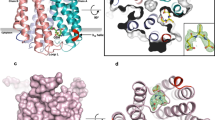
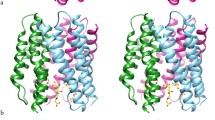
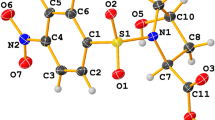
Abstract
Inducible, microsomal prostaglandin E synthase 1 (mPGES-1), the terminal enzyme in the prostaglandin (PG) biosynthetic pathway, constitutes a promising therapeutic target for the development of new anti-inflammatory drugs. To elucidate structure–function relationships and to enable structure-based design, an mPGES-1 homology model was developed using the three-dimensional structure of the closest homologue of the MAPEG family (Membrane Associated Proteins in Eicosanoid and Glutathione metabolism), mGST-1. The ensuing model of mPGES-1 is a homo-trimer, with each monomer consisting of four membrane-spanning segments. Extensive structure refinement revealed an inter-monomer salt bridge (K26-E77) as well as inter-helical interactions within each monomer, including polar hydrogen bonds (e.g. T78-R110-T129) and hydrophobic π-stacking (F82-F103-F106), all contributing to the overall stability of the homo-trimer of mPGES-1. Catalytic co-factor glutathione (GSH) was docked into the mPGES-1 model by flexible optimization of both the ligand and the protein conformations, starting from the initial location ascertained from the mGST-1 structure. Possible binding site for the substrate, prostaglandin H2 (PGH2), was identified by systematically probing the refined molecular structure of mPGES-1. A binding model was generated by induced fit docking of PGH2 in the presence of GSH. The homology model prescribes three potential inhibitor binding sites per mPGES-1 trimer. This was further confirmed experimentally by equilibrium dialysis study which generated a binding stoichiometric ratio of approximately three inhibitor molecules to three mPGES-1 monomers. The structural model that we have derived could serve as a useful tool for structure-guided design of inhibitors for this emergently important therapeutic target.








Similar content being viewed by others
References
Bresell A, Weinander R, Lundqvist G, Raza H, Shimoji M, Sun T-H et al (2005) FEBS J 272:1688. doi:10.1111/j.1742-4658.2005.04596.x
Jakobsson P-J, Thoren S, Morgenstern R, Samuelsson B (1999) Proc Natl Acad Sci USA 96:7220. doi:10.1073/pnas.96.13.7220
Jakobsson P-J, Morgenstern R, Mancini J, Ford-Hutchinson A, Persson B (1999) Protein Sci 8:689
Naraba H, Murakami M, Matsumoto H, Shimbara S, Ueno A, Kudo I et al (1998) J Immunol 160:2974
Pettersson PL, Thoren S, Jakobsson P-J (2005) Methods Enzymol 401:147. doi:10.1016/S0076-6879(05)01009-8
Murakami M, Kudo I (2004) Prog Lipid Res 43:3. doi:10.1016/S0163-7827(03)00037-7
Tanioka T, Nakatani Y, Semmyo N, Murakami M, Kudo I (2000) J Biol Chem 275:32775. doi:10.1074/jbc.M003504200
Zhang Y, Schneider A, Rao R, Lu WJ, Fan X, Davis L et al (2003) Biochim Biophys Acta Mol Cell Biol Lipids 1634:15
Watanabe K, Kurihara K, Tokunaga Y, Hayaishi O (1997) Biochem Biophys Res Commun 235:148. doi:10.1006/bbrc.1997.6708
Tanikawa N, Ohmiya Y, Ohkubo H, Hashimoto K, Kangawa K, Kojima M et al (2002) Biochem Biophys Res Commun 291:884. doi:10.1006/bbrc.2002.6531
Murakami M, Nakashima K, Kamei K, Masuda S, Ishikawa Y, Ishii T et al (2003) J Biol Chem 278:37937. doi:10.1074/jbc.M305108200
Weaver AJ, Sullivan WP, Felts SJ, Owen BA, Toft DO (2000) J Biol Chem 275:23045. doi:10.1074/jbc.M003410200
Kudo I, Murakami M (2005) J Biochem Mol Biol 38:633
Yamada T, Komoto J, Watanabe K, Ohmiya Y, Takusagawa F (2005) J Mol Biol 348:1163. doi:10.1016/j.jmb.2005.03.035
Yamada T, Takusagawa F (2007) Biochemistry 46:8414. doi:10.1021/bi700605m
Thoren S, Weinander R, Saha S, Jegerschold C, Pettersson PL, Samuelsson B et al (2003) J Biol Chem 278:22199. doi:10.1074/jbc.M303227200
Murakami M, Naraba H, Tanioka T, Semmyo N, Nakatani Y, Kojima F et al (2000) J Biol Chem 275:32783. doi:10.1074/jbc.M003505200
Uematsu S, Matsumoto M, Takeda K, Akira S (2002) J Immunol 168:5811
Mabuchi T, Kojima H, Abe T, Takagi K, Sakurai M, Ohmiya Y et al (2004) Neuroreport 15:1395. doi:10.1097/01.wnr.0000129372.89000.31
Kojima F, Naraba H, Miyamoto S, Beppu M, Aoki H, Kawai S (2004) Arthritis Res Ther 6:R355. doi:10.1186/ar1195
Samuelsson B, Morgenstern R, Jakobsson P-J (2007) Pharmacol Rev 59:207. doi:10.1124/pr.59.3.1
Ikeda-Matsuo Y, Ota A, Fukada T, Uematsu S, Akira S, Sasaki Y (2006) Proc Natl Acad Sci USA 103:11790. doi:10.1073/pnas.0604400103
Mnich SJ, Veenhuizen AW, Monahan JB, Sheehan KCF, Lynch KR, Isakson PC et al (1995) J Immunol 155:4437
Kamei D, Yamakawa K, Takegoshi Y, Mikami-Nakanishi M, Nakatani Y, Oh-ishi S et al (2004) J Biol Chem 279:33684. doi:10.1074/jbc.M400199200
Ikeda-Matsuo Y, Ikegaya Y, Matsuki N, Uematsu S, Akira S, Sasaki Y (2005) J Neurochem 94:1546. doi:10.1111/j.1471-4159.2005.03302.x
Inada M, Matsumoto C, Uematsu S, Akira S, Miyaura C (2006) J Immunol 177:1879
Ushikubi F, Segi E, Sugimoto Y, Murata T, Matsuoka T, Kobayashi T et al (1998) Nature 395:281. doi:10.1038/26233
Portanova JP, Zhang Y, Anderson GD, Hauser SD, Masferrer JL, Seibert K et al (1996) J Exp Med 184:883. doi:10.1084/jem.184.3.883
Cheng Y, Austin SC, Rocca B, Koller BH, Coffman TM, Grosser T, Lawson JA, Fitzgerald GA (2002) Science 296:539. doi:10.1126/science.1068711
Murakami M, Kudo I (2006) Curr Pharm Des 12:943. doi:10.2174/138161206776055912
Riendeau D, Aspiotis R, Ethier D, Gareau Y, Grimm EL, Guay J et al (2005) Bioorg Med Chem Lett 15:3352. doi:10.1016/j.bmcl.2005.05.027
Jachak SM (2007) Curr Opin Investig Drugs 8:411
Fahmi H (2004) Curr Opin Rheumatol 16:623. doi:10.1097/01.bor.0000129664.81052.8e
Cheng Y, Wang M, Yu Y, Lawson J, Funk CD, FitzGerald GA (2006) J Clin Invest 116:1391. doi:10.1172/JCI27540
Juteau H, Wu TY-H, Ducharme Y, Friesen RW, Guiral S, Dufresne L et al (2007) Abstracts of papers, 234th ACS national meeting
Mancini JA, Blood K, Guay J, Gordon R, Claveau D, Chan CC (2001) J Biol Chem 276:4469. doi:10.1074/jbc.M006865200
Hauel N, Arndt K, Doods H, Klinder K, Pfau R (2008) PCT Int Appl, WO 2008084218 A1
Côté B, Louise B, Brideau C, Claveau D, Ethier D, Frenette R, Gagnon M, Giroux A, Guay J, Guiral S, Mancini J, Martins E, Massé F, Méthot N, Riendeau D, Rubin J, Xu D, Yu H, Ducharme Y, Friesen RW (2007) Bioorg Med Chem Lett 17:6816. doi:10.1016/j.bmcl.2007.10.033
Ferguson AD, McKeever BM, Xu S, Wisniewski D, Miller DK, Yamin T-T et al (2007) Science 317:510. doi:10.1126/science.1144346
Ago H, Kanaoka Y, Irikura D, Lam BK, Shimamura T, Austen KF et al (2007) Nature 448:609. doi:10.1038/nature05936
Molina DM, Wetterholm A, Kohl A, McCarthy AA, Niegowski D, Ohlson E et al (2007) Nature 448:613. doi:10.1038/nature06009
Holm PJ, Bhakat P, Caroline J, Gyobu N, Mitsuoka K, Fujiyoshi Y et al (2006) J Mol Biol 360:934. doi:10.1016/j.jmb.2006.05.056
Huang X, Yan W, Gao D, Tong M, Tai H-H, Zhan C-G (2006) Bioorg Med Chem 14:3553. doi:10.1016/j.bmc.2006.01.010
AbdulHameed MDM, Hamza A, Liu J, Huang X, Zhan C-G (2008) J Chem Inf Model 48:179. doi:10.1021/ci700315c
Hamza A, AbdulHameed MDM, Zhan C-G (2008) J Phys Chem B 112:7320. doi:10.1021/jp8007688
Schrödinger, LLC, 101 SW Main Street, Suite 1300, Portland, OR 97204; www.schrodinger.com
Sybyl is a product of Tripos, 1699 South Hanley Road, St. Louis, MO 63144; www.tripos.com
Thompson JD, Gibson TJ, Plewniak F, Jeanmougin F, Higgins DG (1997) Nucleic Acids Res 25:4876. doi:10.1093/nar/25.24.4876
Acknowledgements
We thank the scientists at Karolinska Institutet (Dr. H. Hebert, Dr. P.-J. Jakobsson and Dr. R. Morgenstern) for the structural information of mGST-1. The scientists at Schrödinger are acknowledged for their technical supports. The authors are also grateful to Dr. T. Benson for his suggestions on the manuscript.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Xing, L., Kurumbail, R.G., Frazier, R.B. et al. Homo-timeric structural model of human microsomal prostaglandin E synthase-1 and characterization of its substrate/inhibitor binding interactions. J Comput Aided Mol Des 23, 13–24 (2009). https://doi.org/10.1007/s10822-008-9233-4
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-008-9233-4