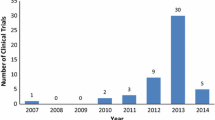
Abstract
Mobile Health Interventions (MHIs) have addressed a range of healthcare challenges and have been evaluated using Randomized Controlled Trials (RCTs) to establish clinical effectiveness. Using PRISMA we conducted a systematic literature review of RCTs for MHIs and identified 70 studies which were analyzed and classified using Nickerson-Varshney-Muntermann (NVM) taxonomy. From the resultant iterations of the taxonomy, we extracted insights from the categorized studies. RCTs cover a wide range of health conditions including chronic diseases, general wellness, unhealthy practices, family planning, end-of-life, and post-transplant care. The MHIs that were utilized by the RCTs were varied as well, although most studies did not find significant differences between MHIs and usual care. The challenges for MHI-based RCTs include the use of technologies, delayed outcomes, patient recruitment, patient retention, and complex regulatory requirements. These variances can lead to a higher rate of Type I/Type II errors. Further considerations are the impact of infrastructure, contextual and cultural factors, and reductions in the technological relevancy of the intervention itself. Finally, due to the delayed effect of most outcomes, RCTs of insufficient duration are unable to measure significant, lasting improvements. Using the insights from seventy identified studies, we developed a classification of existing RCTs along with guidelines for MHI-based RCTs and a research framework for future RCTs. The framework offers opportunities for (a) personalization of MHIs, (b) use of richer technologies, and (c) emerging areas for RCTs.







Similar content being viewed by others
References
S.-I. Kim, H.-S. Kim, Effectiveness of mobile and internet intervention in patients with obese type 2 diabetes, International Journal of Medical Informatics. 77 (2008) 399–404. https://doi.org/10.1016/j.ijmedinf.2007.07.006.
M.-J. Park, H.-S. Kim, Evaluation of mobile phone and Internet intervention on waist circumference and blood pressure in post-menopausal women with abdominal obesity, International Journal of Medical Informatics. 81 (2012) 388–394. https://doi.org/10.1016/j.ijmedinf.2011.12.011.
T.S. Wheeler, T. Michael Vallis, N.B. Giacomantonio, S.R. Abidi, Feasibility and usability of an ontology-based mobile intervention for patients with hypertension, International Journal of Medical Informatics. 119 (2018) 8–16. https://doi.org/10.1016/j.ijmedinf.2018.08.002.
A. Triantafyllidis, H. Kondylakis, K. Votis, D. Tzovaras, N. Maglaveras, K. Rahimi, Features, outcomes, and challenges in mobile health interventions for patients living with chronic diseases: A review of systematic reviews, International Journal of Medical Informatics. 132 (2019) 103984. https://doi.org/10.1016/j.ijmedinf.2019.103984.
D.R. Stukus, N. Farooqui, K.R. Strothman, K. Ryan, J.H. Stevens, D.M. Cohen, Use of a Mobile Health Application in Children with Persistent Asthma: A Randomized Controlled Trial. Journal of Allergy and Clinical Immunology. 139 (2017) AB190.
R.Y. Moon, F.R. Hauck, E.R. Colson, A.L. Kellams, N.L. Geller, T. Heeren, S.M. Kerr, E.E. Drake, K. Tanabe, M. McClain, others, The effect of nursing quality improvement and mobile health interventions on infant sleep practices: a randomized clinical trial, Jama. 318 (2017) 351–359.
L. Afrin, K. Courtney, G. Demiris, W. Goossen, C. Lovis, C. Lynch, M. Sondhi, S. Speedie, V. Vimarlund, Patient-Centered Applications and Mobile Health; Use of IT to Promote Disease Management and Wellness, Journal of the American Medical Informatics Association. 15 (2008) 8–13.
E.M. Geramita, A.J.D. Dabbs, A.F. DiMartini, J.M. Pilewski, G.E. Switzer, D.M. Posluszny, L. Myaskovsky, M.A. Dew, Impact of a mobile health intervention on long-term nonadherence after lung transplantation: follow-up after a randomized controlled trial, Transplantation. 104 (2020) 640–651.
R. Cookson, M. Robson, I. Skarda, T. Doran, Equity-informative methods of health services research, Journal of Health Organization and Management. (2021).
J.-A. Lee, M. Choi, S.A. Lee, N. Jiang, Effective behavioral intervention strategies using mobile health applications for chronic disease management: a systematic review, BMC Medical Informatics and Decision Making. 18 (2018). https://doi.org/10.1186/s12911-018-0591-0.
A.B. Amorim, E. Pappas, M. Simic, M.L. Ferreira, M. Jennings, A. Tiedemann, A.P. Carvalho-e-Silva, E. Caputo, A. Kongsted, P.H. Ferreira, Integrating Mobile-health, health coaching, and physical activity to reduce the burden of chronic low back pain trial (IMPACT): a pilot randomised controlled trial, BMC Musculoskeletal Disorders. 20 (2019) 71.
M. Plow, M. Golding, Using mHealth technology in a self-management intervention to promote physical activity among adults with chronic disabling conditions: randomized controlled trial, JMIR MHealth and UHealth. 5 (2017) e185.
C.H. Lee, B. Cheung, G.-H. Yi, B. Oh, Y.H. Oh, Mobile health, physical activity, and obesity: Subanalysis of a randomized controlled trial, Medicine. 97 (2018).
C.D. Nyström, S. Sandin, P. Henriksson, H. Henriksson, R. Maddison, M. Löf, A 12-month follow-up of a mobile-based (mHealth) obesity prevention intervention in pre-school children: the MINISTOP randomized controlled trial, BMC Public Health. 18 (2018) 658.
B. Oh, G.-H. Yi, M.K. Han, J.S. Kim, C.H. Lee, B. Cho, H.C. Kang, Importance of active participation in obesity management through mobile health care programs: substudy of a randomized controlled trial, JMIR MHealth and UHealth. 6 (2018) e2.
A.L. Schweitzer, J.T. Ross, C.J. Klein, K.Y. Lei, E.R. Mackey, An electronic wellness program to improve diet and exercise in college students: a pilot study, JMIR Research Protocols. 5 (2016) e29.
S.M. Alessi, C.J. Rash, Treatment satisfaction in a randomized clinical trial of mHealth smoking abstinence reinforcement, Journal of Substance Abuse Treatment. 72 (2017) 103–110.
J.E. Glass, J.R. McKay, D.H. Gustafson, R. Kornfield, P.J. Rathouz, F.M. McTavish, A.K. Atwood, A. Isham, A. Quanbeck, D. Shah, Treatment seeking as a mechanism of change in a randomized controlled trial of a mobile health intervention to support recovery from alcohol use disorders, Journal of Substance Abuse Treatment. 77 (2017) 57–66.
M.L. Ybarra, T.L. Prescott, G.L. Phillips, S.S. Bull, J.T. Parsons, B. Mustanski, Pilot RCT results of an mHealth HIV prevention program for sexual minority male adolescents, Pediatrics. 140 (2017).
J. Willcox, S. Wilkinson, M. Lappas, K. Ball, D. Crawford, E. McCarthy, B. Fjeldsoe, R. Whittaker, R. Maddison, K. Campbell. A mobile health intervention promoting healthy gestational weight gain for women entering pregnancy at a high body mass index: the txt4two pilot randomised controlled trial. BJOG: An International Journal of Obstetrics & Gynaecology. 124 (2017) 1718–1728.
A. Kubo, E. Kurtovich, M. McGinnis, S. Aghaee, A. Altschuler, C. Quesenberry Jr, T. Kolevska, A.L. Avins, A randomized controlled trial of mHealth mindfulness intervention for cancer patients and informal cancer caregivers: a feasibility study within an integrated health care delivery system, Integrative Cancer Therapies. 18 (2019) 1534735419850634.
R.I. Vogel, K. Niendorf, S. Petzel, H. Lee, D. Teoh, A.H. Blaes, P. Argenta, C. Rivard, B. Winterhoff, H.Y. Lee, others, A patient-centered mobile health application to motivate use of genetic counseling among women with ovarian cancer: A pilot randomized controlled trial, Gynecologic Oncology. 153 (2019) 100–107.
S. Berrouiguet, M.E. Larsen, C. Mesmeur, M. Gravey, R. Billot, M. Walter, H. Network, C. Lemey, P. Lenca, Toward mHealth brief contact interventions in suicide prevention: case series from the suicide intervention assisted by messages (SIAM) randomized controlled trial, JMIR MHealth and UHealth. 6 (2018) e8.
J. Lorca-Cabrera, C. Grau, R. Martí-Arques, L. Raigal-Aran, A. Falcó-Pegueroles, N. Albacar-Riobóo, Effectiveness of health web-based and mobile app-based interventions designed to improve informal caregiver’s well-being and quality of life: A systematic review, International Journal of Medical Informatics. 134 (2020) 104003. https://doi.org/10.1016/j.ijmedinf.2019.104003.
L. Xue, C.C. Yen, L. Chang, H.C. Chan, B.C. Tai, S.B. Tan, H.B.L. Duh, M. Choolani, An exploratory study of ageing women’s perception on access to health informatics via a mobile phone-based intervention, International Journal of Medical Informatics. 81 (2012) 637–648. https://doi.org/10.1016/j.ijmedinf.2012.04.008.
U. Varshney, Mobile health: Four emerging themes of research, Decision Support Systems. 66 (2014) 20–35. https://doi.org/10.1016/j.dss.2014.06.001.
U. Varshney, Pervasive healthcare and wireless health monitoring, Mobile Networks and Applications. 12 (2007) 113–127.
G. Eysenbach, C.-E. Group, others, CONSORT-EHEALTH: improving and standardizing evaluation reports of Web-based and mobile health interventions, Journal of Medical Internet Research. 13 (2011) e126.
F. Bert, M. Giacometti, M.R. Gualano, R. Siliquini, Smartphones and health promotion: a review of the evidence, Journal of Medical Systems. 38 (2014) 1–11.
B. Rahimi, V. Vimarlund, Methods to evaluate health information systems in healthcare settings: a literature review, Journal of Medical Systems. 31 (2007) 397–432.
Z. Lv, J. Chirivella, P. Gagliardo, Bigdata oriented multimedia mobile health applications, Journal of Medical Systems. 40 (2016) 120.
S.S. Bhuyan, N. Lu, A. Chandak, H. Kim, D. Wyant, J. Bhatt, S. Kedia, C.F. Chang, Use of mobile health applications for health-seeking behavior among US adults, Journal of Medical Systems. 40 (2016) 153.
A.J. Barton, The regulation of mobile health applications, BMC Medicine. 10 (2012) 46.
D. Kotz, S. Avancha, A. Baxi, A privacy framework for mobile health and home-care systems, in: Proceedings of the First ACM Workshop on Security and Privacy in Medical and Home-Care Systems, 2009: pp. 1–12.
C. Doukas, T. Pliakas, I. Maglogiannis, Mobile healthcare information management utilizing Cloud Computing and Android OS, in: 2010 Annual International Conference of the IEEE Engineering in Medicine and Biology, IEEE, 2010: pp. 1037–1040.
I. Asangansi, K. Braa, The emergence of mobile-supported national health information systems in developing countries., in: Medinfo, 2010: pp. 540–544.
B. Martínez-Pérez, I. De La Torre-Díez, M. López-Coronado, Privacy and security in mobile health apps: a review and recommendations, Journal of Medical Systems. 39 (2015) 1–8.
A. Bhide, P.S. Shah, G. Acharya, A simplified guide to randomized controlled trials, Acta Obstetricia et Gynecologica Scandinavica. 97 (2018) 380–387.
S. Hopewell, M. Clarke, D. Moher, E. Wager, P. Middleton, D.G. Altman, K.F. Schulz, and the C. Group, CONSORT for Reporting Randomized Controlled Trials in Journal and Conference Abstracts: Explanation and Elaboration, PLOS Medicine. 5 (2008) e20. https://doi.org/10.1371/journal.pmed.0050020.
D. Moher, S. Hopewell, K.F. Schulz, V. Montori, P.C. Gøtzsche, P.J. Devereaux, D. Elbourne, M. Egger, D.G. Altman, CONSORT 2010 explanation and elaboration: Updated guidelines for reporting parallel group randomised trials, International Journal of Surgery. 10 (2012) 28–55. https://doi.org/10.1016/j.ijsu.2011.10.001.
L. Turner, L. Shamseer, D.G. Altman, L. Weeks, J. Peters, T. Kober, S. Dias, K.F. Schulz, A.C. Plint, D. Moher, Consolidated standards of reporting trials (CONSORT) and the completeness of reporting of randomised controlled trials (RCTs) published in medical journals, Cochrane Database of Systematic Reviews. (2012).
D. Moher, A.R. Jadad, G. Nichol, M. Penman, P. Tugwell, S. Walsh, Assessing the quality of randomized controlled trials: an annotated bibliography of scales and checklists, Controlled Clinical Trials. 16 (1995) 62–73.
D. Moher, L. Shamseer, M. Clarke, D. Ghersi, A. Liberati, M. Petticrew, L. Stewart, Preferred reporting items for systematic review and meta-analysis protocols (PRISMA-P) 2015 statement. Systematic reviews, 4(1) 1 (2015). https://doi.org/10.1136/bmj.g7647.
A. Liberati, D.G. Altman, J. Tetzlaff, C. Mulrow, P.C. Gøtzsche, J.P.A. Ioannidis, M. Clarke, P.J. Devereaux, J. Kleijnen, D. Moher, The PRISMA Statement for Reporting Systematic Reviews and Meta-Analyses of Studies That Evaluate Health Care Interventions: Explanation and Elaboration, PLOS Medicine. 6 (2009) e1000100. https://doi.org/10.1371/journal.pmed.1000100.
R.C. Nickerson, U. Varshney, J. Muntermann, A method for taxonomy development and its application in information systems, European Journal of Information Systems. 22 (2013) 336–359.
N. Singh, U. Varshney, IT-based reminders for medication adherence: systematic review, taxonomy, framework and research directions, European Journal of Information Systems. 29 (2020) 84–108. https://doi.org/10.1080/0960085X.2019.1701956.
D.L. Finfgeld, Metasynthesis: The State of the Art—So Far, Qual Health Res. 13 (2003) 893–904. https://doi.org/10.1177/1049732303253462.
D. Finfgeld-Connett, Use of content analysis to conduct knowledge-building and theory-generating qualitative systematic reviews, Qualitative Research. 14 (2014) 341–352. https://doi.org/10.1177/1468794113481790.
E. Barnett-Page, J. Thomas, Methods for the synthesis of qualitative research: a critical review, BMC Med Res Methodol. 9 (2009) 59. https://doi.org/10.1186/1471-2288-9-59.
M.B. Miles, A.M. Huberman, J. Saldana, Qualitative Data Analysis, SAGE, 2014.
O.S. Albahri, A.S. Albahri, K. Mohammed, A. Zaidan, B. Zaidan, M. Hashim, O.H. Salman, Systematic review of real-time remote health monitoring system in triage and priority-based sensor technology: Taxonomy, open challenges, motivation and recommendations, Journal of Medical Systems. 42 (2018) 1–27.
H.M. Hussien, S.M. Yasin, S. Udzir, A.A. Zaidan, B.B. Zaidan, A systematic review for enabling of develop a blockchain technology in healthcare application: taxonomy, substantially analysis, motivations, challenges, recommendations and future direction, Journal of Medical Systems. 43 (2019) 1–35.
E. Nabovati, H. Vakili-Arki, Z. Taherzadeh, M.R. Saberi, S. Medlock, A. Abu-Hanna, S. Eslami, Information technology-based interventions to improve drug-drug interaction outcomes: a systematic review on features and effects, Journal of Medical Systems. 41 (2017) 1–17.
K.D. Bailey, Typologies and taxonomies: An introduction to classification techniques, Sage, 1994.
S. Agarwal, A.E. LeFevre, J. Lee, K. L’Engle, G. Mehl, C. Sinha, A. Labrique, Guidelines for reporting of health interventions using mobile phones: mobile health (mHealth) evidence reporting and assessment (mERA) checklist, Bmj. 352 (2016) i1174.
J. Zhao, B. Freeman, M. Li, Can mobile phone apps influence people’s health behavior change? An evidence review, Journal of Medical Internet Research. 18 (2016) e287.
S.J. Iribarren, K. Cato, L. Falzon, P.W. Stone, What is the economic evidence for mHealth? A systematic review of economic evaluations of mHealth solutions, PloS One. 12 (2017) e0170581.
S. Kim, T.H. Baek, Examining the antecedents and consequences of mobile app engagement, Telematics and Informatics. 35 (2018) 148–158.
D.K. Owens, K.N. Lohr, D. Atkins, J.R. Treadwell, J.T. Reston, E.B. Bass, S. Chang, M. Helfand, AHRQ Series Paper 5: Grading the strength of a body of evidence when comparing medical interventions—Agency for Healthcare Research and Quality and the Effective Health-Care Program, Journal of Clinical Epidemiology. 63 (2010) 513–523. https://doi.org/10.1016/j.jclinepi.2009.03.009.
D. Atkins, S.M. Chang, G. Gartlehner, D.I. Buckley, E.P. Whitlock, E. Berliner, D. Matchar, Assessing applicability when comparing medical interventions: AHRQ and the Effective Health Care Program, Journal of Clinical Epidemiology. 64 (2011) 1198–1207. https://doi.org/10.1016/j.jclinepi.2010.11.021.
K. Singh, K. Drouin, L.P. Newmark, R. Rozenblum, J. Lee, A. Landman, E. Pabo, E.V. Klinger, D.W. Bates, Developing a framework for evaluating the patient engagement, quality, and safety of mobile health applications, Issue Brief (Common Fund). 5 (2016) 11.
J.L. Baldwin, H. Singh, D.F. Sittig, T.D. Giardina, Patient portals and health apps: Pitfalls, promises, and what one might learn from the other, in: Healthcare, Elsevier, 2017: pp. 81–85.
R.V. Tuckson, M. Edmunds, M.L. Hodgkins, Telehealth, New England Journal of Medicine. 377 (2017) 1585–1592.
K. Singh, K. Drouin, L.P. Newmark, J. Lee, A. Faxvaag, R. Rozenblum, E.A. Pabo, A. Landman, E. Klinger, D.W. Bates, Many mobile health apps target high-need, high-cost populations, but gaps remain, Health Affairs. 35 (2016) 2310–2318.
L. Minary, J. Trompette, J. Kivits, L. Cambon, C. Tarquinio, F. Alla, Which design to evaluate complex interventions? Toward a methodological framework through a systematic review, BMC Med Res Methodol. 19 (2019) 92. https://doi.org/10.1186/s12874-019-0736-6.
R. Ryan, S. Booth, A. Spathis, S. Mollart, A. Clow, Use of salivary diurnal cortisol as an outcome measure in randomised controlled trials: a systematic review, Annals of Behavioral Medicine. 50 (2016) 210–236.
C.D. Kelly-Cirino, J. Nkengasong, H. Kettler, I. Tongio, F. Gay-Andrieu, C. Escadafal, P. Piot, R.W. Peeling, R. Gadde, C. Boehme, Importance of diagnostics in epidemic and pandemic preparedness, BMJ Global Health. 4 (2019) e001179. https://doi.org/10.1136/bmjgh-2018-001179.
L.O. Gostin, J.G. Hodge, S.A. Noe, Reframing the Opioid Epidemic as a National Emergency, JAMA. 318 (2017) 1539–1540. https://doi.org/10.1001/jama.2017.13358.
M. Prince, V. Patel, S. Saxena, M. Maj, J. Maselko, M.R. Phillips, A. Rahman, No health without mental health, The Lancet. 370 (2007) 859–877. https://doi.org/10.1016/S0140-6736(07)61238-0.
D.E. Bloom, D. Canning, A. Lubet, Global Population Aging: Facts, Challenges, Solutions & Perspectives, Daedalus. 144 (2015) 80–92. https://doi.org/10.1162/DAED_a_00332.
I.D. Coulter, E.M. Willis, The rise and rise of complementary and alternative medicine: a sociological perspective, Medical Journal of Australia. 180 (2004) 587–589. https://doi.org/10.5694/j.1326-5377.2004.tb06099.x.
K. Lakshminarayan, S. Westberg, C. Northuis, C.C. Fuller, F. Ikramuddin, M. Ezzeddine, J. Scherber, S. Speedie, A mHealth-based care model for improving hypertension control in stroke survivors: Pilot RCT, Contemporary Clinical Trials. 70 (2018) 24–34.
A. Rubinstein, J.J. Miranda, A. Beratarrechea, F. Diez-Canseco, R. Kanter, L. Gutierrez, A. Bernabé-Ortiz, V. Irazola, A. Fernandez, P. Letona, others, Effectiveness of an mHealth intervention to improve the cardiometabolic profile of people with prehypertension in low-resource urban settings in Latin America: a randomised controlled trial, The Lancet Diabetes & Endocrinology. 4 (2016) 52–63.
M. Allman-Farinelli, S.R. Partridge, K. McGeechan, K. Balestracci, L. Hebden, A. Wong, P. Phongsavan, E. Denney-Wilson, M.F. Harris, A. Bauman, A mobile health lifestyle program for prevention of weight gain in young adults (TXT2BFiT): Nine-month outcomes of a randomized controlled trial, JMIR MHealth and UHealth. 4 (2016) e78.
H. Amoakoh, K. Klipstein-Grobusch, I. Agyepong, P. Zuithoff, M. Amoakoh-Coleman, G. Kayode, C. Sarpong, J. Reitsma, D. Grobbee, E. Ansah, The effect of an mhealth clinical decision-making support system on neonatal mortality in Ghana, European Journal of Public Health. 29 (2019) ckz185–131.
S. Arora, A.L. Peters, C. Agy, M. Menchine, A mobile health intervention for inner city patients with poorly controlled diabetes: proof-of-concept of the TExT-MED program, Diabetes Technology & Therapeutics. 14 (2012) 492–496.
D. Ben-Zeev, R.M. Brian, G. Jonathan, L. Razzano, N. Pashka, E. Carpenter-Song, R.E. Drake, E.A. Scherer, Mobile health (mHealth) versus clinic-based group intervention for people with serious mental illness: a randomized controlled trial, Psychiatric Services. 69 (2018) 978–985.
B.D. Duscha, L.W. Piner, M.P. Patel, L.E. Crawford, W.S. Jones, M.R. Patel, W.E. Kraus, Effects of a 12-week mHealth program on Functional Capacity and physical activity in patients with peripheral Artery disease, The American Journal of Cardiology. 122 (2018) 879–884.
J.M. Ferrante, K.A. Devine, A. Bator, A. Rodgers, P.A. Ohman-Strickland, E.V. Bandera, K.O. Hwang, Feasibility and potential efficacy of commercial mHealth/eHealth tools for weight loss in African American breast cancer survivors: pilot randomized controlled trial, Translational Behavioral Medicine. (2018).
P. Gamito, J. Oliveira, P. Lopes, R. Brito, D. Morais, D. Silva, A. Silva, S. Rebelo, M. Bastos, A. Deus, Executive functioning in alcoholics following an mHealth cognitive stimulation program: randomized controlled trial, Journal of Medical Internet Research. 16 (2014) e102.
P. Ning, P. Cheng, D.C. Schwebel, Y. Yang, R. Yu, J. Deng, S. Li, G. Hu, An app-based intervention for caregivers to prevent unintentional injury among preschoolers: cluster randomized controlled trial, JMIR MHealth and UHealth. 7 (2019) e13519.
E.M. Giesbrecht, W.C. Miller, Effect of an mHealth Wheelchair Skills Training Program for Older Adults: A Feasibility Randomized Controlled Trial, Archives of Physical Medicine and Rehabilitation. 100 (2019) 2159–2166.
S. Grant, J. Hodgkinson, C. Schwartz, P. Bradburn, M. Franssen, F.R. Hobbs, S. Jowett, R.J. McManus, S. Greenfield, Using mHealth for the management of hypertension in UK primary care: an embedded qualitative study of the TASMINH4 randomised controlled trial, British Journal of General Practice. 69 (2019) e612–e620.
B.J. Hall, P. Xiong, X. Guo, E.K.L. Sou, U.I. Chou, Z. Shen, An evaluation of a low intensity mHealth enhanced mindfulness intervention for Chinese university students: A randomized controlled trial, Psychiatry Research. 270 (2018) 394–403.
E.K. Harrington, A.L. Drake, D. Matemo, K. Ronen, A.O. Osoti, G. John-Stewart, J. Kinuthia, J.A. Unger, An mHealth SMS intervention on postpartum contraceptive use among women and couples in Kenya: a randomized controlled trial, American Journal of Public Health. 109 (2019) 934–941.
L. Hebden, A. Cook, H. Van Der Ploeg, L. King, A. Bauman, M. Allman-Farinelli, A mobile health intervention for weight management among young adults: a pilot randomised controlled trial, Journal of Human Nutrition and Dietetics. 27 (2014) 322–332.
H. Holmen, A. Torbjørnsen, A.K. Wahl, A.K. Jenum, M.C. Sm\aastuen, E. \AArsand, L. Ribu, A mobile health intervention for self-management and lifestyle change for persons with type 2 diabetes, part 2: one-year results from the Norwegian randomized controlled trial RENEWING HEALTH, JMIR MHealth and UHealth. 2 (2014) e57.
D. Johnson, R. Juras, P. Riley, M. Chatterji, P. Sloane, S.K. Choi, B. Johns, A randomized controlled trial of the impact of a family planning mHealth service on knowledge and use of contraception, Contraception. 95 (2017) 90–97.
K.A. Kannisto, J. Korhonen, C.E. Adams, M.H. Koivunen, T. Vahlberg, M.A. Välimäki, Factors associated with dropout during recruitment and follow-up periods of a mHealth-based randomized controlled trial for Mobile. Net to encourage treatment adherence for people with serious mental health problems, Journal of Medical Internet Research. 19 (2017) e46.
N.J. Kleinman, A. Shah, S. Shah, S. Phatak, V. Viswanathan, Improved medication adherence and frequency of blood glucose self-testing using an m-Health platform versus usual care in a multisite randomized clinical trial among people with type 2 diabetes in India, Telemedicine and E-Health. 23 (2017) 733–740.
R.C. Kosse, M.L. Bouvy, T.W. de Vries, E.S. Koster, Effect of a mHealth intervention on adherence in adolescents with asthma: a randomized controlled trial, Respiratory Medicine. 149 (2019) 45–51.
J.T. Koufopoulos, M.T. Conner, P.H. Gardner, I. Kellar, A web-based and mobile health social support intervention to promote adherence to inhaled asthma medications: randomized controlled trial, Journal of Medical Internet Research. 18 (2016) e122.
M.A. Kryger, T.M. Crytzer, A. Fairman, E.J. Quinby, M. Karavolis, G. Pramana, I.M.A. Setiawan, G.P. McKernan, B. Parmanto, B.E. Dicianno, The effect of the interactive mobile health and rehabilitation system on health and psychosocial outcomes in spinal cord injury: Randomized controlled trial, Journal of Medical Internet Research. 21 (2019) e14305.
R.L.-T. Lee, C. Leung, H. Chen, L.H. Louie, M. Brown, J.-L. Chen, G. Cheung, P.H. Lee, The impact of a school-based weight management program involving parents via mHealth for overweight and obese children and adolescents with intellectual disability: a randomized controlled trial, International Journal of Environmental Research and Public Health. 14 (2017) 1178.
A.Y. Liu, E. Vittinghoff, P. von Felten, K. Rivet Amico, P.L. Anderson, R. Lester, E. Andrew, I. Estes, P. Serrano, J. Brothers, others, Randomized controlled trial of a mobile health intervention to promote retention and adherence to preexposure prophylaxis among young people at risk for human immunodeficiency virus: The EPIC Study, Clinical Infectious Diseases. 68 (2019) 2010–2017.
Y.-C. Liu, C.-H. Chen, Y.-C. Tsou, Y.-S. Lin, H.-Y. Chen, J.-Y. Yeh, S.Y.-H. Chiu, Evaluating mobile health apps for customized dietary recording for young adults and seniors: randomized controlled trial, JMIR MHealth and UHealth. 7 (2019) e10931.
M. Lozano-Lozano, I. Cantarero-Villanueva, L. Martin-Martin, N. Galiano-Castillo, M.-J. Sanchez, C. Fernández-Lao, P. Postigo-Martin, M. Arroyo-Morales, A Mobile System to Improve Quality of Life Via Energy Balance in Breast Cancer Survivors (BENECA mHealth): Prospective Test-Retest Quasiexperimental Feasibility Study, JMIR MHealth and UHealth. 7 (2019) e14136.
E.B. Lynch, R. Liebman, J. Ventrelle, E.F. Avery, D. Richardson, Peer reviewed: A self-management intervention for African Americans with comorbid diabetes and hypertension: A pilot randomized controlled trial, Preventing Chronic Disease. 11 (2014).
S.R. Partridge, K. McGeechan, L. Hebden, K. Balestracci, A.T. Wong, E. Denney-Wilson, M.F. Harris, P. Phongsavan, A. Bauman, M. Allman-Farinelli, Effectiveness of a mHealth lifestyle program with telephone support (TXT2BFiT) to prevent unhealthy weight gain in young adults: randomized controlled trial, JMIR MHealth and UHealth. 3 (2015) e66.
M.M. MacPherson, K.J. Merry, S.R. Locke, M.E. Jung, Effects of Mobile Health prompts on self-monitoring and exercise behaviors following a diabetes prevention program: secondary analysis from a randomized controlled trial, JMIR MHealth and UHealth. 7 (2019) e12956.
T.J. Somers, S.A. Kelleher, C.S. Dorfman, R.A. Shelby, H.M. Fisher, K.R. Nichols, K.M. Sullivan, N.J. Chao, G.P. Samsa, A.P. Abernethy, others, An mHealth pain coping skills training intervention for hematopoietic stem cell transplantation patients: development and pilot randomized controlled trial, JMIR MHealth and UHealth. 6 (2018) e66.
S.S. Martin, D.I. Feldman, R.S. Blumenthal, S.R. Jones, W.S. Post, R.A. McKibben, E.D. Michos, C.E. Ndumele, E.V. Ratchford, J. Coresh, others, mActive: a randomized clinical trial of an automated mHealth intervention for physical activity promotion, Journal of the American Heart Association. 4 (2015) e002239.
J.W. McGillicuddy, M.J. Gregoski, A.K. Weiland, R.A. Rock, B.M. Brunner-Jackson, S.K. Patel, B.S. Thomas, D.J. Taber, K.D. Chavin, P.K. Baliga, others, Mobile health medication adherence and blood pressure control in renal transplant recipients: a proof-of-concept randomized controlled trial, JMIR Research Protocols. 2 (2013) e32.
W.J. Meurer, M. Dome, D. Brown, D. Delemos, S. Oska, V. Gorom, L. Skolarus, Feasibility of emergency department–initiated, mobile health blood pressure intervention: an exploratory, randomized clinical trial, Academic Emergency Medicine. 26 (2019) 517–527.
J.W. Mitchell, S. Lammert, T. Danh, K.J. Horvath, Optimizing enrollment methods: lessons learned from an mHealth, pilot HIV prevention randomized controlled trial with sexual minority men, International Journal of STD & AIDS. 30 (2019) 703–706.
H. Nawaiseh, W. McIntosh, L. McKyer, Effectiveness of m-Health Application for Improving Healthy Lifestyles in Healthy College Students: A randomized controlled trial, in: APHA’s 2019 Annual Meeting and Expo (Nov. 2-Nov. 6), American Public Health Association, 2019.
S.R. Partridge, K. McGeechan, A. Bauman, P. Phongsavan, M. Allman-Farinelli, Improved eating behaviours mediate weight gain prevention of young adults: moderation and mediation results of a randomised controlled trial of TXT2BFiT, mHealth program, International Journal of Behavioral Nutrition and Physical Activity. 13 (2016) 44.
D. Peiris, D. Praveen, K. Mogulluru, M.A. Ameer, A. Raghu, Q. Li, S. Heritier, S. MacMahon, D. Prabhakaran, G.D. Clifford, others, SMARThealth India: a stepped-wedge, cluster randomised controlled trial of a community health worker managed mobile health intervention for people assessed at high cardiovascular disease risk in rural India, PloS One. 14 (2019) e0213708.
R.J. Petrella, M.I. Stuckey, S. Shapiro, D.P. Gill, Mobile health, exercise and metabolic risk: a randomized controlled trial, BMC Public Health. 14 (2014) 1082.
A. Torbjørnsen, A.K. Jenum, M.C. Sm\aastuen, E. \AArsand, H. Holmen, A.K. Wahl, L. Ribu, A low-intensity mobile health intervention with and without health counseling for persons with type 2 diabetes, part 1: baseline and short-term results from a randomized controlled trial in the Norwegian part of RENEWING HEALTH, JMIR MHealth and UHealth. 2 (2014) e52.
S.G. Quiñonez, M.J.L. Walthouwer, D.N. Schulz, H. de Vries, mHealth or eHealth? Efficacy, use, and appreciation of a Web-based computer-tailored physical activity intervention for Dutch adults: a randomized controlled trial, Journal of Medical Internet Research. 18 (2016) e278.
S. Saleh, A. Farah, H. Dimassi, N. El Arnaout, J. Constantin, M. Osman, C. El Morr, M. Alameddine, Using mobile health to enhance outcomes of noncommunicable diseases care in rural settings and refugee camps: randomized controlled trial, JMIR MHealth and UHealth. 6 (2018) e137.
E.M. Rosenberger, A. DeVito Dabbs, A.F. DiMartini, D.P. Landsittel, J.M. Pilewski, M.A. Dew, Long-term follow-up of a randomized controlled trial evaluating a mobile health intervention for self-management in lung transplant recipients, American Journal of Transplantation. 17 (2017) 1286–1293.
D.N. Schulz, S.P. Kremers, C. Vandelanotte, M.J. Van Adrichem, F. Schneider, M.J. Candel, H. de Vries, Effects of a web-based tailored multiple-lifestyle intervention for adults: a two-year randomized controlled trial comparing sequential and simultaneous delivery modes, Journal of Medical Internet Research. 16 (2014) e26.
A. Serlachius, K. Schache, A. Kieser, B. Arroll, K. Petrie, N. Dalbeth, Association between user engagement of a mobile health app for gout and improvements in self-care behaviors: randomized controlled trial, JMIR MHealth and UHealth. 7 (2019) e15021.
D. Shellmer, A.D. Dabbs, M. Dew, B. Feingold, G. Mazariegos, R. Shapiro, A Randomized Controlled Trial of a Dyadic Mobile Health Application to Promote Medication Adherence Among Solid Organ Adolescent Transplant Patients.: Abstract# A484, Transplantation. 98 (2014) 237–238.
S. Shorey, Y. Lau, C.-L. Dennis, Y.S. Chan, W.W. Tam, Y.H. Chan, A randomized-controlled trial to examine the effectiveness of the ‘Home-but not Alone’mobile-health application educational programme on parental outcomes, Journal of Advanced Nursing. 73 (2017) 2103–2117.
B. Spring, C. Pellegrini, H. McFadden, A.F. Pfammatter, T.K. Stump, J. Siddique, A.C. King, D. Hedeker, Multicomponent mHealth intervention for large, sustained change in multiple diet and activity risk behaviors: the make better choices 2 randomized controlled trial, Journal of Medical Internet Research. 20 (2018) e10528.
A. Sridhar, A. Chen, D. Glik, Plan a birth control: randomized controlled trial of a mobile health application, Contraception. 88 (2013) 463.
R. Sutherland, N. Nathan, A. Brown, S. Yoong, M. Finch, C. Lecathelinais, R. Reynolds, A. Walton, L. Janssen, C. Desmet, others, A randomized controlled trial to assess the potential efficacy, feasibility and acceptability of an m-health intervention targeting parents of school aged children to improve the nutritional quality of foods packed in the lunchbox ‘SWAP IT,’ International Journal of Behavioral Nutrition and Physical Activity. 16 (2019) 54.
J. Tighe, F. Shand, R. Ridani, A. Mackinnon, N. De La Mata, H. Christensen, Ibobbly mobile health intervention for suicide prevention in Australian Indigenous youth: a pilot randomised controlled trial, BMJ Open. 7 (2017) e013518.
A. Torbjørnsen, M.C. Sm\aastuen, A.K. Jenum, E. \AArsand, L. Ribu, Acceptability of an mHealth app intervention for persons with type 2 diabetes and its associations with initial self-management: randomized controlled trial, JMIR MHealth and UHealth. 6 (2018) e125.
C. Sun, L. Sun, S. Xi, H. Zhang, H. Wang, Y. Feng, Y. Deng, H. Wang, X. Xiao, G. Wang, others, Mobile phone–based telemedicine practice in older chinese patients with type 2 diabetes mellitus: randomized controlled trial, JMIR MHealth and UHealth. 7 (2019) e10664.
S. Yu, Z. Duan, P.B. Redmon, M.P. Eriksen, J.P. Koplan, C. Huang, mHealth intervention is effective in creating smoke-free homes for newborns: A randomized controlled trial study in China, Scientific Reports. 7 (2017) 1–9.
X. Zhu, W. Zhang, D. Operario, Y. Zhao, A. Shi, Z. Zhang, P. Gao, A. Perez, J. Wang, N. Zaller, others, Effects of a mobile health intervention to promote HIV self-testing with MSM in China: a randomized controlled trial, AIDS and Behavior. 23 (2019) 3129–3139.
Funding
This research did not receive any specific grant from funding agencies in the public, commercial, or not-for-profit sectors.
Author information
Authors and Affiliations
Contributions
All authors contributed in identifying the research objective and strategy, conducted the data gathering, systematic review of articles, analysis for taxonomy development, identification of challenges for MHI-based RCTS followed by development of guidelines, and development of research framework as well as the article preparation and write-up.
Corresponding author
Ethics declarations
Competing interests
The authors have no competing interests to declare.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
This article is part of the Topical Collection on Mobile & Wireless Health
Appendices
Appendices
Appendix A. Systematic literature review of 70 articles
# | Article | Intervention Objective | Condition | Findings and Limitation |
|---|---|---|---|---|
1 | [17] | Incentives for being smoking-negative | Smoking cessation | mHealth reinforcement led to longest durations of abstinence |
2 | [74] | Nutritional recommendations for fruits, vegs, and takeout meals | Obesity prevention | Intervention group met nutrition recommendations more frequently |
3 | [75] | Effectiveness of mHealth as a decision support intervention | Neonatal mortality | Intervention arm had increased odds of neonatal death (2.09) due to multiple confounding factors |
4 | [11] | Effectiveness of coaching and mobile health in reducing care-seeking, pain, and disability | Chronic low back pain | 38% reduction in care-seeking for the Intervention group |
5 | [76] | Intervention for clinical outcomes and healthy behaviors | Diabetes patients | Hemoglobin levels decreased in intervention group by 1.05% and 0.6% in control: not significant |
6 | [77] | Intervention for engagement, satisfaction, improvement in clinical symptoms and QoL | Mental illness | Higher participation for treatment (90 to 58) and engagement (56 to 40) and no significant outcome |
7 | [23] | Effectiveness of messaging for suicide prevention | Suicide prevention | Message contact can reconnect individuals with support services |
8 | [78] | Effectiveness of activity trackers and coaching for sustaining fitness & physical activity | cardiac rehabilitation | Mhealth increased moderate high physical activity |
9 | [79] | Effectiveness of activity tracker for weight management | Weight management | Significant in waist circumference, logged food per week and weight loss |
10 | [80] | Mobile games for decreasing alcohol dependence using cognitive simulation | Alcohol dependence | Frontal lobe function (executive decision) increases in the intervention group |
11 | [8] | To study adherence of recipients after trial period | Lung transplant | No sustained impact. Age, graft rejection and chronic anxiety associated with nonadherence |
12 | [81] | Effectiveness of an informational m-health app for child safety | Car seat child safety | Significant improvement in car seat type (1.84) and inspection (1.73) |
13 | [82] | mHealth treatment or tablet cognitive gaming for skills | Wheelchair skills | Safety with skill performance is significant |
14 | [18] | Intervention for ‘risky drinking days’ for alcoholics | Alcohol dependence | Significant outpatient treatment adherence and reduction in risky drinking days by 11% |
15 | [83] | Intervention for self-monitoring of blood pressure | Patients with hypertension | Intervention had more rapid communication between patients and providers |
16 | [84] | Use of mindfulness as an intervention | Mental wellbeing for students | Significant decrease in depression, anxiety, and stress, and improved sleep quality |
17 | [85] | Postpartum contraceptive among women and couples | Postpartum contraceptive | Statistically significant (69.9% vs 57.4%) contraceptive use |
18 | [86] | E-mail and SMS for managing weight | Weight control | Reduced body weight, increased activity and vegetable intake, and decreased beverage intake (not statistically significant due to low engagement) |
19 | [87] | Intervention and counseling for self-management and lifestyle change | Diabetes | Statistical improvement in HbA1c level > = 7.1% |
20 | [88] | Effect of m-health on family planning using testing of consumer’s knowledge | Family planning | Access to m-health significant in testing consumer’s knowledge but cannot lead to behavior change |
21 | [89] | Study of recruitment and the trial follow-up of RCT participants to picture of the dropout predictors | Mental health | Engagement using SMS to integrate support for people with serious mental health problems |
22 | [90] | M-health platform on clinical outcomes, patient and provider satisfaction, and app usage | Diabetes | Significantly improved medication adherence and BG testing |
23 | [91] | Mhealth intervention (ADAPT) for medication adherence | Asthma patients | Positive effect on MA with uncontrolled asthma but no effect on adolescents with asthma |
24 | [92] | Online community intervention for adherence to inhaler | Asthma adherence | No statistically significant improvement in adherence |
25 | [93] | Impact of iMHere (interactive mHealth) on health outcomes | Spinal Cord Injury | Reduction in mood symptoms meeting the threshold for clinical significance |
26 | [21] | A mindfulness intervention for cancer patients and caregivers | QoL for cancer patients | Significant improvement in QoL among patients |
27 | [72] | A smart phone and wireless BP monitor as an intervention | Post stroke care | 32% vs 14% improvement of hypertension controlled in small test group of 28 patients |
28 | [94] | M-health intervention for parents in school-based weight management program | Weight management for children and adolescents | Increased in students’ health knowledge and positive psychological impact |
29 | [13] | Sub-analysis of RCT for Smart Care Services of Obese patients | Obesity | Anthropometric indices and laboratory test results improved |
30 | [95] | Intervention to Promote Retention and Adherence to Preexposure Prophylaxis | HIV risk | 16% more likely to attend study visits and 15% more likely to have tenofovir diphosphate levels consistent with < 4 doses/week |
31 | [96] | Study of two prototypes for dietary recoding utilization | Food intake | No control group, test was on usability of applications rather than on modification of health-related behaviors |
32 | [97] | Comparative efficacy of lifestyle application combined with a supervised rehabilitation program versus the lifestyle application alone | QoL and functional outcomes of breast cancer survivors | Reported clinical improvement on QoL was higher with combined app and rehab (57.5% vs 26.3%, P = 0.008) |
33 | [98] | MHealth intervention for self-management of type 2 diabetes | Diabetes | No statistical difference in HbA1c level but an increase in the self-management skills |
34 | [99] | Efficacy of an mHealth prevention program, TXT2BFiT | Weight management in young adults | Modest weight loss and more vegetables, fewer sugary soft drinks, and fewer energy-dense takeout meals |
35 | [100] | Mhealth intervention for self-monitoring and exercise behaviors as a follow up for diabetes prevention program | Diabetes | Prompts effective in the first half of the year but no changes in the second half of the year |
36 | [101] | Web-based mobile pain coping skills training (mPCST) protocol | HCT patients | Significant improvements in pre-treatment and post-treatment pain, self-efficacy, and in the two-minute walk test. No significant changes in pain severity |
37 | [102] | Mhealth intervention for physical activity promotion | Physical activity | Significant change in daily steps by 2534 |
38 | [103] | Mhealth intervention for improving medication adherence and BP control | Renal Transplant recipient | Proof-of-Concept RCT |
39 | [104] | Feasibility of an intervention for blood pressure (BP) self-monitoring | Hypertension | Significant blood pressure reduction over 3-week period |
40 | [105] | Evaluation of a multi-step enrollment process for an RCT to promote regular HIV testing | HIV risk | The need for pre-determined enrollment decision rules based on available resources |
41 | [6] | Effectiveness of 2 interventions separately and combined to promote infant safe sleep practices | Infant sleep | Nursing quality improvement was not significant for all outcomes |
42 | [106] | Mhealth intervention to improve physical activity in college students | Physical activity | Physical activity was higher in step-count but no significant effects for BMI and body fat |
43 | [14] | MINISTOP intervention to improve fat mass index (FMI) using dietary and physical activities | Weight management | No statistically significant difference |
44 | [15] | Assessment of the weight loss achieved by using SmartCare service based on adherence and participation | Obesity Management | Statistically significant improvement to BMI and weight but lipid profile not statistically different between two groups |
45 | [107] | Moderation and Mediation to study improved eating behaviors of young adults | Eating behavior | Weight changes significant and increased vegetable and decreased sweetened beverage intake accounted for indirect weight change effects |
46 | [108] | CHI using mobile devices for risk patients for BP | Pre-BP in rural India | Increased treatment rate, but no outcome on BP |
47 | [109] | Comparing Exercise vs m-health + exercise group | Exercise | Negative outcome (Intervention group did worse in 12 weeks but the same in 52 weeks) |
48 | [12] | Self-management for physical activity | Neurological disability | Commercial MHA improve physical activity |
49 | [110] | MHI and health counseling (3 groups) | Diabetes | No statistical difference in HbA1c in 3 groups at 4 months |
50 | [111] | Comparison of e-health intervention with a mobile health intervention | Physical Activity | eHealth outperformed mHealth for usability and user appreciation, but limited impact on user health and health behaviors |
51 | [112] | Effectiveness of low-cost m-health intervention (text, reminder, messaging) | Noncommunicable diseases in rural and refugee environment | The intervention reduced symptoms in rural but less in refugee camps |
52 | [113] | Self-monitoring using Pocket PATH for patients | Lung Transplant | Promising and proposed for the long-term |
53 | [73] | Mobile health intervention for wellness | Food, weight, BP, & physical activity | No significant difference with small impact of MHI |
54 | [114] | Use of BodiMojo for healthy lifestyle | Diabetes in Young people | No differences in self-efficacy or A1c (82% believed it helped them achieve health goals) |
55 | [16] | Intervention (Calls, TM, apps, resources) for Wellness | Weight management in young people | Modest weight loss, improved dietary behavior and no change in physical activity |
56 | [115] | Use of a self-management app | GOUT | No difference in self-care |
57 | [116] | Effectiveness of Teen Pocket PATH app for patients and their caregivers | Solid organ transplant | Promise in promoting medication adherence among adolescents and caregivers |
58 | [117] | Effectiveness of “Home but not alone” app | Post-partum app | Improved self-efficacy of parents, social support and parental satisfaction |
59 | [118] | Effectiveness of m-health diet and activity intervention + coaching | Diet and health activity risk | The intervention reduces risk |
60 | [119] | Plan ABC app as an educational intervention | Birth control methods for women | No difference in app and regular health counseling group |
61 | [5] | Clinical efficacy of AsthmaCare app | Asthma in children | 72% reduction in urgent care trips |
62 | [120] | M-health intervention for improving the nutritional value in lunchboxes | Nutritional food in the lunchbox | Significant increase in recommended foods in lunchboxes |
63 | [121] | Effectiveness of a therapeutic intervention | Suicide prevention | Reduces distress and depression, but no reduction in suicide ideation or impulsivity |
64 | [122] | Low cost INTV with or without counseling | Diabetes Management | Only 1 of 8 outcomes (skills and technique acquisition) improved |
65 | [22] | Effectiveness of genetic counseling app | Ovarian Cancer | No increased uptake of genetic counseling but increased knowledge, self-efficacy, and communications with family |
66 | [123] | Effectiveness of an app for managing Diabetes | Diabetes | The frequency of hospitalization & rehospitalization is reduced |
67 | [20] | The role of health diets, physical activity, and GWG guidelines | Gestational weight gain | Same food consumption, but less decrease in physical activities, and less weight gain |
68 | [19] | Sexual minority males (Guy2Guy: Text messaging intervention) | Sexual minority males | Improves HIV testing, increased sexual activity and less risky sex |
69 | [124] | Educational intervention for parents to create smoke free home for newborns | Smoking cessation | Increases smoking cessation |
70 | [125] | Feasibility of WeTest as m-health intervention | HIV Testing | Higher rate of testing and condom use with partners and non-partners |
Appendix B. Taxonomy – mobile health interventions evaluated with an RCT
Iteration 1 | Iteration 2 | Iteration 3 | ||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Article | Duration (Months) | Stages | Arms | Blinding | Participant Design | Number of Participants | Intervention Target | Post RCT Evaluation | Health Condition | |||||||||||||||||||
< 3 | 3—6 | > 6 | 1 | 2 | ≥ 3 | 1 | 2 | 3 | Single Blind | Double Blind | Unblinded or Unspecified | Parallel | Crossover | Not Specified | 0 to 10 | 11 to 25 | 26 to 50 | 50 + | Patient | Professional | Both | None | < 3 Months | 3—6 Months | > 6 Months | Existing Condition | Preventative | |
[17] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[74] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[75] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[11] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[76] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[77] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[23] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[78] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[79] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[80] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[8] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[82] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[18] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[83] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[84] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[85] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[86] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[87] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[88] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[89] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[90] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[91] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[92] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[93] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[21] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[72] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[13] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[94] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[95] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[96] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[97] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[98] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[100] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[102] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[103] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[104] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[105] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[6] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[106] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[81] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[14] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[15] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[99] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[107] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[108] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[109] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[12] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[111] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[113] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[73] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[112] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[114] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[16] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[115] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[116] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[117] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[101] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[118] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[119] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[5] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[123] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[120] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[121] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[110] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[122] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[22] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[20] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[19] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[124] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
[125] | x | x | x | x | x | x | x | x | x | |||||||||||||||||||
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Yang, A., Singh, N. & Varshney, U. Mobile Health Interventions and RCTs: Structured Taxonomy and Research Framework. J Med Syst 46, 66 (2022). https://doi.org/10.1007/s10916-022-01856-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10916-022-01856-6