Abstract
Purpose
Accurate lung tumor segmentation is a prerequisite for effective radiation therapy and surgical planning. However, tumor delineation is challenging when the tumor boundaries are indistinct on PET or CT. To address this problem, we developed a segmentation method to improve the delineation of primary lung tumors from PET–CT images.
Methods
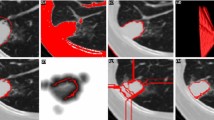
We formulated the segmentation problem as a label information propagation process in an iterative manner. Our model incorporates spatial–topological information from PET and local intensity changes from CT. The topological information of the regions was extracted based on the metabolic activity of different tissues. The spatial–topological information moderates the amount of label information that a pixel receives: The label information attenuates as the spatial distance increases and when crossing different topological regions. Thus, the spatial–topological constraint assists accurate tumor delineation and separation. The label information propagation and transition model are solved under a random walk framework.
Results
Our method achieved an average DSC of \(0.848 \pm 0.036\) and HD (mm) of \(8.652 \pm 4.532\) on 40 patients with lung cancer. The t test showed a significant improvement (p value \(<\) 0.05) in segmentation accuracy when compared to eight other methods. Our method was better able to delineate tumors that had heterogeneous FDG uptake and which abutted adjacent structures that had similar densities.
Conclusions
Our method, using a spatial–topological constraint, provided better lung tumor delineation, in particular, when the tumor involved or abutted the chest wall and the mediastinum.














Similar content being viewed by others
References
MacManus M et al (2009) Use of PET and PET/CT for radiation therapy planning: IAEA expert report 2006–2007. Radiother Oncol 91(1):85–94
Jemal A, Bray F, Center MM, Ferlay J, Ward E, Forman D (2011) Global cancer statistics. Cancer J Clin 61(2):69–90
Kirov A, Fanchon L (2014) Pathology-validated PET image data sets and their role in PET segmentation. Clin Transl Imaging 2(3):253–267
Simona B-H, Peter E (2009) 18F-FDG PET and PET/CT in the evaluation of cancer treatment response. J Nucl Med 50(1):88–99
Boellaard R (2012) Mutatis mutandis: harmonize the standard!. J Nucl Med 53(1):1–3
Hatt M et al (2009) A fuzzy locally adaptive Bayesian segmentation approach for volume determination in PET. IEEE Trans Med Imaging 28(6):881–893
Hatt M et al (2010) Accurate automatic delineation of heterogeneous functional volumes in positron emission tomography for oncology applications. Int J Radiat Oncol Biol Phys 77(1):301–308
Bagci U et al (2013) Joint segmentation of anatomical and functional images: applications in quantification of lesions from PET, PET–CT, MRI–PET, and MRI–PET–CT images. Med Image Anal 17(8):929–945
Foster B et al (2014) Segmentation of PET images for computer-aided functional quantification of tuberculosis in small animal models. IEEE Trans Biomed Eng 61(3):711–724
Foster B et al (2014) A review on segmentation of positron emission tomography images. Comput Biol Med 50:76–96
Wang X et al (2014) Lung tumor delineation based on novel tumor-background likelihood models in PET–CT images. IEEE Trans Nucl Sci 61(1):218–224
Han D et al (2011) Globally optimal tumor segmentation in PET-CT images: a graph-based co-segmentation method. Inf Process Med Imaging 22:245–256
Xu Z et al (2014) Fuzzy connectedness image co-segmentation for hybrid PET/MRI and PET/CT scans. In: Computational methods for molecular imaging workshop, medical image computing and computer-assisted intervention (MICCAI), Boston
Boykov Y, Veksler O, Zabih R (2001) Fast approximate energy minimization via graph cuts. IEEE Trans Pattern Anal Mach Intell 23(11):1222–1239
Grady L (2006) Random walks for image segmentation. IEEE Trans Pattern Anal Mach Intell 28(11):1768–1783
Qi S et al (2013) Optimal co-segmentation of tumor in PET–CT images with context information. IEEE Trans Med Imaging 32(9):1685–1697
Grady L et al (2005) Random walks for interactive organ segmentation in two and three dimensions: implementation and validation. In: Medical image computing and computer-assisted intervention, pp 773–780
Chen M et al (2011) Random walk-based automated segmentation for the prognosis of malignant pleural mesothelioma. In: IEEE international symposium on biomedical imaging: from nano to macro, pp 1978–1981
Carr H, Snoeyink J, Axen U (2003) Computing contour trees in all dimensions. Comput Geom Theory Appl 24(2):75–94
Zhou J (2012) Gaining insights into volumetric data visualization: a semi-automatic transfer function generation approach using contour tree analyses. LAP LAMBERT Academic Publishing, Germany
Cui H et al (2014) Topology constraint graph-based model for non-small-cell lung tumor segmentation from PET volumes. In: IEEE international symposium on biomedical imaging (ISBI), pp 1243–1246
RIDER Phantom PET–CT: University of Washington. https://wiki.cancerimagingarchive.net/display/Public/RIDER+Collections
Black QC et al (2004) Defining a radiotherapy target with positron emission tomography. Int J Radiat Oncol Biol Phys 60(4):1272–1282
Ballangan C et al (2011) Automated delineation of lung tumors in PET images based on monotonicity and a tumor-customized criterion. IEEE Trans Inf Technol Biomed 15(5):691–702
Kim TH, Lee KM, Lee SU (2010) Nonparametric higher-order learning for interactive segmentation. In: IEEE conference on computer vision and pattern recognition, pp 3201–3208
Grady L (2003) The graph analysis toolbox: image processing on arbitrary graphs 2003. Boston University, Boston, MA, Tech. Rep. TR-03-021
Kim TH. Generative image segmentation using random walks with restart. http://cv.snu.ac.kr/thkim/RWR_ECCV08/index.html
Kim TH, Lee KM, Lee SU (2008) Generative image segmentation using random walks with restart. In: European conference on computer vision, pp 264–275
Kinahan PE, Doot RK, Wanner-Roybal M, Bidaut LM, Armato SG, Meyer CR, McLennan G (2009) PET/CT assessment of response to therapy: tumor change measurement, truth data, and error. Transl Oncol 2(4):223–230
Shah B et al (2012) Intra-reader reliability of FDG PET volumetric tumor parameters: effects of primary tumor size and segmentation methods. Ann Nucl Med 26(9):707–714
Zhou J, Xiao C, Takatsuka M (2013) A multi-dimensional importance metric for contour tree simplification. J Vis 16(4):341–349
Conflict of interest
Hui Cui, Xiuying Wang, Weiran Lin, Jianlong Zhou, Stefan Eberl, Dagan Feng, and Michael Fulham declare that they have no conflict of interest.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Cui, H., Wang, X., Lin, W. et al. Primary lung tumor segmentation from PET–CT volumes with spatial–topological constraint. Int J CARS 11, 19–29 (2016). https://doi.org/10.1007/s11548-015-1231-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-015-1231-0