:23–34
Kulka K, Hatfull G, Ojha AK (2012) Growth of Mycobacterium tuberculosis biofilms. J Vis Exp. https://doi.org/10.3791/3820
Malapati P, Siva Krishna V, Nallangi R, Meda N, Srilakshmi RR, Sriram D (2018) Lead identification and optimization of bacterial glutamate racemase inhibitors. Bioorg Med Chem 26:177–190
Reck F, Alm RA, Brassil P et al (2012) Novel N-linked aminopiperidine inhibitors of bacterial topoisomerase type II with reduced pKa: antibacterial agents with an improved safety profile. J Med Chem 55:6916–6933
Biketov S, Mukamolova GV, Potapov V et al (2000) Culturability of Mycobacterium tuberculosis cells isolated from murine macrophages: a bacterial growth factor promotes recovery. FEMS Immunol Med Microbiol 29:233–240
Acknowledgements
VSK and EMR are thankful to Department of Science and Technology, Government of India for the INSPIRE fellowship. DS is thankful to Department of Biotechnology, Government of India for the Tata innovation fellowship (BT/HRD/35/01/04/2015). LWG is supported by an NHMRC Project Grant 1147297.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Conflict of interest
The authors declare no conflict of interest.
Additional information
Publisher’s Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Krishna, V.S., Zheng, S., Rekha, E.M. et al. Discovery and evaluation of novel Mycobacterium tuberculosis ketol-acid reductoisomerase inhibitors as therapeutic drug leads. J Comput Aided Mol Des 33, 357–366 (2019). https://doi.org/10.1007/s10822-019-00184-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-019-00184-1