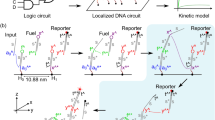
Abstract
Implementing DNA computing circuits using components tethered to a surface offers several advantages over using components that freely diffuse in bulk solution. However, automated computational modeling of tethered circuits is far more challenging than for solution-phase circuits, because molecular geometry must be taken into account when deciding whether two tethered species may interact. Here, we tackle this issue by translating a tethered molecular circuit into a constraint problem that encodes the possible physical configurations of the components, using a simple biophysical model. We use a satisfaction modulo theories solver to determine whether the constraint problem associated with a given structure is satisfiable, which corresponds to whether that structure is physically realizable given the constraints imposed by the tether geometry. We apply this technique to example structures from the literature, and discuss how this approach could be integrated with a reaction enumerator to enable fully automated analysis of tethered molecular computing systems. This paper is a significantly revised and extended version of a conference publication: Lakin and Phillips (in Brijder and Qian (eds) Proceedings of the 23rd International Conference on DNA Computing and Molecular Programming. Lecture Notes in Computer Science, vol 10467, pp 1–16, 2017).




Similar content being viewed by others
Notes
See the Supporting Information (available from the first author’s web page) for full details of the examples and corresponding constraints.
References
Badelt S, Shin SW, Johnson RF, Dong Q, Thachuk C, Winfree E (2017) A general-purpose CRN-to-DSD compiler with formal verification, optimization, and simulation capabilities. In: R Brijder, L Qian (eds) Proceedings of DNA23. LNCS, vol 10467, pp 232–248. https://doi.org/10.1007/978-3-319-66799-7_15
Bui H, Miao V, Garg S, Mokhtar R, Song T, Reif J (2017) Design and analysis of localized DNA hybridization chain reactions. Small 13(12):1602983. https://doi.org/10.1002/smll.201602983
Chatterjee G, Dalchau N, Muscat RA, Phillips A, Seelig G (2017) A spatially localized architecture for fast and modular DNA computing. Nat Nanotechnol 12:920–927. https://doi.org/10.1038/nnano.2017.127
Chen YJ, Dalchau N, Srinivas N, Phillips A, Cardelli L, Soloveichik D, Seelig G (2013) Programmable chemical controllers made from DNA. Nat Nanotechnol 8:755–762. https://doi.org/10.1038/nnano.2013.189
Cook M, Soloveichik D, Winfree E, Bruck J (2009) Programmability of chemical reaction networks. In: Algorithmic Bioprocesses. Springer, pp 543–584
Dalchau N, Chandran H, Gopalkrishnan N, Phillips A, Reif J (2015) Probabilistic analysis of localized DNA hybridization circuits. ACS Synth Biol 4(8):898–913. https://doi.org/10.1021/acssynbio.5b00044
de Moura L, Bjørner N (2008) Z3: an efficient SMT solver. In: Proceedings of TACAS 2008. LNCS, vol 4963. Springer, pp 337–340. https://doi.org/10.1007/978-3-540-78800-3_24
Dirks RM, Pierce NA (2004) An algorithm for computing nucleic acid base-pairing probabilities including pseudoknots. J Comput Chem 25:1295–1304. https://doi.org/10.1002/jcc.20057
Doye JPK, Ouldridge TE, Louis AA, Romano F, Šulc P, Matek C, Snodin BEK, Rovigatti L, Schreck JS, Harrison RM, Smith WPJ (2013) Coarse-graining DNA for simulations of DNA nanotechnology. Phys Chem Chem Phys 15:20395–20414
Elbaz J, Cecconello A, Fan Z, Govorov AO, Willner I (2013) Powering the programmed nanostructure and function of gold nanoparticles with catenated DNA machines. Nat Commun 4:2000. https://doi.org/10.1038/ncomms3000
Genot AJ, Zhang DY, Bath J, Turberfield AJ (2011) Remote toehold: a mechanism for flexible control of DNA hybridization kinetics. J Am Chem Soc 133:2177–2182
Grun C, Werfel J, Zhang DY, Yin P (2015) DyNAMiC workbench: an integrated development environment for dynamic DNA nanotechnology. JRS Interface 12:20150580. https://doi.org/10.1098/rsif.2015.0580
Jovanović D, de Moura L (2012) Solving non-linear arithmetic. In: Proceedings of IJCAR 2012. LNCS, vol 7364. Springer, pp 339–354. https://doi.org/10.1007/978-3-642-31365-3_27
Lakin MR, Phillips A (2017) Automated, constraint-based analysis of tethered DNA nanostructures. In Proceedings of DNA23. LNCS, vol 10467, pp 1–16
Lakin MR, Petersen R, Gray KE, Phillips A (2014) Abstract modelling of tethered DNA circuits. In: Proceedings of DNA20. LNCS, vol 8727. Springer, pp 132–147. https://doi.org/10.1007/978-3-319-11295-4_9
Lakin MR, Youssef S, Polo F, Emmott S, Phillips A (2011) Visual DSD: a design and analysis tool for DNA strand displacement systems. Bioinformatics 27(22):3211–3213
Lakin MR, Parker D, Cardelli L, Kwiatkowska M, Phillips A (2012) Design and analysis of DNA strand displacement devices using probabilistic model checking. JRS Interface 9(72):1470–1485
Lakin MR, Youssef S, Cardelli L, Phillips A (2012) Abstractions for DNA circuit design. JRS Interface 9(68):470–486
List J, Falgenhauer E, Kopperger E, Pardatcher G, Simmel FC (2016) Long-range movement of large mechanically interlocked DNA nanostructures. Nat Commun 7:12414. https://doi.org/10.1038/ncomms12414
Muscat RA, Strauss K, Ceze L, Seelig G (2013) DNA-based molecular architecture with spatially localized components. In: Proceedings of ISCA 13
Ouldridge TE, Louis AA, Doye JPK (2010) DNA nanotweezers studied with a coarse-grained model of DNA. Phys Rev Lett 104:178101–178105
Ouldridge TE, Hoare RL, Louis AA, Doye JPK, Bath J, Turberfield AJ (2013) Optimizing DNA nanotechnology through coarse-grained modeling: a two-footed DNA walker. ACS Nano 7:2479–2490
Petersen RL, Lakin MR, Phillips A (2016) A strand graph semantics for DNA-based computation. Theor Comput Sci 632:43–73. https://doi.org/10.1016/j.tcs.2015.07.041
Qian L, Winfree E (2011) Scaling up digital circuit computation with DNA strand displacement cascades. Science 332:1196–1201. https://doi.org/10.1126/science.1200520
Qian L, Winfree E, Bruck J (2011) Neural network computation with DNA strand displacement cascades. Nature 475:368–372. https://doi.org/10.1038/nature10262
Rothemund PWK (2006) Folding DNA to create nanoscale shapes and patterns. Nature 440:297–302
Snodin BE, Randisi F, Mosayebi M, Šulc P, Schreck JS, Romano F, Ouldridge TE, Tsukanov R, Nir E, Louis AA, Doye JP (2015) Introducing improved structural properties and salt dependence into a coarse-grained model of DNA. J Chem Phys 142:234901
Soloveichik D, Seelig G, Winfree E (2010) DNA as a universal substrate for chemical kinetics. PNAS 107(12):5393–5398
Šulc P, Romano F, Ouldridge TE, Rovigatti L, Doye JPK, Louis AA (2012) Sequence-dependent thermodynamics of a coarse-grained DNA model. J Chem Phys 137:135101–135114
Thubagere AJ, Li W, Johnson RF, Chen Z, Doroudi S, Lee YL, Izatt G, Wittman S, Srinivas N, Woods D, Winfree E, Qian L (2017) A cargo-sorting DNA robot. Science 357(6356):1112. https://doi.org/10.1126/science.aan6558
Tikhomirov G, Petersen P, Qian L (2017) Programmable disorder in random DNA tilings. Nat Nanotechnol 12:251–259. https://doi.org/10.1038/nnano.2016.256
Walsh AS, Yin H, Erben CM, Wood MJA, Turberfield AJ (2011) DNA cage delivery to mammalian cells. ACS Nano 5(7):5427–5432
Woo S, Rothemund PWK (2011) Programmable molecular recognition based on the geometry of DNA nanostructures. Nat Chem 3:620–627. https://doi.org/10.1038/nchem.1070
Zhang DY, Seelig G (2011) Dynamic DNA nanotechnology using strand-displacement reactions. Nat Chem 3(2):103–113. https://doi.org/10.1038/nchem.957
Acknowledgements
The authors thank Neil Dalchau and Rasmus Petersen for productive discussions.
Author information
Authors and Affiliations
Corresponding author
Additional information
This material is based upon work supported by the National Science Foundation under Grants 1525553, 1518861, and 1318833.
Rights and permissions
About this article
Cite this article
Lakin, M.R., Phillips, A. Automated analysis of tethered DNA nanostructures using constraint solving. Nat Comput 17, 709–722 (2018). https://doi.org/10.1007/s11047-018-9693-y
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11047-018-9693-y