Abstract
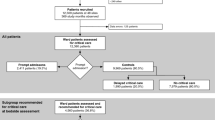
Supply–demand mismatch of ward resources (“ward capacity strain”) alters care and outcomes. Narrow strain definitions and heterogeneous populations limit strain literature. Evaluate the predictive utility of a large set of candidate strain variables for in-hospital mortality and discharge destination among acute respiratory failure (ARF) survivors. In a retrospective cohort of ARF survivors transferred from intensive care units (ICUs) to wards in five hospitals from 4/2017–12/2019, we applied 11 machine learning (ML) models to identify ward strain measures during the first 24 hours after transfer most predictive of outcomes. Measures spanned patient volume (census, admissions, discharges), staff workload (medications administered, off-ward transports, transfusions, isolation precautions, patients per respiratory therapist and nurse), and average patient acuity (Laboratory Acute Physiology Score version 2, ICU transfers) domains. The cohort included 5,052 visits in 43 wards. Median age was 65 years (IQR 56–73); 2,865 (57%) were male; and 2,865 (57%) were white. 770 (15%) patients died in the hospital or had hospice discharges, and 2,628 (61%) were discharged home and 964 (23%) to skilled nursing facilities (SNFs). Ward admissions, isolation precautions, and hospital admissions most consistently predicted in-hospital mortality across ML models. Patients per nurse most consistently predicted discharge to home and SNF, and medications administered predicted SNF discharge. In this hypothesis-generating analysis of candidate ward strain variables’ prediction of outcomes among ARF survivors, several variables emerged as consistently predictive of key outcomes across ML models. These findings suggest targets for future inferential studies to elucidate mechanisms of ward strain’s adverse effects.


Similar content being viewed by others
Data Availability
The authors will consider sharing de-identified data made by reasonable request to the corresponding author.
References
Halpern SD. ICU capacity strain and the quality and allocation of critical care. Curr Opin Crit Care. Dec 2011;17(6):648-57. https://doi.org/10.1097/MCC.0b013e32834c7a53
Kerlin MP, Harhay MO, Vranas KC, Cooney E, Ratcliffe SJ, Halpern SD. Objective factors associated with physicians' and nurses' perceptions of intensive care unit capacity strain. Ann Am Thorac Soc. Feb 2014;11(2):167-72. https://doi.org/10.1513/AnnalsATS.201306-141OC
Bagshaw SM, Opgenorth D, Potestio M, et al. Healthcare Provider Perceptions of Causes and Consequences of ICU Capacity Strain in a Large Publicly Funded Integrated Health Region: A Qualitative Study. Crit Care Med. Apr 2017;45(4):e347-e356. https://doi.org/10.1097/CCM.0000000000002093
Carr BG, Addyson DK, Kahn JM. Variation in critical care beds per capita in the United States: implications for pandemic and disaster planning. JAMA. Apr 2010;303(14):1371-2. https://doi.org/10.1001/jama.2010.394
Wagner J, Gabler NB, Ratcliffe SJ, Brown SE, Strom BL, Halpern SD. Outcomes among patients discharged from busy intensive care units. Ann Intern Med. Oct 1 2013;159(7):447-55. https://doi.org/10.7326/0003-4819-159-7-201310010-00004
Gabler NB, Ratcliffe SJ, Wagner J, et al. Mortality among patients admitted to strained intensive care units. Am J Respir Crit Care Med. Oct 1 2013;188(7):800-6. https://doi.org/10.1164/rccm.201304-0622OC
Brown SE, Rey MM, Pardo D, et al. The allocation of intensivists' rounding time under conditions of intensive care unit capacity strain. Am J Respir Crit Care Med. Oct 1 2014;190(7):831-4. https://doi.org/10.1164/rccm.201406-1127LE
Weissman GE, Gabler NB, Brown SE, Halpern SD. Intensive care unit capacity strain and adherence to prophylaxis guidelines. J Crit Care. Dec 2015;30(6):1303-9. https://doi.org/10.1016/j.jcrc.2015.08.015
Hua M, Halpern SD, Gabler NB, Wunsch H. Effect of ICU strain on timing of limitations in life-sustaining therapy and on death. Intensive Care Med. Jun 2016;42(6):987-94. https://doi.org/10.1007/s00134-016-4240-8
Hefter Y, Madahar P, Eisen LA, Gong MN. A Time-Motion Study of ICU Workflow and the Impact of Strain. Crit Care Med. Aug 2016;44(8):1482-9. https://doi.org/10.1097/CCM.0000000000001719
Rose L, Scales DC, Atzema C, et al. Emergency Department Length of Stay for Critical Care Admissions. A Population-based Study. Ann Am Thorac Soc. Aug 2016;13(8):1324–32. https://doi.org/10.1513/AnnalsATS.201511-773OC
Pines JM, Localio AR, Hollander JE, et al. The impact of emergency department crowding measures on time to antibiotics for patients with community-acquired pneumonia. Ann Emerg Med. Nov 2007;50(5):510-6. https://doi.org/10.1016/j.annemergmed.2007.07.021
Schull MJ, Vermeulen M, Slaughter G, Morrison L, Daly P. Emergency department crowding and thrombolysis delays in acute myocardial infarction. Ann Emerg Med. Dec 2004;44(6):577-85. https://doi.org/10.1016/S0196064404005232
Weiss SJ, Derlet R, Arndahl J, et al. Estimating the degree of emergency department overcrowding in academic medical centers: results of the National ED Overcrowding Study (NEDOCS). Acad Emerg Med. Jan 2004;11(1):38–50.
Hwang U, McCarthy ML, Aronsky D, et al. Measures of crowding in the emergency department: a systematic review. Acad Emerg Med. May 2011;18(5):527-38. https://doi.org/10.1111/j.1553-2712.2011.01054.x
Morley C, Unwin M, Peterson GM, Stankovich J, Kinsman L. Emergency department crowding: A systematic review of causes, consequences and solutions. PLoS One. 2018;13(8):e0203316. https://doi.org/10.1371/journal.pone.0203316
Michtalik HJ, Pronovost PJ, Marsteller JA, Spetz J, Brotman DJ. Developing a model for attending physician workload and outcomes. JAMA Intern Med. Jun 10 2013;173(11):1026-8. https://doi.org/10.1001/jamainternmed.2013.405
Needleman J, Buerhaus P, Pankratz VS, Leibson CL, Stevens SR, Harris M. Nurse staffing and inpatient hospital mortality. N Engl J Med. Mar 17 2011;364(11):1037-45. https://doi.org/10.1056/NEJMsa1001025
Volchenboum SL, Mayampurath A, Goksu-Gursoy G, Edelson DP, Howell MD, Churpek MM. Association Between In-Hospital Critical Illness Events and Outcomes in Patients on the Same Ward. JAMA. Dec 27 2016;316(24):2674-2675. https://doi.org/10.1001/jama.2016.15505
Kohn R, Harhay MO, Weissman GE, et al. Ward Capacity Strain: A Novel Predictor of Delays in Intensive Care Unit Survivor Throughput. Ann Am Thorac Soc. 2019;16(3):387-390.
Kohn R, Harhay MO, Bayes B, et al. Ward Capacity Strain: A Novel Predictor of 30-Day Hospital Readmissions. J Gen Intern Med. 2018;33(11):1851-1853.
Kohn R, Harhay MO, Bayes B, et al. Influence of bedspacing on outcomes of hospitalised medicine service patients: a retrospective cohort study. BMJ Qual Saf. Apr 2020. https://doi.org/10.1136/bmjqs-2019-010675
Healthcare Cost and Utilization Project (HCUP) Statistical Briefs. 2006.
Fonarow GC, Abraham WT, Albert NM, et al. Day of admission and clinical outcomes for patients hospitalized for heart failure: findings from the Organized Program to Initiate Lifesaving Treatment in Hospitalized Patients With Heart Failure (OPTIMIZE-HF). Circ Heart Fail. May 2008;1(1):50-7. https://doi.org/10.1161/CIRCHEARTFAILURE.107.748376
Earnest A, Chen MI, Seow E. Exploring if day and time of admission is associated with average length of stay among inpatients from a tertiary hospital in Singapore: an analytic study based on routine admission data. BMC Health Serv Res. Jan 22 2006;6:6. https://doi.org/10.1186/1472-6963-6-6
Tran K, Bell C, Stall N, et al. The Effect of Hospital Isolation Precautions on Patient Outcomes and Cost of Care: A Multi-Site, Retrospective, Propensity Score-Matched Cohort Study. J Gen Intern Med. Mar 2017;32(3):262-268. https://doi.org/10.1007/s11606-016-3862-4
Stelfox HT, Bates DW, Redelmeier DA. Safety of patients isolated for infection control. JAMA. Oct 08 2003;290(14):1899-905. https://doi.org/10.1001/jama.290.14.1899
Colice GL, Carnathan B, Sung J, Paramore LC. A respiratory therapist-directed protocol for managing inpatients with asthma and COPD incorporating a long-acting bronchodilator. J Asthma. Feb 2005;42(1):29-34. https://doi.org/10.1081/jas-200044765
Harbrecht BG, Delgado E, Tuttle RP, Cohen-Melamed MH, Saul MI, Valenta CA. Improved outcomes with routine respiratory therapist evaluation of non-intensive-care-unit surgery patients. Respir Care. Jul 2009;54(7):861-7. https://doi.org/10.4187/002013209793800457
Kallam A, Meyerink K, Modrykamien AM. Physician-ordered aerosol therapy versus respiratory therapist-driven aerosol protocol: the effect on resource utilization. Respir Care. Mar 2013;58(3):431-7. https://doi.org/10.4187/respcare.01907
Kollef MH, Shapiro SD, Clinkscale D, et al. The effect of respiratory therapist-initiated treatment protocols on patient outcomes and resource utilization. Chest. Feb 2000;117(2):467-75. https://doi.org/10.1378/chest.117.2.467
Stoller JK, Mascha EJ, Kester L, Haney D. Randomized controlled trial of physician-directed versus respiratory therapy consult service-directed respiratory care to adult non-ICU inpatients. Am J Respir Crit Care Med. Oct 1998;158(4):1068-75. https://doi.org/10.1164/ajrccm.158.4.9709076
Aiken LH, Clarke SP, Sloane DM, Sochalski J, Silber JH. Hospital nurse staffing and patient mortality, nurse burnout, and job dissatisfaction. JAMA. Oct 23–30 2002;288(16):1987–93.
Amaravadi RK, Dimick JB, Pronovost PJ, Lipsett PA. ICU nurse-to-patient ratio is associated with complications and resource use after esophagectomy. Intensive Care Med. Dec 2000;26(12):1857-62.
Checkley W, Martin GS, Brown SM, et al. Structure, Process, and Annual ICU Mortality Across 69 Centers: United States Critical Illness and Injury Trials Group Critical Illness Outcomes Study. Critical Care Medicine. Feb 2014;42(2):344-356. https://doi.org/10.1097/CCM.0b013e3182a275d7
Dang D, Johantgen ME, Pronovost PJ, Jenckes MW, Bass EB. Postoperative complications: does intensive care unit staff nursing make a difference? Heart Lung. May-Jun 2002;31(3):219-28.
Dimick JB, Swoboda SM, Pronovost PJ, Lipsett PA. Effect of nurse-to-patient ratio in the intensive care unit on pulmonary complications and resource use after hepatectomy. Am J Crit Care. Nov 2001;10(6):376-82.
Neuraz A, Guerin C, Payet C, et al. Patient Mortality Is Associated With Staff Resources and Workload in the ICU: A Multicenter Observational Study. Crit Care Med. Aug 2015;43(8):1587-94. https://doi.org/10.1097/CCM.0000000000001015
Sakr Y, Moreira CL, Rhodes A, et al. The Impact of Hospital and ICU Organizational Factors on Outcome in Critically III Patients: Results From the Extended Prevalence of Infection in Intensive Care Study. Critical Care Medicine. Mar 2015;43(3):519-526. https://doi.org/10.1097/Ccm.0000000000000754
Escobar GJ, Greene JD, Scheirer P, Gardner MN, Draper D, Kipnis P. Risk-adjusting hospital inpatient mortality using automated inpatient, outpatient, and laboratory databases. Med Care. Mar 2008;46(3):232-9. https://doi.org/10.1097/MLR.0b013e3181589bb6
Liu V, Kipnis P, Gould MK, Escobar GJ. Length of stay predictions: improvements through the use of automated laboratory and comorbidity variables. Med Care. Aug 2010;48(8):739-44. https://doi.org/10.1097/MLR.0b013e3181e359f3
Anesi GL, Chowdhury M, Small DS, et al. Association of a Novel Index of Hospital Capacity Strain with Admission to Intensive Care Units. Ann Am Thorac Soc. 11 2020;17(11):1440–1447. https://doi.org/10.1513/AnnalsATS.202003-228OC
Wong J, Taljaard M, Forster AJ, Escobar GJ, van Walraven C. Derivation and validation of a model to predict daily risk of death in hospital. Med Care. Aug 2011;49(8):734-43. https://doi.org/10.1097/MLR.0b013e318215d266
Kohn R, Weissman GE, Wang W, et al. Prediction of In-hospital Mortality Among Intensive Care Unit Patients Using Modified Daily Laboratory-based Acute Physiology Score, Version 2. Med Care. Jun 12 2023;https://doi.org/10.1097/MLR.0000000000001878
Reineck LA, Pike F, Le TQ, Cicero BD, Iwashyna TJ, Kahn JM. Hospital factors associated with discharge bias in ICU performance measurement. Crit Care Med. May 2014;42(5):1055-64. https://doi.org/10.1097/CCM.0000000000000132
Warraich HJ, Xu H, DeVore AD, et al. Trends in Hospice Discharge and Relative Outcomes Among Medicare Patients in the Get With The Guidelines-Heart Failure Registry. JAMA Cardiol. 10 01 2018;3(10):917–926. https://doi.org/10.1001/jamacardio.2018.2678
Ma J, Chi S, Buettner B, et al. Early Palliative Care Consultation in the Medical ICU: A Cluster Randomized Crossover Trial. Crit Care Med. 12 2019;47(12):1707–1715. https://doi.org/10.1097/CCM.0000000000004016
Obermeyer Z, Makar M, Abujaber S, Dominici F, Block S, Cutler DM. Association between the Medicare hospice benefit and health care utilization and costs for patients with poor-prognosis cancer. JAMA. Nov 12 2014;312(18):1888-96. https://doi.org/10.1001/jama.2014.14950
Teno JM, Gozalo PL, Bynum JP, et al. Change in end-of-life care for Medicare beneficiaries: site of death, place of care, and health care transitions in 2000, 2005, and 2009. JAMA. Feb 06 2013;309(5):470-7. https://doi.org/10.1001/jama.2012.207624
Olmsted CL, Johnson AM, Kaboli P, Cullen J, Vaughan-Sarrazin MS. Use of palliative care and hospice among surgical and medical specialties in the Veterans Health Administration. JAMA Surg. Nov 2014;149(11):1169-75. https://doi.org/10.1001/jamasurg.2014.2101
Anesi GL, Liu VX, Gabler NB, et al. Associations of ICU Capacity Strain with Disposition and Outcomes of Patients with Sepsis Presenting to the Emergency Department. Ann Am Thorac Soc. Aug 2018;https://doi.org/10.1513/AnnalsATS.201804-241OC
Elixhauser A, Steiner C, Harris DR, Coffey RM. Comorbidity measures for use with administrative data. Med Care. Jan 1998;36(1):8-27.
Quan H, Parsons GA, Ghali WA. Validity of information on comorbidity derived rom ICD-9-CCM administrative data. Med Care. Aug 2002;40(8):675-85. https://doi.org/10.1097/01.MLR.0000020927.46398.5D
Quan H, Sundararajan V, Halfon P, et al. Coding algorithms for defining comorbidities in ICD-9-CM and ICD-10 administrative data. Med Care. Nov 2005;43(11):1130-9.
Escobar GJ, Gardner MN, Greene JD, Draper D, Kipnis P. Risk-adjusting hospital mortality using a comprehensive electronic record in an integrated health care delivery system. Med Care. May 2013;51(5):446-53. https://doi.org/10.1097/MLR.0b013e3182881c8e
Hu W, Chan CW, Zubizarreta JR, Escobar GJ. Incorporating Longitudinal Comorbidity and Acute Physiology Data in Template Matching for Assessing Hospital Quality: An Exploratory Study in an Integrated Health Care Delivery System. Med Care. May 2018;56(5):448-454. https://doi.org/10.1097/MLR.0000000000000891
Nicholls HL, John CR, Watson DS, Munroe PB, Barnes MR, Cabrera CP. Reaching the End-Game for GWAS: Machine Learning Approaches for the Prioritization of Complex Disease Loci. Front Genet. 2020;11:350. https://doi.org/10.3389/fgene.2020.00350
Kohavi R. A study of cross-validation and bootstrap for accuracy estimation and model selection. Proceedings of the 14th international joint conference on artificial intelligence. vol 2. 1995.
Efron B, Hastie T, Johnstone I, Tibshirani R. Least angle regression. Ann Statist. 2004;32:407-99.
Hastie T, Tibshirani R, Wainwright M. Statistical Learning with Sparsity: The Lasso and Generalizations. CRC Press; 2015.
Friedman J, Hastie T, Tibshirani R. Regularization Paths for Generalized Linear Models via Coordinate Descent. J Stat Softw. 2010;33(1):1-22.
Torio CM, Moore BJ. National Inpatient Hospital Costs: The Most Expensive Conditions by Payer, 2013. HCUP Statistical Brief #204. Rockville, MD: Agency for Healthcare Research and Quality; 2016.
Orlenko A, Kofink D, Lyytikäinen LP, et al. Model selection for metabolomics: predicting diagnosis of coronary artery disease using automated machine learning. Bioinformatics. 03 01 2020;36(6):1772–1778. https://doi.org/10.1093/bioinformatics/btz796
Olson RS, Moore JH. TPOT: A Tree-based Pipeline Optimization Tool for Automating Machine Learning. JMLR: Workshop and Conference Proceedings. 2016;64:66–74.
Olson RS, Bartley N, Urbanowicz RJ, Moore JH. Evaluation of a tree-based pipeline optimization tool for automating data science. GECCO '16: Proceedings of the Genetic and Evolutionary Computation Conference 2016. 2016:485–492.
Manduchi E, Fu W, Romano JD, Ruberto S, Moore JH. Embedding covariate adjustments in tree-based automated machine learning for biomedical big data analyses. BMC Bioinformatics. 2020;21(1):430. https://doi.org/10.1186/s12859-020-03755-4
Le TT, Fu W, Moore JH. Scaling tree-based automated machine learning to biomedical big data with a feature set selector. Bioinformatics. 01 01 2020;36(1):250–256. https://doi.org/10.1093/bioinformatics/btz470
Tree-based models. C3.ai. Accessed July 5, 2023. https://c3.ai/glossary/data-science/tree-based-models/#:~:text=Tree%2Dbased%20models%20are%20a,both%20classification%20and%20regression%20models.
Garland A, Olafson K, Ramsey CD, Yogendran M, Fransoo R. A population-based observational study of intensive care unit-related outcomes. With emphasis on post-hospital outcomes. Ann Am Thorac Soc. 2015;12(2):202–8. https://doi.org/10.1513/AnnalsATS.201405-201CME
Funding
Dr. Kohn was supported by NIH/NHLBI K23 HL146894. Dr. Harhay was supported by NIH/NHLBI R00 HL141678.
Author information
Authors and Affiliations
Contributions
RK, MOH, SDH, and MPK contributed to the conception and design of the study. RK, MOH, GEW, RU, WW, SS, and BB contributed to the acquisition, analysis, or interpretation of data. RK, MOH, GEW, RU, WW, GLA, SS, BB, SRG, SDH, and MPK contributed to the drafting or revising of the manuscript.
Corresponding author
Ethics declarations
Ethical Approval
The University of Pennsylvania Institutional Review Board deemed this protocol exempt (protocol #833060).
Conflict of Interest
Dr. Kohn received an honorarium from the University of Nebraska for presenting her research portfolio at Pulmonary and Critical Care Medicine Grand Rounds. No authors have additional financial disclosures or conflicts of interest related to this work.
Informed Consent
Not applicable.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Kohn, R., Harhay, M.O., Weissman, G.E. et al. A Data-Driven Analysis of Ward Capacity Strain Metrics That Predict Clinical Outcomes Among Survivors of Acute Respiratory Failure. J Med Syst 47, 83 (2023). https://doi.org/10.1007/s10916-023-01978-5
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s10916-023-01978-5