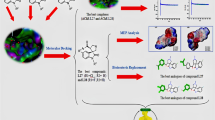
Abstract
Nowadays, the importance of computational methods in the design of therapeutic agents in a more efficient way is indisputable. Particularly, these methods have been important in the design of novel acetylcholinesterase enzyme inhibitors related to Alzheimer’s disease. In this sense, in this report a computational model of linear prediction of acetylcholinesterase inhibitory activity of steroids and triterpenes is presented. The model is based in a correlation between binding energies obtained from molecular dynamic simulations (after docking studies) and \(\hbox {IC}_{{50}}\) values of a training set. This set includes a family of natural and semi-synthetic structurally related alkaloids reported in bibliography. These types of compounds, with some structural complexity, could be used as building blocks for the synthesis of many important biologically active compounds Therefore, the present study proposes an alternative based on the use of conventional and easily accessible tools to make progress on the rational design of molecules with biological activity.





Similar content being viewed by others
References
Gammon K (2014) Nature 515:299. https://doi.org/10.1038/nj7526-299a
Patterson C (2018) World Alzheimer report 2018. The state of the art of dementia research: New frontiers. Tech. rep., Alzheimer’s Disease International
World Alzheimer report (2019) Attitudes to dementia. Tech. rep., Alzheimer’s Disease International
Wimo A, Jönsson L, Bond J, Prince M, Winblad B (2013) Alzheimer’s Dementia 9(1):1. https://doi.org/10.1016/j.jalz.2012.11.006
Mount C, Downton C (2006) Nat Med 12:780. https://doi.org/10.1038/nm0706-780
Haas C (2012) J Alzheimer’s Dis 28:241. https://doi.org/10.3233/JAD-2011-110986
Kumar A, Singh A (2015) Ekavali. Pharmacol Rep 67(2):195. https://doi.org/10.1016/j.pharep.2014.09.004
Hurtado-Puerto AM, Russo C, Fregni F (2018) Alzheimer’s disease. Springer, New York
Husna Ibrahim N, Yahaya MF, Mohamed W, Teoh SL, Hui CK, Kumar J (2020) Front Pharmacol 11:261. https://doi.org/10.3389/fphar.2020.00261
Montanari S, Bartolini M, Neviani P, Belluti F, Gobbi S, Pruccoli L, Tarozzi A, Falchi F, Andrisano V, Miszta P, Cavalli A, Filipek S, Bisi A, Rampa A (2016) ChemMedChem 11(12):1296. https://doi.org/10.1002/cmdc.201500392
Prati F, Bergamini C, Fato R, Soukup O, Korabecny J, Andrisano V, Bartolini M, Bolognesi ML (2016) ChemMedChem 11(12):1284. https://doi.org/10.1002/cmdc.201600014
Viayna E, Gómez T, Galdeano C, Ramríez L, Ratia M, Badia A, Clos MV, Verdaguer E, Junyent F, Camins A, Pallàs M, Bartolini M, Mancini F, Andrisano V, Arce MP, Rodríguez-Franco MI, Bidon-Chanal A, Luque FJ, Camps P, Muñoz-Torrero D (2010) ChemMedChem 5(11):1855. https://doi.org/10.1002/cmdc.201000322
Galdeano C, Viayna E, Arroyo P, Bidon-Chanal A, Ramon Blas J, Munoz-Torrero D, Javier Luque F (2010) Curr Pharm Des 16(25):2818. https://doi.org/10.2174/138161210793176536
Bachurin SO, Bovina EV, Ustyugov AA (2017) Med Res Rev 37(5):1186. https://doi.org/10.1002/med.21434
Cole MA, Seabrook GR (2020) Alzheimer’s Dementia 6(1):e12009. https://doi.org/10.1002/trc2.12009
Makin S (2018) Nature 559(7715):S4–S7. https://doi.org/10.1038/d41586-018-05719-4
Alkadhi K, Eriksen J (2011) Curr Neuropharmacol 9(4):586. https://doi.org/10.2174/157015911798376235
Carreiras MC, Mendes E, Perry MJ, Francisco AP, Marco-Contelles J (2013) Curr Top Med Chem 13(15):1745. https://doi.org/10.2174/15680266113139990135
Contestabile A (2011) Behav Brain Res 221(2):334. https://doi.org/10.1016/j.bbr.2009.12.044
Whitehouse P, Price D, Struble R, Clark A, Coyle J, Delon M (1982) Science 215(4537):1237. https://doi.org/10.1126/science.7058341
Chrispin P (1995) The Lancet 345:1248. https://doi.org/10.5555/uri:pii:S0140673695920366
Liu JS, Zhu YL, Yu CM, Zhou YZ, Han YY, Wu FW, Qi BF (1986) Can J Chem 64(4):837. https://doi.org/10.1139/v86-137
Ha GT, Wong RK, Zhang Y (2011) Chem Biodivers 8(7):1189. https://doi.org/10.1002/cbdv.201000269
Wilcock GK, Lilienfeld S, Gaens E (2000) BMJ 321(7274):1445. https://doi.org/10.1136/bmj.321.7274.1445
Boit HG (1954) Chem Berichte 87(5):724. https://doi.org/10.1002/cber.19540870517
Quinn DM (1987) Chem Rev 87(5):955. https://doi.org/10.1021/cr00081a005
Silman I, Sussman JL (2008) Chemico-Biol Interact 175(1–3):3. https://doi.org/10.1016/j.cbi.2008.05.035
Dvir H, Silman I, Harel M, Rosenberry TL, Sussman JL (2010) Chemico-Biol Interact 187(1–3):10. https://doi.org/10.1016/j.cbi.2010.01.042
Cras P, Kawai M, Lowery D, Gonzalez-DeWhitt P, Greenberg B, Perry G (1991) Proc Natl Acad Sci 88(17):7552. https://doi.org/10.1073/pnas.88.17.7552
Jakob-Roetne R, Jacobsen H (2009) Angew Chem Int Ed 48(17):3030. https://doi.org/10.1002/anie.200802808
Dunkel PI, Chai CL, Sperlágh B, Huleatt PB, Mátyus P (2012) Expert Opin Investig Drugs 21(9):1267. https://doi.org/10.1517/13543784.2012.703178
Zinchenko E, Navolokin N, Shirokov A, Khlebtsov B, Dubrovsky A, Saranceva E, Abdurashitov A, Khorovodov A, Terskov A, Mamedova A, Klimova M, Agranovich I, Martinov D, Tuchin V, Semyachkina-Glushkovskaya O, Kurts J (2019) Biomed Opt Express 10(8):4003. https://doi.org/10.1364/BOE.10.004003
De Ferrari GV, Canales MA, Shin I, Weiner LM, Silman I, Inestrosa NC (2001) Biochemistry 40(35):10447. https://doi.org/10.1021/bi0101392
Inestrosa NC, Alvarez A, Pérez CA, Moreno RD, Vicente M, Linker C, Casanueva OI, Soto C, Garrido J (1996) Neuron 16:881. https://doi.org/10.1016/S0896-6273(00)80108-7
Rees T, Hammond P, Soreq H, Younkin S, Brimijoin S (2003) Neurobiol Aging 24:777. https://doi.org/10.1016/S0197-4580(02)00230-0
Muñoz FJ, Inestrosa NC (1999) FEBS Lett 450(3):205. https://doi.org/10.1016/S0014-5793(99)00468-8
Bartolini M, Bertucci C, Cavrini V, Andrisano V (2003) Biochem Pharmacol 65(3):407. https://doi.org/10.1016/S0006-2952(02)01514-9
Piazzi L, Rampa A, Bisi A, Gobbi S, Belluti F, Cavalli A, Bartolini M, Andrisano V, Valenti P, Recanatini M (2003) J Med Chem 46(12):2279. https://doi.org/10.1021/jm0340602
Bolognesi ML, Cavalli A, Valgimigli L, Bartolini M, Rosini M, Andrisano V, Recanatini M, Melchiorre C (2007) J Med Chem 50(26):6446. https://doi.org/10.1021/jm701225u
Munoz-Torrero D (2008) Curr Med Chem 15(24):2433. https://doi.org/10.2174/092986708785909067
Agatonovic-Kustrin S, Kettle C, Morton DW (2018) Biomed Pharmacother 106:553. https://doi.org/10.1016/j.biopha.2018.06.147
Galdeano C, Viayna E, Sola I, Formosa X, Camps P, Badia A, Clos MV, Relat J, Ratia M, Bartolini M, Mancini F, Andrisano V, Salmona M, Minguillón C, González-Muñoz GC, Rodrí-guez-Franco MI, Bidon-Chanal A, Luque FJ, Muñoz-Torrero D (2012) J Med Chem 55(2):661. https://doi.org/10.1021/jm200840c
Cai P, Fang SQ, Yang HL, Yang XL, Liu QH, Kong LY, Wang XB (2018) Eur J Med Chem 157:161. https://doi.org/10.1016/j.ejmech.2018.08.005
Kryger G, Silman I, Sussman JL (1999) Structure 7(3):297. https://doi.org/10.1016/S0969-2126(99)80040-9
Cheung J, Rudolph MJ, Burshteyn F, Cassidy MS, Gary EN, Love J, Franklin MC, Height JJ (2012) J Med Chem 55(22):10282. https://doi.org/10.1021/jm300871x PMID: 23035744
Gerlits O, Ho KY, Cheng X, Blumenthal D, Taylor P, Kovalevsky A, Radić Z (2019) Chemico-Biol Interact 309:108698. https://doi.org/10.1016/j.cbi.2019.06.011
Houghton PJ, Ren Y, Howes MJ (2006) Nat Prod Rep 23:181. https://doi.org/10.1039/B508966M
Mukherjee PK, Kumar V, Mal M, Houghton PJ (2007) Phytomedicine 14:289. https://doi.org/10.1016/j.phymed.2007.02.002
Konrath EL, Passos CDS, Klein-Júnior LC, Henriques AT (2013) J Pharm Pharmacol 65(12):1701. https://doi.org/10.1111/jphp.12090
Burke MD, Schreiber SL (2004) Angew Chem Int Ed 43(1):46. https://doi.org/10.1002/anie.200300626
Newman DJ, Cragg GM (2020) J Nat Prod 83(3):770. https://doi.org/10.1021/acs.jnatprod.9b01285
Davison EK, Brimble MA (2019) Curr Opin Chem Biol 52:1. https://doi.org/10.1016/j.cbpa.2018.12.007
García ME, Borioni JL, Cavallaro V, Puiatti M, Pierini AB, Murray AP, Peñéñory AB (2015) Steroids 104:95. https://doi.org/10.1016/j.steroids.2015.09.001
Sauvaître T, Barlier M, Herlem D, Gresh N, Chiaroni A, Guenard D, Guillou C (2007) J Med Chem 50(22):5311. https://doi.org/10.1021/jm070536w
Gilson M, Straatsma T, McCammon J, Ripoll D, Faerman C, Axelsen P, Silman I, Sussman J (1994) Science 263(5151):1276. https://doi.org/10.1126/science.8122110
Cheng S, Song W, Yuan X, Xu Y (2017) Sci Rep 7:3219. https://doi.org/10.1038/s41598-017-03088-y
Bermudez-Lugo JA, Rosales-Hernandez MC, Deeb O, Trujillo-Ferrara J, Correa-Basurto J (2011) Curr Med Chem 18(8):1122. https://doi.org/10.2174/092986711795029681
Daoud I, Melkemi N, Salah T, Ghalem S (2018) Comput Biol Chem 74:304. https://doi.org/10.1016/j.compbiolchem.2018.03.021
Cavas L, Topcam G, Gundogdu-Hizliates C, Ergun Y (2019) Interdisciplinary Sci 11(1):95. https://doi.org/10.1007/s12539-017-0245-4
Hansson T, Marelius J, Åqvist J (1998) J Comput-Aid Mol Des 12(1):27. https://doi.org/10.1023/A:1007930623000
Ul Haq Z, Halim SA, Uddin R, Madura JD (2010) J Mol Gr Model 28(8):870. https://doi.org/10.1016/j.jmgm.2010.03.007
Lan NT, Vu KB, Dao Ngoc MK, Tran PT, Hiep DM, Tung NT, Ngo ST (2019) J Mol Gr Model 93:107441. https://doi.org/10.1016/j.jmgm.2019.107441
Nascimento ÉCM, Oliva M, Andrés J (2018) J Comput-Aid Mol Des 32(5):607. https://doi.org/10.1007/s10822-018-0114-1
Ngo ST, Mai BK, Hiep DM, Li MS (2015) Chem Biol Drug Des 86(4):546. https://doi.org/10.1111/cbdd.12518
Myers J, Grothaus G, Narayanan S, Onufriev A (2006) Proteins 63(4):928. https://doi.org/10.1002/prot.20922
Anandakrishnan R, Aguilar B, Onufriev AV (2012) Nucleic Acids Res 40(W1):W537. https://doi.org/10.1093/nar/gks375
Szegezdi J, Csizmadia F (2007) American chemical society spring meeting
Boström J, Greenwood JR, Gottfries J (2003) J Mol Gr Model 21(5):449. https://doi.org/10.1016/S1093-3263(02)00204-8
McGann MR, Almond HR, Nicholls A, Grant JA, Brown FK (2003) Biopolymers 68(1):76. https://doi.org/10.1002/bip.10207
McGaughey GB, Sheridan RP, Bayly CI, Culberson JC, Kreatsoulas C, Lindsley S, Maiorov V, Truchon JF, Cornell WD (2007) J Chem Inf Model 47(4):1504. https://doi.org/10.1021/ci700052x
Humphrey W, Dalke A, Schulten K (1996) J Mol Gr 14(1):33. https://doi.org/10.1016/0263-7855(96)00018-5
Wang J, Wolf RM, Caldwell JW, Kollman PA, Case DA (2004) J Comput Chem 25(9):1157. https://doi.org/10.1002/jcc.20035
Jakalian A, Bush BL, Jack DB, Bayly CI (2000) J Comput Chem 21(2):132. https://doi.org/10.1002/(SICI)1096-987X(20000130)21:2<132::AID-JCC5>3.0.CO;2-P
Wang J, Cieplak P, Kollman PA (2000) J Comput Chem 21(12):1049. https://doi.org/10.1002/1096-987X(200009)21:12<1049::AID-JCC3>3.0.CO;2-F
Case D, Darden TA, Cheatham I, Simmerling CL, Wang J, Duke RE et al (2010) AMBER11. University of California, San Francisco
Jorgensen WL, Chandrasekhar J, Madura JD, Impey RW, Klein ML (1983) J Chem Phys 79(2):926. https://doi.org/10.1063/1.445869
Phillips JC, Braun R, Wang W, Gumbart J, Tajkhorshid E, Villa E, Chipot C, Skeel RD, Kalé L, Schulten K (2005) J Comput Chem 26(16):1781. https://doi.org/10.1002/jcc.20289
Golosov AA, Karplus M (2007) J Phys Chem B 111(6):1482. https://doi.org/10.1021/jp065493u
Kollman PA, Massova I, Reyes C, Kuhn B, Huo S, Chong L, Lee M, Lee T, Duan Y, Wang W, Donini O, Cieplak P, Srinivasan J, Case DA, Cheatham TE (2000) Acc Chem Res 33(12):889. https://doi.org/10.1021/ar000033j
Wang C, Nguyen PH, Pham K, Huynh D, Le TBN, Wang H, Ren P, Luo R (2016) J Comput Chem 37(27):2436. https://doi.org/10.1002/jcc.24467
Ellman GL, Courtney KD, Andresjr V, Featherstone RM (1961) Biochem Pharmacol 7(2):88. https://doi.org/10.1016/0006-2952(61)90145-9
Choudhary MI, Devkota K, Nawaz SA, Shaheen F, ur Rahman A (2004) Helvetica Chimica Acta 87(5):1099. https://doi.org/10.1002/hlca.200490100
ur Rahman A, ul Haq Z, Khalid A, Anjum S, Khan MR, Choudhary MI (2002) Helvetica Chimica Acta 85(2):678. https://doi.org/10.1002/1522-2675(200202)85:2<678::AID-HLCA678>3.0.CO;2-2
Rouleau J, Iorga BI, Guillou C (2011) Eur J Med Chem 46(6):2193. https://doi.org/10.1016/j.ejmech.2011.02.073
Genheden S, Ryde U (2015) Expert Opin Drug Discov 10(5):449. https://doi.org/10.1517/17460441.2015.1032936
Yang CY, Wang S (2010) ACS Med Chem Lett 1(3):125. https://doi.org/10.1021/ml100026a
Hou T, Wang J, Li Y, Wang W (2011) J Chem Inf Model 51(1):69. https://doi.org/10.1021/ci100275a
Weissenberg M (2001) Phytochemistry 58(3):501. https://doi.org/10.1016/S0031-9422(01)00185-6
Bhattacharya S, Kohli S, Chaudhary AS (2013) Aust-Asian J Cancer 12(3):199
Acknowledgements
We would like to thank E. Daiann Sosa Carrizo for helpful comments and discussions and F. Brigante for the revision of this manuscript. This work was supported in part by the Consejo Nacional de Investigaciones Científicas y Tecnológicas (CONICET), the Agencia Nacional de Promoción Científica y Tecnológica (FONCYT, Argentina), the Agencia Córdoba Ciencia and the Secretaría de Ciencia y Técnica (SECYT) of the Universidad Nacional de Córdoba. INFIQC and IMBIV are jointly sponsored by CONICET and the Universidad Nacional de Córdoba. All calculations were performed with computational resources from the Centro de Computación de Alto Desempeño—Universidad Nacional de Córdoba (http://ccad.unc.edu.ar/), in particular the Mendieta Cluster that belongs to the Facultad de Matemática, Astronomía y Física, that is also part of SNCAD, República Argentina.
Author information
Authors and Affiliations
Corresponding author
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Borioni, J.L., Cavallaro, V., Pierini, A.B. et al. An activity prediction model for steroidal and triterpenoidal inhibitors of Acetylcholinesterase enzyme. J Comput Aided Mol Des 34, 1079–1090 (2020). https://doi.org/10.1007/s10822-020-00324-y
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-020-00324-y