Abstract
Object
Developments in open magnetic resonance imaging (MRI) magnets have made possible the use of reproducible thermosensitive sequences to determine temperature distribution inside biological tissue. This study aimed to compare MR thermal mapping during laser-induced interstitial thermal therapy (LITT) with macroscopically observed thermal lesions in order to estimate the 3D size of the coagulative necrosis.
Materials and methods
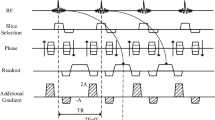
Laser irradiation was performed ex vivo with a 980-nm laser in pig liver in an open low-magnetic field (0.2 T) scanner. Laser light was transmitted through a 1,040/600 μm (outer/core diameter) bare-tipped silica fiber. Laser energy was applied in a pulsed mode (10 s laser-on, 10 s laser-off) for 12 min, power 6 W, energy 2,160 J. Gradient-echo images acquired during laser irradiation were used for real-time temperature mapping by the MR-T1 method. The method was then validated by a comparison between calculated 60°C isotherm and macroscopic lesion size.
Results
Temperature accuracy was 2.2°C, temporal resolution was 20 s. and spatial resolution was 2.5 × 2.5 × 2.5 mm3 (0.8% of the mean volume of coagulative necrosis). The mean lesion volume was 1830 mm3 ± 189 (standard error), σ (standard deviation) = 499 and range (min = 1281; max = 2591) mm3. Volumes calculated from MRI isotherms were correlated (correlation coefficient r 2 = 0.70) significantly (P = 0.08) to lesion size determined from macroscopic measurements.
Conclusion
Using fast gradient-echo sequence, laser monitoring is achieved efficiently with fast temperature mapping. T1-weighted images appear promising in monitoring lesion size evolution in future low magnetic field in vivo studies.
Similar content being viewed by others
References
Amin Z, Donald JJ, Masters A, Kant R, Steger AC, Bown SG and Lees WR (1993). Hepatic metastases: interstitial laser photocoagulation with real-time us monitoring and dynamic ct evaluation of treatment. Radiology 187(2): 339–347
Vogl TJ, Eichler K, Zangos S and Mack MG (2005). Interstitial laser therapy of liver tumors. Med Laser Appl 20(2): 115–118
Dickinson RJ, Hall AS, Hind AJ and Young IR (1986). Measurement of changes in tissue temperature using mr imaging. J Comput Assist Tomogr 10(3): 468–472
Vogl TJ, Straub R, Zangos S, Mack MG and Eichler K (2004). MR-guided laser-induced thermotherapy (litt) of liver tumours: experimental and clinical data. Int J Hyperthermia 20(7): 713–724
Germain D, Chevallier P, Laurent A, Savart M, Wassef M and Saint-Jalmes H (2001). MR monitoring of laser-induced lesions of the liver in vivo in a low-field open magnet: temperature mapping and lesion size prediction. J Magn Reson Imaging 13(1): 42–49
Dick EA, Wragg P, Joarder R, de Jode M, Lamb G, Gould S and Gedroyc WMW (2003). Feasibility of abdomino-pelvic T1-weighted real-time thermal mapping of laser ablation. J Magn Reson Imaging 17(2): 197–205
Germer CT, Albrecht D, Isbert C, Ritz J, Roggan A and Buhr HJ (1999). Diffusing fibre tip for the minimally invasive treatment of liver tumours by interstitial laser coagulation (ilc): an experimental ex vivo study. Lasers Med Sci 14(1): 32–39
Ivarsson K, Olsrud J, Sturesson C, Moller PH, Persson BR and Tranberg KG (1998). Feedback interstitial diode laser (805 nm) thermotherapy system: ex vivo evaluation and mathematical modeling with one and four-fibers. Lasers Surg Med 22(2): 86–96
Jolesz FA (1988). MR imaging of laser-tissue interactions. Radiology 168(1): 249–253
McNichols RJ, Gowda A, Kangasniemi M, Bankson JA, Price RE and Hazle JD (2004). MR thermometry-based feedback control of laser interstitial thermal therapy at 980 nm. Lasers Surgery Med 34(1): 48–55
Wlodarczyk W, Hentschel M, Wust P, Noeske R, Hosten N, Rinneberg H and Felix R (1999). Comparison of four magnetic resonance methods for mapping small temperature changes. Phys Med Biol 44(2): 607–624
Eyrich GK, Bruder E, Hilfiker P, Dubno B, Quick HH, Patak MA, Gratz KW and Sailer HF (2000). Temperature mapping of magnetic resonance-guided laser interstitial thermal therapy (litt) in lymphangiomas of the head and neck. Lasers Surg Med 26(5): 467–476
Kangasniemi M, McNichols RJ, Bankson JA, Gowda A, Price RE and Hazle JD (2004). Thermal therapy of canine cerebral tumors using a 980 nm diode laser with MR temperature-sensitive imaging feedback. Lasers Surg Med 35(1): 41–50
Jolesz FA (2005). Future perspectives for intraoperative MRI. Neurosurg Clin North Am 16(1): 201–213
Mack MG, Lehnert T, Eichler K and Vogl TJ (2005). MR-guided laser ablation. Magn Reson Imaging Clin North Am 13(3): 583–594
Gedroyc WM (2005). Magnetic resonance guidance of thermal ablation. Topics Magn Reson Imaging 16(5): 339–353
McNichols RJ, Kangasniemi M, Gowda A, Bankson JA, Price RE and Hazle JD (2004). Technical developments for cerebral thermal treatment: water-cooled diffusing laser fibre tips and temperature-sensitive mri using intersecting image planes. Int J Hyperthermia 20(1): 45–56
Germain D, Vahala E, Ehnholm GJ, Vaara T, Ylihautala M, Savart M, Laurent A, Tanttu J and Saint-Jalmes H (2002). MR temperature measurement in liver tissue at 0. 23 T with a steady-state free precession sequence. Magn Reson Med 47(5): 940–947
Prudhomme M, Mattei-Gazagnes M, Fabbro-Peray P, Puche P, Chabalier JP, Delacretaz G, Francois-Michel LF and Godlewski G (2003). MRI thermodosimetry in laser-induced interstitial thermotherapy. Lasers Surg Med 32(1): 54–60
Muralidharan V and Christophi C (2001). Interstitial laser thermotherapy in the treatment of colorectal liver metastases. J Surg Oncol 76(1): 73–81
Sturesson C, Liu DL, Stenram U and ndersson-Engels S (1997). Hepatic inflow occlusion increases the efficacy of interstitial laser-induced thermotherapy in rat. J Surg Res 71(1): 67–72
Hines-Peralta AU, Pirani N, Clegg P, Cronin N, Ryan TP, Liu Z and Goldberg SN (2006). Microwave Ablation: Results with a 2.45-GHz Applicator in ex vivo bovine and in vivo porcine liver. Radiology 239: 94–102
Aschoff AJ, Merkle EM, Wong V, Zhang Q, Mendez MM, Duerk JL and Lewin JS (2001). How does alteration of hepatic blood flow affect liver perfusion and radiofrequency-induced thermal lesion size in rabbit liver?. J Magn Reson Imaging 13(1): 57–63
Veenendaal LM, de Jager A, Stapper G, Borel Rinkes I and van Hillegersberg R (2006). Multiple fiber laser-induced thermotherapy for ablation of large intrahepatic tumors. Photomed Laser Surg 24(1): 3–9
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Viard, R., Piron, B., Steiner, A. et al. Non-invasive 3d magnetic resonance thermal mapping: determination of the lesion size during laser-therapy in ex vivo tissues. Int J CARS 2, 327–334 (2008). https://doi.org/10.1007/s11548-007-0141-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-007-0141-1