Abstract
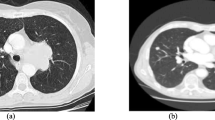
Interstitial Lung Disease (ILD) encompasses a wide array of diseases that share some common radiologic characteristics. When diagnosing such diseases, radiologists can be affected by heavy workload and fatigue thus decreasing diagnostic accuracy. Automatic segmentation is the first step in implementing a Computer Aided Diagnosis (CAD) that will help radiologists to improve diagnostic accuracy thereby reducing manual interpretation. Automatic segmentation proposed uses an initial thresholding and morphology based segmentation coupled with feedback that detects large deviations with a corrective segmentation. This feedback is analogous to a control system which allows detection of abnormal or severe lung disease and provides a feedback to an online segmentation improving the overall performance of the system. This feedback system encompasses a texture paradigm. In this study we studied 48 males and 48 female patients consisting of 15 normal and 81 abnormal patients. A senior radiologist chose the five levels needed for ILD diagnosis. The results of segmentation were displayed by showing the comparison of the automated and ground truth boundaries (courtesy of ImgTracer™ 1.0, AtheroPoint™ LLC, Roseville, CA, USA). The left lung’s performance of segmentation was 96.52 % for Jaccard Index and 98.21 % for Dice Similarity, 0.61 mm for Polyline Distance Metric (PDM), −1.15 % for Relative Area Error and 4.09 % Area Overlap Error. The right lung’s performance of segmentation was 97.24 % for Jaccard Index, 98.58 % for Dice Similarity, 0.61 mm for PDM, −0.03 % for Relative Area Error and 3.53 % for Area Overlap Error. The segmentation overall has an overall similarity of 98.4 %. The segmentation proposed is an accurate and fully automated system.


















Similar content being viewed by others
References
Schwarz, M. I., Matthay, R. A., Sahn, S. A., Stanford, R. E., Marmorstein, B. L., and Scheinhorn, D. J., Interstitial lung disease in polymyositis and dermatomyositis: analysis of six cases and review of the literature. Medicine 55(1):89–104, 1976.
Peroš-Golubičić, T., and Sharma, O., Clinical atlas of interstitial lung disease. Springer, London, 2006.
Sharman, P., and Wood-Baker, R., Interstitial lung disease due to fumes from heat-cutting polymer rope. Occup. Med. 63(6):451–453, 2013.
O’Dwyer, D. N., Armstrong, M. E., Cooke, G., Dodd, J. D., Veale, D. J., and Donnelly, S. C., Rheumatoid Arthritis (RA) associated interstitial lung disease (ILD). Eur. J. Intern Med. 24(7):597–603, 2013.
Washko, G. R., Hunninghake, G. M., Fernandez, I. E., Nishino, M., Okajima, Y., Yamashiro, T., et al., Lung volumes and emphysema in smokers with interstitial lung abnormalities. N. Engl. J. Med. 364(10):897–906, 2011.
Henne, E., Anderson, J. C., Lowe, N., and Kesten, S., Comparison of human lung tissue mass measurements from ex vivo lungs and high resolution CT software analysis. BMC Pulm. Med. 12(1):18, 2012.
Krupinski, E. A., Berbaum, K. S., Does reader visual fatigue impact interpretation accuracy? Proc. SPIE. Med. Imaging. 7627, 76270M-1-6, 2010.
Jiang, Y., Nishikawa, R. M., Schmidt, R. A., Metz, C. E., Giger, M. L., and Doi, K., Improving breast cancer diagnosis with computer-aided diagnosis. Acad. Radiol. 6(1):22–33, 1999.
Doi, K., Computer-aided diagnosis in medical imaging : Historical review, current status and future potential. Comput. Med. Imaging Graph. 31(4):198–211, 2007. doi:10.1016/j.compmedimag.2007.02.002.
Kobayashi, T., Xu, X. W., MacMahon, H., Metz, C. E., and Doi, K., Effect of a computer-aided diagnosis scheme on radiologists’ performance in detection of lung nodules on radiographs. Radiology 199(3):843–848, 1996.
Nagaraj, S., Rao, G. N., and Koteswararao, K., The role of pattern recognition in computer-aided diagnosis and computer-aided detection in medical imaging: A clinical validation. Int. J. Comput. Appl. 8(5):18–22, 2010.
van Rikxoort, E. M., de Hoop, B., Viergever, M. A., Prokop, M., and van Ginneken, B., Automatic lung segmentation from thoracic computed tomography scans using a hybrid approach with error detection. Med. Phys. 36(7):2934–2947, 2009. doi:10.1118/1.3147146.
van Rikxoort, E. M., and van Ginneken, B., Automated segmentation of pulmonary structures in thoracic computed tomography scans : A review. Phys. Med. Biol. 58(17):R187, 2013. doi:10.1088/0031-9155/58/17/R187.
Sluimer, I., Prokop, M., and van Ginneken, B., Toward automated segmentation of the pathological lung in CT. IEEE Trans. Med. Imaging 24(8):1025–1038, 2005. doi:10.1109/TMI.2005.851757.
El-Baz, A., Suri, J. S., (Eds) Lung Imaging and Computer Aided Diagnosis: CRC Press, Boca Raton, 2011.
Nandy, K., Interactive segmentation and tracking in optical microscopic images. Cytom. Part A 81(5):357–359, 2012.
Otsu, N., A threshold selection method from gray-level histograms. Automatica 11:23–27, 1975.
Churg, A., Thurlbeck’s Pathology of the Lung. Thieme, New York, 2005.
Li, L.-N., Ouyang, J.-H., Chen, H.-L., and Liu, D.-Y., A computer aided diagnosis system for thyroid disease using extreme learning machine. J. Med. Syst. 36(5):3327–3337, 2012. doi:10.1007/s10916-012-9825-3.
Sheikhtaheri, A., Sadoughi, F., and Hashemi, D. Z., Developing and using expert systems and neural networks in medicine: A review on benefits and challenges. J. Med. Syst. 38(9):1–6, 2014. doi:10.1007/s10916-014-0110-5.
Wang, Q., Zhu, W., and Wang, B., Three-Dimensional SVM with Latent Variable: Application for detection of lung lesions in CT images. J. Med. Syst. 39(1):171, 2015. doi:10.1007/s10916-014-0171-5.
Hu, S., Hoffman, E. A., and Reinhardt, J. M., Automatic lung segmentation for accurate quantitation of volumetric X-ray CT images. IEEE Trans. Med. Imaging 20(6):490–498, 2001. doi:10.1109/42.929615.
Itai, Y., Kim, H., Ishikawa, S., Yamamoto, A., and Nakamura, K., A segmentation method of lung areas by using snakes. Int. J. Innov. Comput. Inf. Control 3(2):277–284, 2007.
Boykov, Y., Jolly, M. P., Interactive organ segmentation using graph cuts. In Medical Image Computing and Computer-Assisted Intervention--MICCAI2000, 276–286, 2000.
Osareh, A., and Shadgar, B., A segmentation method of lung cavities using region aided geometric snakes. J. Med. Syst. 34(4):419–433, 2010.
Korfiatis, P., Kalogeropoulou, C., Karahaliou, A., Kazantzi, A., Skiadopoulos, S., and Costaridou, L., Texture classification-based segmentation of lung affected by interstitial pneumonia in high-resolution CT. Med. Phys. 35(12):5290–5302, 2008.
van Rikxoort, E. M., de Hoop, B., van de Vorst, S., Prokop, M., and van Ginneken, B., Automatic segmentation of pulmonary segments from volumetric chest CT scans. IEEE Trans. Med. Imaging 28(4):621–630, 2009. doi:10.1109/TMI.2008.2008968.
Kakar, M., and Olsen, D. R., Automatic segmentation and recognition of lungs and lesion from CT scans of thorax. Comput. Med. Imaging Graph. 33(1):72–82, 2009. doi:10.1016/j.compmedimag.2008.10.009.
Zhou, X., Hayashi, T., Hara, T., Fujita, H., Yokoyama, R., Kiryu, T., and Hoshi, H., Automatic segmentation and recognition of anatomical lung structures from high-resolution chest CT images. Comput. Med. Imaging Graph. 30(5):299–313, 2006. doi:10.1016/j.compmedimag.2006.06.002.
Wang, J., Li, F., and Li, Q., Automated segmentation of lungs with severe interstitial lung disease in CT. Med. Phys. 36(10):4592–9, 2009. doi:10.1118/1.3222872.
Massoptier, L., Misra, A., Sowmya, A., Automatic lung segmentation in HRCT images with diffuse parenchymal lung disease using graph-cut. 24th Inter Conf In Image and Vision Computing New Zealand, 2009. IVCNZ’09. 266–270, 2009.
Abbas, Q., Khan, M. T. A., Farooq, A., and Celebi, M. E., Segmentation of lungs in HRCT scan images using particle swarm optimization. Int. J. Innov. Comput. Inf. Control 9(5):2155–2165, 2013.
Boykov, Y., and Kolmogorov, V., An experimental comparison of min-cut/max-flow algorithms for energy minimization in vision. IEEE Trans. Pattern Anal. Mach. Intell. 26(9):1124–1137, 2004. doi:10.1109/TPAMI.2004.60.
Acknowledgments
We would like to thank all the radiologists and clinicians for making this study a success. We would like to express our gratitude to Mr. Ng Chuen Rue for helping to edit this manuscript. We are grateful to AtheroPoint™ LLC, Roseville, CA, USA for gracefully letting us use ImgTracer™ 1.0 software for tracing the manual borders of the lung. This study was partly funded by Universiti Teknologi Malaysia research fund (06H35).
Author information
Authors and Affiliations
Corresponding author
Additional information
This article is part of the Topical Collection on Systems-Level Quality Improvement
Rights and permissions
About this article
Cite this article
Noor, N.M., Than, J.C.M., Rijal, O.M. et al. Automatic Lung Segmentation Using Control Feedback System: Morphology and Texture Paradigm. J Med Syst 39, 22 (2015). https://doi.org/10.1007/s10916-015-0214-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10916-015-0214-6