Abstract
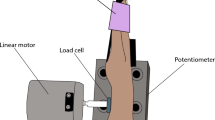
Studies have reported the benefits of sensory noise in motor performance, but it is not clear if this phenomenon is influenced by muscle contraction intensity. Additionally, most of the studies investigated the role of a stochastic noise on the improvement of motor control and there is no evidence that a sinusoidal vibrotactile stimulation could also enhance motor performance. Eleven participants performed a sensorimotor task while sinusoidal vibrations were applied to the finger skin. The effects of an optimal vibration (OV) on force steadiness were evaluated in different contraction intensities. We assessed the standard deviation (SD) and coefficient of variation (CoV) of force signals. OV significantly decreased force SD irrespective of contraction intensity, but the decrease in force CoV was significantly higher for low-intensity contraction. To the best of our knowledge, our findings are the first evidence that sinusoidal vibrotactile stimulation can enhance force steadiness in a motor task. Also, the significant improvement caused by OV during low-intensity contractions is probably due to the higher sensitivity of the motor system to the synaptic noise. These results add to the current knowledge on the effects of vibrotactile stimulation in motor control and have potential implications for the development of wearable haptic devices.

In this work the effects of a sinusoidal vibrotactile stimulation on force steadiness was investigated. Index finger sensorimotor tasks were performed in three levels of isometric contraction of the FDI muscle: 5, 10 and 15 %MVC. An optimal level of vibration significantly improved force steadiness, but the decrease in force CoV caused by vibration was more pronounced in contractions at 5 %MVC.






Similar content being viewed by others
References
Proske U, Gandevia SC (2012) The proprioceptive senses: their roles in signaling body shape, body position and movement, and muscle force. Physiol Rev 92:1651–1697. https://doi.org/10.1152/physrev.00048.2011
Prochazka A, Ellaway P (2012) Sensory systems in the control of movement. Compr Physiol 2:2615–2627. https://doi.org/10.1002/cphy.c100086
Samoudi G, Jivegard M, Mulavara AP, Bergquist F (2015) Effects of stochastic vestibular galvanic stimulation and LDOPA on balance and motor symptoms in patients with Parkinson’s disease. Brain Stimul 8:474–480. https://doi.org/10.1016/j.brs.2014.11.019
Mulavara AP, Fiedler MJ, Kofman IS, Wood SJ, Serrador JM, Peters B, Cohen HS, Reschke MF, Bloomberg JJ (2011) Improving balance function using vestibular stochastic resonance: optimizing stimulus characteristics. Exp Brain Res 210:303–312. https://doi.org/10.1007/s00221-011-2633-z
Priplata A, Niemi J, Salen M, Harry J, Lipsitz LA, Collins JJ (2002) Noise-enhanced human balance control. Phys Rev Lett 89:238101. https://doi.org/10.1103/PhysRevLett.89.238101
Priplata AA, Niemi JB, Harry JD, Lipsitz LA, Collins JJ (2003) Vibrating insoles and balance control in elderly people. Lancet 362:1123–1124. https://doi.org/10.1016/S0140-6736(03)14470-4
Priplata AA, Patritti BL, Niemi JB, Hughes R, Gravelle DC, Lipsitz LA, Veves A, Stein J, Bonato P, Collins JJ (2006) Noise-enhanced balance control in patients with diabetes and patients with stroke. Ann Neurol 59:4–12. https://doi.org/10.1002/ana.20670
Dettmer M, Pourmoghaddam A, Lee B-C, Layne CS (2015) Effects of aging and tactile stochastic resonance on postural performance and postural control in a sensory conflict task. Somatosens Mot Res 32:1–8. https://doi.org/10.3109/08990220.2015.1004045
Magalhaes FH, Kohn AF (2011) Vibratory noise to the fingertip enhances balance improvement associated with light touch. Exp Brain Res 209:139–151. https://doi.org/10.1007/s00221-010-2529-3
Collins JJ, Imhoff TT, Grigg P (1996) Noise-enhanced tactile sensation. Nature 383:770–770. https://doi.org/10.1038/383770a0
Lakshminarayanan K, Lauer AW, Ramakrishnan V, Webster JG, Seo NJ (2015) Application of vibration to wrist and hand skin affects fingertip tactile sensation. Phys Rep 3:e12465. https://doi.org/10.14814/phy2.12465
Mendez-Balbuena I, Manjarrez E, Schulte-Monting J, Huethe F, Tapia JA, Hepp-Reymond MC, Kristeva R (2012) Improved sensorimotor performance via stochastic resonance. J Neurosci 32:12612–12618. https://doi.org/10.1523/JNEUROSCI.0680-12.2012
Toledo DR, Barela JA, Kohn AF (2017) Improved proprioceptive function by application of subsensory electrical noise: effects of aging and task-demand. Neuroscience. 358:103–114. https://doi.org/10.1016/j.neuroscience.2017.06.045
Manjarrez E, Diez-Martı́nez O, Méndez I, Flores A (2002) Stochastic resonance in human electroencephalographic activity elicited by mechanical tactile stimuli. Neurosci Lett 324:213–216. https://doi.org/10.1016/S0304-3940(02)00212-4
Manjarrez E, Rojas-Piloni G, Mendez I, Flores A (2003) Stochastic resonance within the somatosensory system: effects of noise on evoked field potentials elicited by tactile stimuli. J Neurosci 23:1997–2001
Trenado C, Amtage F, Huethe F, Schulte-Mönting J, Mendez-Balbuena I, Baker SN, Baker M, Hepp-Reymond MC, Manjarrez E, Kristeva R (2014) Suppression of enhanced physiological tremor via stochastic noise: initial observations. PLoS One 9:e112782. https://doi.org/10.1371/journal.pone.0112782
Trenado C, Mikulić A, Manjarrez E, Mendez-Balbuena I, Schulte-Mönting J, Huethe F, Hepp-Reymond MC, Kristeva R (2014) Broad-band Gaussian noise is most effective in improving motor performance and is most pleasant. Front Hum Neurosci 8:22. https://doi.org/10.3389/fnhum.2014.00022
McDonnell MD, Ward LM (2011) The benefits of noise in neural systems: bridging theory and experiment. Nat Rev Neurosci 12:415–426. https://doi.org/10.1038/nrn3061
Moss F (2004) Stochastic resonance and sensory information processing: a tutorial and review of application. Clin Neurophysiol 115:267–281. https://doi.org/10.1016/j.clinph.2003.09.014
McDonnell MD, Abbott D (2009) What is stochastic resonance? Definitions, misconceptions, debates, and its relevance to biology. PLoS Comput Biol 5:e1000348. https://doi.org/10.1371/journal.pcbi.1000348
Durand DM, Kawaguchi M, Mino H (2013) Reverse stochastic resonance in a hippocampal CA1 neuron model. In: 2013 35th Annu. Int. Conf. IEEE Eng. Med. Biol. Soc. IEEE, pp 5242–5245
Iliopoulos F, Nierhaus T, Villringer A (2014) Electrical noise modulates perception of electrical pulses in humans: sensation enhancement via stochastic resonance. J Neurophysiol 111:1238–1248. https://doi.org/10.1152/jn.00392.2013
Slifkin AB, Newell KM (2000) Variability and noise in continuous force production. J Mot Behav 32:141–150. https://doi.org/10.1080/00222890009601366
Jones KE, Hamilton AF, Wolpert DM (2002) Sources of signal-dependent noise during isometric force production. J Neurophysiol 88:1533–1544
Watanabe RN, Magalhães FH, Elias LA, Chaud VM, Mello EM, Kohn AF (2013) Influences of premotoneuronal command statistics on the scaling of motor output variability during isometric plantar flexion. J Neurophysiol 110:2592–2606. https://doi.org/10.1152/jn.00073.2013
Dideriksen JL, Negro F, Enoka RM, Farina D (2012) Motor unit recruitment strategies and muscle properties determine the influence of synaptic noise on force steadiness. J Neurophysiol 107:3357–3369. https://doi.org/10.1152/jn.00938.2011
Kurita Y, Shinohara M, Ueda J (2013) Wearable sensorimotor enhancer for fingertip based on stochastic resonance effect. IEEE Trans Human-Machine Syst 43:333–337. https://doi.org/10.1109/TSMC.2013.2242886
Germer CM, Moreira LS, Elias LA (2017) Enhancement of force steadiness induced by sinusoidal vibrotactile stimulation depends on contraction intensity. 47th Annu. Meet. Soc. Neurosci
Oldfield RC (1971) The assessment and analysis of handedness: the Edinburgh Inventory. Neuropsychologia 9:97–113. https://doi.org/10.1016/0028-3932(71)90067-4
Abraira VE, Ginty DD (2013) The sensory neurons of touch. Neuron 79:618–639. https://doi.org/10.1016/j.neuron.2013.07.051
Sato M (1961) Response of Pacinian corpuscles to sinusoidal vibration. J Physiol 159:391–409. https://doi.org/10.1113/jphysiol.1961.sp006817
Germer C, Moreira L, Elias LA (2018) Force control with vibrotactile stimulation. https://doi.org/10.6084/m9.figshare.7447907.v1
Trenado C, Mendez-Balbuena I, Manjarrez E, Huethe F, Schulte-Mönting J, Feige B, Hepp-Reymond MC, Kristeva R (2014) Enhanced corticomuscular coherence by external stochastic noise. Front Hum Neurosci 8:1–10. https://doi.org/10.3389/fnhum.2014.00325
Slifkin AB, Newell KM (1999) Noise, information transmission, and force variability. J Exp Psychol 25:837–851
Pincus SM (1991) Approximate entropy as a measure of system complexity. Proc Natl Acad Sci 88:2297–2301. https://doi.org/10.1073/pnas.88.6.2297
Lodha N, Christou EA (2017) Low-frequency oscillations and control of the motor output. Front Physiol 8:1–9. https://doi.org/10.3389/fphys.2017.00078
Negro F, Holobar A, Farina D (2009) Fluctuations in isometric muscle force can be described by one linear projection of low-frequency components of motor unit discharge rates. J Physiol 587:5925–5938. https://doi.org/10.1113/jphysiol.2009.178509
Novak T, Newell KM (2017) Physiological tremor (8–12Hz component) in isometric force control. Neurosci Lett 641:87–93. https://doi.org/10.1016/j.neulet.2017.01.034
Cohen J (1988) Statistical power analysis for the behavioral sciences, 2nd ed. Hillsdale: Lawrence Erlbaum. p 567
Seo NJ, Kosmopoulos ML, Enders LR, Hur P (2014) Effect of remote sensory noise on hand function post stroke. Front Hum Neurosci 8:1–19. https://doi.org/10.3389/fnhum.2014.00934
Freeman AW, Johnson KO (1982) Cutaneous mechanoreceptors in macaque monkey: temporal discharge patterns evoked by vibration, and a receptor model. J Physiol 323:21–41. https://doi.org/10.1113/jphysiol.1982.sp014059
Baweja HS, Patel BK, Martinkewiz JD, Vu J, Christou EA (2009) Removal of visual feedback alters muscle activity and reduces force variability during constant isometric contractions. Exp Brain Res 197:35–47. https://doi.org/10.1007/s00221-009-1883-5
Harris CM, Wolpert DM (1998) Signal-dependent noise determines motor planning. Nature 394:780–784. https://doi.org/10.1038/29528
Keenan KG, Valero-Cuevas FJ (2007) Experimentally valid predictions of muscle force and EMG in models of motor-unit function are Most sensitive to neural properties. J Neurophysiol 98:1581–1590. https://doi.org/10.1152/jn.00577.2007
Calvin WH, Stevens CF (1967) Synaptic noise as a source of variability in the interval between action potentials. Science 155(80):842–844. https://doi.org/10.1126/science.155.3764.842
Moritz CT, Barry BK, Pascoe MA, Enoka RM (2005) Discharge rate variability influences the variation in force fluctuations across the working range of a hand muscle. J Neurophysiol 93:2449–2459. https://doi.org/10.1152/jn.01122.2004
Ofori E, Loucks TMJ, Sosnoff JJ (2012) Visuomotor and audiomotor processing in continuous force production of oral and manual effectors. J Mot Behav 44:87–96. https://doi.org/10.1080/00222895.2012.654523
Sosnoff JJ, Newell KM (2005) Intermittent visual information and the multiple time scales of visual motor control of continuous isometric force production. Percept Psychophys 67:335–344
Moon H, Kim C, Kwon M, Chen YT, Onushko T, Lodha N, Christou EA (2014) Force control is related to low-frequency oscillations in force and surface EMG. PLoS One 9:e109202. https://doi.org/10.1371/journal.pone.0109202
Nagamori A, Laine CM, Valero-Cuevas FJ (2018) Cardinal features of involuntary force variability can arise from the closed-loop control of viscoelastic afferented muscles. PLoS Comput Biol 14:e1005884. https://doi.org/10.1371/journal.pcbi.1005884
Aimonetti JM, Vedel JP, Schmied A, Pagni S (2000) Mechanical cutaneous stimulation alters Ia presynaptic inhibition in human wrist extensor muscles: a single motor unit study. J Physiol 522:137–145. https://doi.org/10.1111/j.1469-7793.2000.0137m.x
Nakashima K, Rothwell JC, Day BL, Thompson PD, Marsden CD (1990) Cutaneous effects on presynaptic inhibition of flexor Ia afferents in the human forearm. J Physiol 426:369–380
Kurita Y, Sueda Y, Ishikawa T, Hattori M, Sawada H, Egi H, Ohdan H, Ueda J, Tsuji T (2016) Surgical grasping forceps with enhanced sensorimotor capability via the stochastic resonance effect. IEEE/ASME Trans Mechatronics 21:2624–2634. https://doi.org/10.1109/TMECH.2016.2591591
Acknowledgments
The authors are thankful to Mr. Carlos Silva, Mr. Mauro Martinazo, Mr. Renato Moura, and Mr. Flavio Santos (Center for Biomedical Engineering, UNICAMP) for their technical support. C.M.G and L.S.M are recipients of PhD scholarships from Coordination for the Improvement of Higher Education Personnel (CAPES, Brazil).
Funding
L.A.E was funded by Research Grants from Teaching, Research, and Extension Support Fund of the University of Campinas (FAEPEX/UNICAMP, procs. nos. 1483/14 and 3289/16), CNPq (Brazilian NSF, proc. no. 312442/2017-3), and FAPESP (The Sao Paulo Research Foundation, proc. no. 2017/22191-3).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki declaration and its later amendments or comparable ethical standards.
Additional information
Publisher’s note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
About this article
Cite this article
Germer, C.M., Moreira, L.S. & Elias, L.A. Sinusoidal vibrotactile stimulation differentially improves force steadiness depending on contraction intensity. Med Biol Eng Comput 57, 1813–1822 (2019). https://doi.org/10.1007/s11517-019-01999-8
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11517-019-01999-8