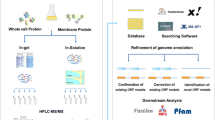
Abstract
Bordetella pertussis Tohama I, Bordetella parapertussis 12822, and Bordetella bronchiseptica RB50 are pathogenic β-proteobacteria. The B. pertussis and B. parapertussis are the main pathogenic species which cause whooping cough in humans. The availability of whole genome sequences of different strains of Bordetella has open up new avenues towards identification of genomic islands. The genomic islands are clusters of genes which are acquired from other species by horizontal transfer and have substantial impact on microbial adaptations which are associated with clinical and environmental aspects. In the present work, identification and comparative functional analysis of genomic islands have been carried out against the genome of these three species of Bordetella. A total of 15 genomic islands have been identified which encodes for 251 proteins. Out of these, 108 proteins are functionally annotated while 143 proteins were found to be hypothetical proteins. Among 108 proteins, 32 proteins were present in B. bronchiseptica, 03 in B. Parapertussis, and 73 in B. Pertussis. In addition, BLAST-P analysis was carried out to find the occurrence of these proteins in other species of Bordetella. Several unique proteins have been found in B. Bronchiseptica (06) and B. Pertussis (21) as they were not expressed in others species of Bordetella. Functional annotation of 108 proteins further revealed that 26 proteins are pathogen-associated, 53 proteins are involved in resistance, 19 proteins are responsible for virulence, and 10 proteins are associated with both virulence and pathogenicity. This study revealed that a significant number of genomic islands are acquired by horizontal gene transfer which play significant role in the evolution behaviour of Bordetella species. In conclusion, proteins encoded by predicted genomic islands might be associated with the development of increased virulence, pathogenicity, and host restriction for humans to B. pertussis in comparison to B. parapertussis and B. bronchiseptica. The novel encoded proteins can be exploited as potential therapeutic drug targets against Bordetella species.




Similar content being viewed by others
Data availability
The authors confirm that the data supporting the findings of this study are available within the article [and/or] its supplementary materials.
References
Bertelli C, Laird MR, Williams KP, Simon Fraser University Research Computing Group, Lau BY, Hoad G, Winsor GL, Brinkman FSL (2017) IslandViewer 4 expanded prediction of genomic islands for larger-scale datasets. Nucleic Acids Res 45(W1):W30–W35. https://doi.org/10.1093/nar/gkx343
Bertelli C, Tilley KE, Brinkman FSL (2019) Microbial genomic island discovery, visualization and analysis. Brief Bioinform 20(5):1685–1698. https://doi.org/10.1093/bib/bby042
Bertelli C, Gray KL, Woods N, Lim AC, Tilley KE, Winsor GL, Hoad GR, Roudgar A, Spencer A, Peltier J, Warren D, Raphenya AR, McArthur AG, Brinkman FSL (2022) Enabling genomic island prediction and comparison in multiple genomes to investigate bacterial evolution and outbreaks. Microb Genom 8(5):mgen000818
Bie L, Fang M, Li Z, Wang M, Xu H (2018) Identification and characterization of new resistance-conferring SGI1s (Salmonella Genomic Island 1) in Proteus mirabilis. Front Microbiol 9:3172. https://doi.org/10.3389/fmicb.2018.03172
Burrus V, Pavlovic G, Decaris B, Guedon G (2002) Conjugative transposons: the tip of the iceberg. Mol Microbiol 46:601–610
Che D, Hasan MS, Wang H, Fazekas J, Huang J, Liu Q (2011) EGID: an ensemble algorithm for improved genomic island detection in genomic sequences. Bioinformation 7(6):311–314. https://doi.org/10.6026/007/97320630007311
Chen L, Xiong Z, Sun L, Yang J, Jin Q (2012) VFDB 2012 update: toward the genetic diversity and molecular evolution of bacterial virulence factors. Nucleic Acids Res 40:D641-5. https://doi.org/10.1093/nar/gkr989
da Silva AR, Quiambao LG, Sutherland A, Soldevila F, Dhanda SK, Armstrong SK, Brickman TJ, Merkel T, Peters B, Sette A (2020) Development and validation of a bordetella pertussis whole-genome screening strategy. J Immunol Res. https://doi.org/10.1155/2020/8202067
Dobrindt U, Hochhut B, Hentschel U, Hacker J (2004) Genomic islands in pathogenic and environmental microorganisms. Nat Rev Microbiol 2:414–424
Gaillard M, Vallaeys T, Vorholter FJ (2006) The clc element of Pseudomonas sp. strain B13, a genomic island with various catabolic properties. J Bacteriol 188:1999–2013
Ghazali AK, Eng SA, Khoo JS, Teoh S, Hoh CC, Nathan S (2021) Whole-genome comparative analysis of Malaysian Burkholderia pseudomallei clinical isolates. Microb Genom 7(2):000527. https://doi.org/10.1099/mgen.0.000527
Hacker J, Carniel E (2001) Ecological fitness, genomic islands and bacterial pathogenicity. a darwinian view of the evolution of microbes. EMBO Rep 5:376–381. https://doi.org/10.1093/embo-reports/kve097
Hacker J, Kaper JB (2000) Pathogenicity islands and the evolution of microbes. Annu Rev Microbiol 54:641–679. https://doi.org/10.1146/annurev.micro.54.1.641
Hacker J, Bender L, Ott M, Wingender J, Lund B, Marre R, Goebel W (1990) Deletions of chromosomal regions coding for fimbriae and hemolysins occur in vitro and in vivo in various extraintestinal Escherichia coli isolates. Microb Pathog 8:213–225
Hamidou Soumana I, Linz B, Harvill ET (2017) Environmental origin of the Genus Bordetella. Front Microbiol 8:28. https://doi.org/10.3389/fmicb.2017.00028
Ho Sui SJ, Fedynak A, Hsiao WW, Langille MG, Brinkman FS (2009) The association of virulence factors with genomic islands. PLoS One 4(12):e8094. https://doi.org/10.1371/journal.pone.0008094
Hsiao WW, Ung K, Aeschliman D, Bryan J, Finlay BB, Brinkman FS (2005) Evidence of a large novel gene pool associated with prokaryotic genomic islands. PLoS Genet 1:e62. https://doi.org/10.1371/journal.pgen.0010062
Jia B, Raphenya AR, Alcock B, Waglechner N, Guo P, Tsang KK, Lago BA, Dave BM, Pereira S, Sharma AN, Doshi S, Courtot M, Lo R, Williams LE, Frye JG, Elsayegh T, Sardar D, Westman EL, Pawlowski AC, Johnson TA, Brinkman FS, Wright GD, McArthur AG (2017) CARD 2017: expansion and model-centric curation of the comprehensive antibiotic resistance database. Nucleic Acids Res 45(D1):D566–D573. https://doi.org/10.1093/nar/gkw1004
Juhas M, van der Meer JR, Gaillard M, Harding RM, Hood DW, Crook DW (2009) Genomic islands: tools of bacterial horizontal gene transfer and evolution. FEMS Microbiol Rev 33(2):376–393. https://doi.org/10.1111/j.1574-6976.2008.00136.x
Kilgore PE, Salim AM, Zervos MJ, Schmitt H-J (2016) Pertussis: microbiology, disease, treatment, and prevention. Clin Microbiol Rev 29:449–486. https://doi.org/10.1128/CMR.00083-15
Langille M, Hsiao W, Brinkman F (2010) Detecting genomic islands using bioinformatics approaches. Nat Rev Microbiol 8:373–382. https://doi.org/10.1038/nrmicro2350
Linz B, Ivanov YV, Preston A et al (2016) Acquisition and loss of virulence-associated factors during genome evolution and speciation in three clades of Bordetella species. BMC Genom 17:767. https://doi.org/10.1186/s12864-016-3112-5
McCarlie SJ, Boucher CE, Bragg RR (2023) Genomic islands identified in highly resistant serratia sp. HRI: a pathway to discover new disinfectant resistance elements. Microorganisms 11(2):515. https://doi.org/10.3390/microorganisms11020515
Nieves DJ, Heininger U (2016) Bordetella pertussis. Microbiol Spectr. https://doi.org/10.1128/microbiolspec.EI10-0008-2015
Park J, Zhang Y, Buboltz AM et al (2012) Comparative genomics of the classical Bordetella subspecies: the evolution and exchange of virulence-associated diversity amongst closely related pathogens. BMC Genom 13:545. https://doi.org/10.1186/1471-2164-13-545
Parkhill J, Sebaihia M, Preston A, Murphy LD, Thomson N, Harris DE, Holden MT, Churcher CM, Bentley SD, Mungall KL, Cerdeño-Tárraga AM, Temple L, James K, Harris B, Quail MA, Achtman M, Atkin R, Baker S, Basham D, Bason N, Cherevach I, Chillingworth T, Collins M, Cronin A, Davis P, Doggett J, Feltwell T, Goble A, Hamlin N, Hauser H, Holroyd S, Jagels K, Leather S, Moule S, Norberczak H, O’Neil S, Ormond D, Price C, Rabbinowitsch E, Rutter S, Sanders M, Saunders D, Seeger K, Sharp S, Simmonds M, Skelton J, Squares R, Squares S, Stevens K, Unwin L, Whitehead S, Barrell BG, Maskell DJ (2003) Comparative analysis of the genome sequences of Bordetella pertussis, Bordetella parapertussis and Bordetella bronchiseptica. Nat Genet 35(1):32–40. https://doi.org/10.1038/ng1227
Rao RT, Sharma S, Sivakumar N, Jayakumar K (2020) Genomic islands and the evolution of livestock-associated Staphylococcus aureus genomes. Biosci Rep 40(11):BSR20202287. https://doi.org/10.1042/BSR20202287
Schmidt H, Hensel M (2004) Pathogenicity islands in bacterial pathogenesis. Clin Microbiol Rev 17:14–56
Sharn HO, Singh DB, Singh S (2021) Comparative structural analysis of bifunctional methylenetetrahydrofolate dehydrogenase/methenyltetrahydrofolate cyclohydrolase from Bordetella pertussis and Bordetella parapertussis: a drug target against pertussis. In Silico Pharmacol 9(1):7. https://doi.org/10.1007/s40203-020-00069-4
van der Meer JR, Sentchilo V (2003) Genomic islands and the evolution of catabolic pathways in bacteria. Curr Opin Biotechnol 14(3):248–254. https://doi.org/10.1016/s0958-1669(03)00058-2. (PMID: 12849776)
Vernikos GS, Parkhill J (2008) Resolving the structural features of genomic islands: a machine learning approach. Genome Res 18:331–342
Wattam AR, Abraham D, Dalay O, Disz TL, Driscoll T, Gabbard JL, Gillespie JJ, Gough R, Hix D, Kenyon R, Machi D, Mao C, Nordberg EK, Olson R, Overbeek R, Pusch GD, Shukla M, Schulman J, Stevens RL, Sullivan DE, Vonstein V, Warren A, Will R, Wilson MJ, Yoo HS, Zhang C, Zhang Y, Sobral BW (2014) PATRIC, the bacterial bioinformatics database and analysis resource. Nucleic Acids Res 42:D581-91. https://doi.org/10.1093/nar/gkt1099
Weigand MR, Peng Y, Loparev V, Batra D, Bowden KE, Burroughs M, Cassiday PK, Davis JK, Johnson T, Juieng P, Knipe K, Mathis MH, Pruitt AM, Rowe L, Sheth M, Tondella ML, Williams MM (2017) The history of Bordetella pertussis genome evolution includes structural rearrangement. J Bacteriol 199:e00806-e816. https://doi.org/10.1128/JB.00806-16
Woolfrey BF, Moody JA (1991) Human infections associated with Bordetella bronchiseptica. Clin Microbiol Rev 4(3):243–255. https://doi.org/10.1128/CMR.4.3.243
Acknowledgements
The authors are grateful to the Department of Computational Biology and Bioinformatics, JIBB, Sam Higginbottom University of Agriculture, Technology & Sciences, Prayagraj, India for providing the facilities and support to complete this research work.
Author information
Authors and Affiliations
Corresponding authors
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Below is the link to the electronic supplementary material.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Sharn, H.O., Singh, D.B., Yadav, P.K. et al. Genome annotation and comparative functional analysis of genomic islands in Bordetella pertussis Tohama I, Bordetella parapertussis 12822, and Bordetella bronchiseptica RB50 genomes. Netw Model Anal Health Inform Bioinforma 12, 23 (2023). https://doi.org/10.1007/s13721-023-00418-1
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1007/s13721-023-00418-1