Abstract
Purpose
Radiographic parameters (RPs) provide objective support for effective decision making in determining clinical treatment of distal radius fractures (DRFs). This paper presents a novel automatic RP computation pipeline for computing the six anatomical RPs associated with DRFs in anteroposterior (AP) and lateral (LAT) forearm radiographs.
Methods
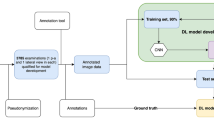
The pipeline consists of: (1) segmentation of the distal radius and ulna bones with six 2D Dynamic U-Net deep learning models; (2) landmark points detection and distal radius axis computation from the segmentations with geometric methods; (3) RP computation and generation of a quantitative DRF report and composite AP and LAT radiograph images. This hybrid approach combines the advantages of deep learning and model-based methods.
Results
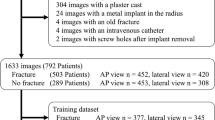
The pipeline was evaluated on 90 AP and 93 LAT radiographs for which ground truth distal radius and ulna segmentations and RP landmarks were manually obtained by expert clinicians. It achieves an accuracy of 94 and 86% on the AP and LAT RPs, within the observer variability, and an RP measurement difference of 1.4 ± 1.2° for the radial angle, 0.5 ± 0.6 mm for the radial length, 0.9 ± 0.7 mm for the radial shift, 0.7 ± 0.5 mm for the ulnar variance, 2.9 ± 3.3° for the palmar tilt and 1.2 ± 1.0 mm for the dorsal shift.
Conclusion
Our pipeline is the first fully automatic method that accurately and robustly computes the RPs for a wide variety of clinical forearm radiographs from different sources, hand orientations, with and without cast. The computed accurate and reliable RF measurements may support fracture severity assessment and clinical management.





Similar content being viewed by others
References
Crowe S, Massenburg B, Massenburg C, Morrison B, Morrison D (2020) Global trends of hand and wrist trauma: a systematic analysis of fracture and digit amputation using the Global Burden of Disease 2017 Study. Inj Prev 26(2):i115–i124
Rundgren J, Bojan A, Mellstrand Navarro C, Enocson A (2020) Epidemiology, classification, treatment and mortality of distal radius fractures in adults: an observational study of 23,394 fractures from the National Swedish fracture register. BMC Musculoskelet Disord 21(1):1–9
Kamal RN, Shapiro LM (2020) American Academy of Orthopaedic Surgeons. American Society for Surgery of the Hand Clinical Practice Guideline summary management of distal radius fractures. J Am Acad Orthopaedic Surg 30(4):e480ee486.
Solgaard S (1988) Function after distal radius fracture. Acta Orthop Scand 59(1):39–42
Brogren E, Hofer M, Petranek M, Wagner P, Dahlin LB, Atroshi I (2011) Relationship between distal radius fracture malunion and arm-related disability: a prospective population-based cohort study with 1-year follow-up. BMC Musculoskelet Disord 12(1):1–9
Wilcke MK, Abbaszadegan H, Adolphson PY (2007) Patient-perceived outcome after displaced distal radius fractures: a comparison between radiological parameters, objective physical variables and the DASH score. J Hand Therapy 20(4):290–299
Kreder HJ, Hanel DP, McKee M, Jupiter J, McGillivary G, Swiontkowski MF (1996) X-ray film measurements for healed distal radius fractures. J Hand Surgery 21(1):31–39
Best practice for management of Distal Radial Fractures (DRFs). British Orthopaedic Association and British Society for Surgery of the Hand, 2018. https://www.bssh.ac.uk/professionals/management_of_distal_radial_fractures.aspx
Management of Distal Radius Fractures evidence-based clinical practice guideline. American Academy of Orthopedic Surgeons, 5 December 2020.
Johnson P, Szabo RM (1993) Angle measurements of the distal radius: a cadaver study. Skeletal Radiol 22(4):243–246
Watson NJ, Asadollahi S, Parrish F, Ridgway J, Tran P, Keating JL (2016) Reliability of radiographic measurements for acute distal radius fractures. BMC Med Imag 16(1):1–9
Hossain M, Andrew J (2008) Reliability of a digital radiographic system in measuring distal radial fracture displacement parameters. Euro J Orthop Surg Traumatol 18(8):565–569
Raisuddin AM, Vaattovaara E, Nevalainen M, Nikki M, Järvenpää E, Makkonen K, Pinola P, Palsio T, Niemensivu A, Tervonen O, Tiulpin A (2021) Critical evaluation of deep neural networks for wrist fracture detection. Sci Rep 11(1):6006. https://doi.org/10.1038/s41598-021-85570-2
Davidson A, Suna A, Joskowicz L, Weil Y (2022) Computer generated radiographic measurements of distal radius fractures—does it help with decision making? J Hand Surg Am. https://doi.org/10.1016/j.jhsa.2022.09.015
Payer C, Štern D, Bischof H, Urschler M (2019) Integrating spatial configuration into heatmap regression based CNNs for landmark localization. Med Image Anal 54:207–219
Rouzrokh P, Wyles CC, Kurian SJ, Ramazanian T, Cai JC, Huang Q, Erickson BJ (2022) Deep learning for radiographic measurement of femoral component subsidence following total hip arthroplasty. Radiol Artif Intell 4(3):e210206.
Zheng Q, Shellikeri S, Huang H, Hwang M, Sze RW (2020) Deep learning measurement of leg length discrepancy in children based on radiographs. Radiology 296(1):152–158
Yan K, Tang Y, Peng Y, Sandfort V, Bagheri M, Lu Z, Summers RM (2019) MULAN: multitask universal lesion analysis network for joint lesion detection, tagging, and segmentation. In: Proceedings of 21st international conference on medical image computing and computer-assisted intervention, pp. 194–202.
Bano S, Dromey B, Vasconcelos F, Napolitano R, David AL, Peebles DM, Stoyanov D (2021) AutoFB: Automating fetal biometry estimation from standard ultrasound planes. In: Proceedings of 23rd international conference on medical image computing and computer-assisted intervention, pp. 228–238.
Avisdris N, Yehuda B, Ben-Zvi O, Link-Sourani D, Ben-Sira L, Miller E, Joskowicz L (2021) Automatic linear measurements of the fetal brain on MRI with deep neural networks. Int J Comput Assist Radiol Surg 16(9):1481–1492.
Kang BK, Han Y, Oh J, Lim J, Ryu J, Yoon MS, Ryu S (2022) Automatic segmentation for favourable delineation of ten wrist bones on wrist radiographs using Convolutional Neural Network. J Personalized Med 12(5):776
Lindsey R, Daluiski A, Chopra S, Lachapelle A, Mozer M, Sicular S, Potter H (2018) Deep neural network improves fracture detection by clinicians. Proc Natl Acad Sci 115(45):11591–11596
Thian YL, Li Y, Jagmohan P, Sia D, Chan, VEY, Tan RT (2019) Convolutional neural networks for automated fracture detection and localization on wrist radiographs. Radiol Artif Intell 1(1).
Blüthgen C, Becker AS, de Martini IV, Meier A, Martini K, Frauenfelder T (2020) Detection and localization of distal radius fractures: deep learning system versus radiologists. Euro J Radiol 126:108925
Suzuki T, Maki S, Yamazaki T, Wakita H, Toguchi Y, Horii M, Ohtori S (2022) Detecting distal radial fractures from wrist radiographs using a deep convolutional neural network with an accuracy comparable to hand orthopedic surgeons. J Digital Imaging 35(1):39–46
Khan MA, Sharif M, Akram T, Bukhari SAC, Nayak RS (2020) Developed Newton-Raphson based deep features selection framework for skin lesion recognition. Pattern Recogn Lett 129:293–303
Lin TY, Goyal P, Girshick R, He K, Dollár P (2017) Focal loss for dense object detection. In: Proceedings of IEEE international conference on computer vision, pp. 2980–2988.
MURA dataset: bone X-Ray deep learning competition, Stanford Machine Learning Group, https://stanfordmlgroup.github.io/competitions/mura/, last visited Nov 14, 2022.
Yushkevich PA, Gao Y, Gerig G (2016) ITK-SNAP: An interactive tool for semi-automatic segmentation of multi-modality biomedical images. In: Proceedings of 38th International IEEE Conference on Engineering, Medicine and Biology. IEEE, New York, pp 3342–3345.
Howard J, Gugger S (2020) Fastai: a layered API for deep learning. Information 11(2):108
Vingelmann P. Fitzek FHP. NVIDIA CUDA Release 10.2.89, 2020.
Reyes-Aldasoro CC, Ngan KH, Ananda A, d'Avila Garcez A, Appelboam A, Knapp KM (2020) Geometric semi-automatic analysis of radiographs of Colles' fractures PLoS ONE 15(9):e0238926. https://doi.org/10.1371/journal.pone.0238926.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Ethical approval
All procedures performed in studies involving human participants were in accordance with the ethical standards of the institutional and/or national research committee and with the 1964 Helsinki Declaration and its later amendments or comparable ethical standards.
Informed consent
There was no need for informed consent required for the work reported in this manuscript.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Suna, A., Davidson, A., Weil, Y. et al. Automated computation of radiographic parameters of distal radial metaphyseal fractures in forearm X-rays. Int J CARS 18, 2179–2189 (2023). https://doi.org/10.1007/s11548-023-02907-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s11548-023-02907-0