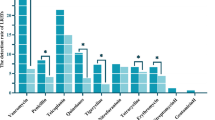
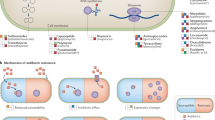
Abstract
The rise of antimicrobial resistance (AMR) in bacteria that cause infectious diseases poses a significant global health challenge, necessitating advanced prediction technology for effective management and response. This study aims to evaluate the predictive capabilities of several machine learning (ML) and deep learning (DL) models in identifying patterns of antibiotic resistance. We have proposed a comprehensive experimental framework that combines multiple machine learning algorithms and deep learning architectures to evaluate their efficacy and accuracy in predicting antibiotic resistance. We have developed DnnARs, an artificial intelligence pipeline designed to identify antibiotic-resistant strains of E. coli bacteria, which are mostly responsible for urinary tract infections (UTIs). DnnARs utilizes Synthetic Minority Over-sampling Technique (SMOTE) and applies feature engineering using principal component analysis (PCA) and t-distributed Stochastic neighbor embedding (t-SNE) on our dataset to evaluate the effectiveness of various AI models. Our model comparison demonstrates that DL models outperform ML models when the dataset is fine-tuned using SMOTE and feature engineering. Our DnnARs achieve a classification accuracy of 97%, a specificity of 91%, and a remarkable sensitivity of 100%. The DL models that were examined exhibit an average classification accuracy that is 3.66% higher than that of the ML models. The findings demonstrate the capability of deep learning methodology to enhance predictive analytics for antimicrobial resistance, offering a robust and efficient alternative to traditional machine learning techniques. This research aims to enhance diagnostic tools and medications, improve the management of infectious diseases, reduce antibiotic resistance, and integrate deep learning models into clinical workflows to enable immediate decision-making.









Similar content being viewed by others
Explore related subjects
Discover the latest articles, news and stories from top researchers in related subjects.Data Availability
The datasets utilized for this study are freely available and can be found at NCBI with accession numbers GSE98505 and GSE96706.
References
Bloom DE, Cadarette D. Infectious disease threats in the twenty-first century: strengthening the global response. Front Immunol. 2019;10:549.
Zhao AP et al., AI for science: Predicting infectious diseases. J Saf Sci Resilienc. 2024.
Abavisani M, Khoshrou A, Foroushan SK, Sahebkar A, Chatting with artificial intelligence to combat antibiotic resistance: Opportunities and challenges. Curr Res Biotechnol. 2024;7:100197. https://www.sciencedirect.com/science/article/pii/S2590262824000236
Tang KWK, Millar BC, Moore JE. Antimicrobial resistance (AMR). Br J Biomed Sci. 2023;80:11387.
Paneri M, Sevta P, "Overview of antimicrobial resistance: An emerging silent pandemic. Glob J Med Pharm Biomed Update. 2023;18. https://gjmpbu.org/overview-of-antimicrobial-resistance-an-emerging-silentpandemic/
Dadgostar P, "Antimicrobial resistance: implications and costs. Infect Drug Resis. 2019;12:3903–3910. https://www.tandfonline.com/doi/full/10.2147/IDR.S234610
Walsh TR, Gales AC, Laxminarayan R, Dodd PC Antimicrobial resistance: addressing a global threat to humanity, vol. 20, ed: Public Library of Science San Francisco, CA USA, 2023, e1004264.
Alowais SA, et al. Revolutionizing healthcare: the role of artificial intelligence in clinical practice. BMC Med Educ. 2023;23(1):689.
Noman SM, et al. Machine learning techniques for antimicrobial resistance prediction of pseudomonas aeruginosa from whole genome sequence data. Comput Intell Neurosci. 2023;2023(1):5236168.
Mutisya F, Kanguha R. AntiMicro. ai: an artificial Intelligence powered web application for predicting antibacterial/antifungal susceptibility and constructing personalized machine learning models. Wellcome Open Res. 2024;9:273.
Boulaamane Y, et al. Antibiotic discovery with artificial intelligence for the treatment of Acinetobacter baumannii infections. Msystems. 2024. https://doi.org/10.1128/msystems.00325-24.
Jin C, Jia C, Hu W, Xu H, Shen Y, Yue M. Predicting antimicrobial resistance in E. coli with discriminative position fused deep learning classifier. Comput Struct Biotechnol J. 2024;23:559–65.
Ahmad A, Hettiarachchi R, Khezri A, Singh Ahluwalia B, Wadduwage DN, Ahmad R. Highly sensitive quantitative phase microscopy and deep learning aided with whole genome sequencing for rapid detection of infection and antimicrobial resistance. Front Microbiol. 2023;14:1154620.
Tharmakulasingam M, Wang W, Kerby M, La Ragione R, Fernando A. "TransAMR: an interpretable transformer model for accurate prediction of antimicrobial resistance using antibiotic administration data. IEEE Access. 2023. https://doi.org/10.1109/ACCESS.2023.3296221.
Kim J, et al. Predicting antimicrobial resistance of bacterial pathogens using time series analysis. Front Microbiol. 2023;14:1160224.
Nayak DSK, et al. aiGeneR 10: an artificial intelligence technique for the revelation of informative and antibiotic resistant genes in escherichia coli. Front Biosci Landmark. 2024;29(2):82.
Nsubuga M, Galiwango R, Jjingo D, Mboowa G. Generalizability of machine learning in predicting antimicrobial resistance in E. coli: a multi-country case study in Africa. BMC Genom. 2024;25(1):287.
Nayak DSK et al. Artificial Intelligence in Battle against Antimicrobial Resistance: Opportunities and Challenges. Int J Recent Innovation Trends Comput Commun 2023;11(9):427–437. https://doi.org/10.17762/ijritcc.v11i9.8824
"GSE98505." https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE98505. Accessed 04 Feb 2024.
"GSE96706." https://www.ncbi.nlm.nih.gov/geo/query/acc.cgi?acc=GSE96706. Accessed 01 Feb 2024.
Mohanty S, Nayak DSK, Swarnkar T, "A neural network framework for predicting Adenocarcinoma cancer using high-throughput gene expression data," In AIP Conference Proceedings, 2023, 2819(1): AIP Publishing.
Nayak DSK, Mohapatra S, Al-Dabass D, Swarnkar T, "Deep learning approaches for high dimension cancer microarray data feature prediction: A review. Comput Intell Cancer Diagn. 2023;1:13–41. https://www.sciencedirect.com/science/article/abs/pii/B9780323852401000183
Nayak DSK, Pati A, Panigrahi A, Sahoo S, Swarnkar T, "ReCuRandom: A hybrid machine learning model for significant gene identification," In AIP Conference Proceedings, 2023, 2819(1): AIP Publishing.
Das R, Nayak DSK, Rout CP, Jena L, Swarnkar T, "Deep Learning Techniques for Identification of Pneumonia: A CNN Approach," In:2024 International Conference on Advancements in Smart, Secure and Intelligent Computing (ASSIC), 2024: IEEE, pp. 1–5.
Al‐Shaebi Z, Akdeniz M, Ahmed AO, Altunbek M, Aydin O, Breakthrough Solution for Antimicrobial Resistance Detection: Surface‐Enhanced Raman Spectroscopy‐based on Artificial Intelligence. Adv Mater Interfaces. 2023:13–41, 2300664. https://onlinelibrary.wiley.com/doi/full/10.1002/admi.202300664
Singh MP, Gollapalli P, Bagadi S, Ragul N, Sharma Y Assessment of Machine Learning Algorithms as an Emerging Model for Translational Research to Predict Antimicrobial Resistance in Clinically Relevant Pathogens, Available at SSRN 4817064.
Yan J, Cai J, Zhang B, Wang Y, Wong DF, Siu SW. Recent progress in the discovery and design of antimicrobial peptides using traditional machine learning and deep learning. Antibiotics. 2022;11(10):1451.
Jeatrakul P, Wong KW, Fung CC "Classification of imbalanced data by combining the complementary neural network and SMOTE algorithm," In Neural Information Processing. Models and Applications: 17th International Conference, ICONIP 2010, Sydney, Australia, November 22–25, 2010, Proceedings, Part II 17, 2010: Springer, 152–159.
Lusa L Evaluation of smote for high-dimensional class-imbalanced microarray data, In: 2012 11th international conference on machine learning and applications, 2012, vol. 2: IEEE, pp. 89–94.
Slatko BE, Gardner AF, Ausubel FM. Overview of next-generation sequencing technologies. Curr Protoc Mol Biol. 2018;122(1):e59.
Nayak DSK, Das J, Swarnkar T Quality Control Pipeline for Next Generation Sequencing Data Analysis, In Intelligent and Cloud Computing: Proceedings of ICICC 2021: Springer, 2022, pp. 215–225.
Nayak DSK, Mahapatra S, Swarnkar T Gene selection and enrichment for microarray data—a comparative network based approach, In Progress in Advanced Computing and Intelligent Engineering: Proceedings of ICACIE 2016, Volume 2, 2018: Springer, pp. 417–427.
"Python 3.7." https://www.python.org/downloads/release/python-370/. Accessed 03 Apr 2024.
Funding
The authors have received no external funding for this work.
Author information
Authors and Affiliations
Contributions
DSKN, SS, and TS designed the model and the computational framework and analyzed the data. DSKN carried out the simulations and implementation. DSKN, AP, PM, and TD performed the calculations. DSKN wrote the manuscript with input from all authors. DSKN, SS, and TS conceived the study and were in charge of overall direction and planning.
Corresponding author
Ethics declarations
Conflict of interest
The authors have no conflict of interest related to this publication.
Additional information
Publisher's Note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Nayak, D.S.K., Priyadarshini, A., Mahanta, P. et al. DnnARs: An Artificial Intelligence Technique for Prediction of Antimicrobial Resistant Strains in E. coli Bacteria Causing Urine Tract Infection. SN COMPUT. SCI. 5, 1091 (2024). https://doi.org/10.1007/s42979-024-03452-6
Received:
Accepted:
Published:
DOI: https://doi.org/10.1007/s42979-024-03452-6