Abstract
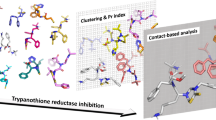
Chagas disease remains a major health problem in South America, and throughout the world. The two drugs clinically available for its treatment have limited efficacy and cause serious adverse effects. Cruzain is an established therapeutic target of Trypanosoma cruzi, the protozoan that causes Chagas disease. Our group recently identified a competitive cruzain inhibitor (compound 1) with an IC50 = 15 µM that is also more synthetically accessible than the previously reported lead, compound 2. Prior studies, however, did not propose a binding mode for compound 1, hindering understanding of the structure–activity relationship and optimization. Here, the cruzain binding mode of compound 1 was investigated using docking, molecular dynamics (MD) simulations with ab initio derived parameters, ab initio calculations, and MM/PBSA. Two ligand protonation states and four binding poses were evaluated. A careful ligand parameterization method was employed to derive more physically meaningful parameters than those obtained by automated tools. The poses of unprotonated 1 were unstable in MD, showing large conformational changes and diffusing away from the binding site, whereas the protonated form showed higher stability and interaction with negatively charged residues Asp161 and Cys25. MM/PBSA also suggested that these two residues contribute favorably to binding of compound 1. By combining results from MD, ab initio calculations, and MM/PBSA, a binding mode of 1 is proposed. The results also provide insights for further optimization of 1, an interesting lead compound for the development of new cruzain inhibitors.








Similar content being viewed by others
References
World Health Organization (2015) Chagas disease in Latin America: an epidemiological update based on 2010 estimates. Wkly Epidemiol Rec 6:33–44. https://doi.org/10.2147/IBPC.S70402
World Health Organization (2013) Sustaining the drive to overcome the global impact of neglected tropical diseases. Second WHO report on neglected tropical diseases, vol 3, no 9. World Health Organization, Geneva, pp 67–71
Morillo CA, Marin-Neto JA, Avezum A et al (2015) Randomized trial of benznidazole for chronic Chagas’ cardiomyopathy. N Engl J Med 373:1295–1306. https://doi.org/10.1056/NEJMoa1507574
das Neves Pinto AY, da Costa Valente V, Coura JR et al (2013) Clinical follow-up of responses to treatment with benznidazol in amazon: a cohort study of acute Chagas disease. PLoS ONE 8:e64450. https://doi.org/10.1371/journal.pone.0064450
Guedes PM, Silva GK, Gutierrez FR, Silva JS (2011) Current status of Chagas disease chemotherapy. Expert Rev Anti Infect Ther 9:609–620. https://doi.org/10.1586/eri.11.31
Carlos Pinto Dias J, Novaes Ramos A, Dias Gontijo E et al (2016) II Consenso Brasileiro em Doença de Chagas, 2015. Epidemiol e Serviços Saúde 25:1–10. https://doi.org/10.5123/S1679-49742016000500002
Keenan M, Chaplin JH (2015) A new era for Chagas disease drug discovery? Progress in medicinal chemistry, 1st edn. Elsevier, Oxford, pp 185–230
Duschak VG, Ciaccio M, Nasser JR, Basombrío MA (2001) Enzymatic activity, protein expressionm, and gene sequence of cruzipain in virulent and attenuated Trypanosoma cruzi strains. J Parasitol 87:1016–1022
McKerrow J, Doyle P, Engel J et al (2009) Two approaches to discovering and developing new drugs for Chagas disease. Mem Inst Oswaldo Cruz 104:263–269. https://doi.org/10.1590/S0074-02762009000900034
Engel JC, Doyle PS, Hsieh I, McKerrow JH (1998) Cysteine protease inhibitors cure an experimental Trypanosoma cruzi infection. J Exp Med 188:725–734
Ferreira RS, Simeonov A, Jadhav A et al (2010) Complementarity between a docking and a high-throughput screen in discovering new cruzain inhibitors. J Med Chem 53:4891–4905. https://doi.org/10.1021/jm100488w
Braga SFP, Martins LC, da Silva EB et al (2017) Synthesis and biological evaluation of potential inhibitors of the cysteine proteases cruzain and rhodesain designed by molecular simplification. Bioorg Med Chem 25:1889–1900. https://doi.org/10.1016/j.bmc.2017.02.009
Cino EA, Choy W-Y, Karttunen M (2013) Conformational biases of linear motifs. J Phys Chem B 117:15943–15957. https://doi.org/10.1021/jp407536p
Cino EA, Killoran RC, Karttunen M, Choy W-Y (2013) Binding of disordered proteins to a protein hub. Sci Rep 3:2305. https://doi.org/10.1038/srep02305
Kuhn B, Tichý M, Wang L et al (2017) Prospective evaluation of free energy calculations for the prioritization of Cathepsin L inhibitors. J Med Chem 60:2485–2497. https://doi.org/10.1021/acs.jmedchem.6b01881
Matter H, Sotriffer C (2011) Applications and success stories in virtual screening. In: Sotriffer C (ed) Virtual screening: principles, challenges, and practical guidelines. Wiley, New York, pp 319–358
De Vivo M, Masetti M, Bottegoni G, Cavalli A (2016) Role of molecular dynamics and related methods in drug discovery. J Med Chem 59:4035–4061. https://doi.org/10.1021/acs.jmedchem.5b01684
Wang L, Wu Y, Deng Y et al (2015) Accurate and reliable prediction of relative ligand binding potency in prospective drug discovery by way of a modern free-energy calculation protocol and force field. J Am Chem Soc 137:2695–2703. https://doi.org/10.1021/ja512751q
Gordon MS, Schmidt MW (2005) Advances in electronic structure theory. In: Dykstra CE, Frenking G, Kim KS, Scuseria GE (eds) Theory and applications of computational chemistry. Elsevier, Amsterdam, pp 1167–1189
Janssen CL, Nielsen IB, Leininger ML et al (2008) The massively parallel quantum chemistry program (MPQC). Sandia National Laboratories, Livermore
Thapa B, Schlegel HB (2016) Density functional theory calculation of pKa’s of thiols in aqueous solution using explicit water molecules and the polarizable continuum model. J Phys Chem A 120:5726–5735. https://doi.org/10.1021/acs.jpca.6b05040
Thapa B, Schlegel HB (2015) Calculations of pKa’s and redox potentials of nucleobases with explicit waters and polarizable continuum solvation. J Phys Chem A 119:5134–5144. https://doi.org/10.1021/jp5088866
Linstrom PJ, Mallard WG (2014) NIST chemistry webBook, NIST standard reference database number 69. National Institute of Standards and Technology 20899: doi: citeulike-article-id:3211271
Camaioni DM, Schwerdtfeger CA (2005) Comment on “Accurate experimental values for the free energies of hydration of H+, OH-, and H3O+”. J Phys Chem A 109:10795–10797. https://doi.org/10.1021/jp054088k
Welch BL (1947) The generalization of “Student”s’ problem when several different population variances are involved. Biometrika 34:28–35. https://doi.org/10.1093/biomet/34.1-2.28
Efron B, Tibshirani RJ (1993) An introduction to the bootstrap. Springer, Boston
Mann HB, Whitney DR (1947) On a test of whether one of two random variables is stochastically larger than the other. Ann Math Stat 18:50–60. https://doi.org/10.1214/aoms/1177730491
Wilcoxon F (1947) Probability tables for individual comparisons by ranking methods. Biometrics 3:119. https://doi.org/10.2307/3001946
Kruskal WH, Wallis WA (1952) Use of ranks in one-criterion variance analysis. J Am Stat Assoc 47:583–621. https://doi.org/10.1080/01621459.1952.10483441
Vanquelef E, Simon S, Marquant G et al (2011) R.E.D. Server: a web service for deriving RESP and ESP charges and building force field libraries for new molecules and molecular fragments. Nucleic Acids Res 39:W511–W517. https://doi.org/10.1093/nar/gkr288
Sousa da Silva AW, Vranken WF (2012) ACPYPE—antechamber python parser interface. BMC Res Notes 5:367. https://doi.org/10.1186/1756-0500-5-367
Pappu RV, Hart RK, Ponder JW (1988) Tinker: a package for molecular dynamics simulation. J Phys Chem B 102:9725–9742
Schlee D, Sneath PHA, Sokal RR, Freeman WH (1975) Numerical taxonomy. The principles and practice of numerical classification. Syst Zool 24:263. https://doi.org/10.2307/2412767
O’Boyle NM, Banck M, James C et al (2011) Open babel: an open chemical toolbox. J Cheminform 3:33. https://doi.org/10.1186/1758-2946-3-33
Connolly ML (1983) Analytical molecular surface calculation. J Appl Crystallogr 16:548–558. https://doi.org/10.1107/S0021889883010985
Dupradeau F-Y, Pigache A, Zaffran T et al (2010) The R.E.D. tools: advances in RESP and ESP charge derivation and force field library building. Phys Chem Chem Phys 12:7821. https://doi.org/10.1039/c0cp00111b
Morris GM, Huey R, Lindstrom W et al (2009) AutoDock4 and AutoDockTools4: automated docking with selective receptor flexibility. J Comput Chem 30:2785–2791. https://doi.org/10.1002/jcc.21256
Pronk S, Páll S, Schulz R et al (2013) GROMACS 4.5: a high-throughput and highly parallel open source molecular simulation toolkit. Bioinformatics 29:845–854. https://doi.org/10.1093/bioinformatics/btt055
Jorgensen WL, Chandrasekhar J, Madura JD et al (1983) Comparison of simple potential functions for simulating liquid water. J Chem Phys 79:926–935. https://doi.org/10.1063/1.445869
Shevchuk R, Prada-Gracia D, Rao F (2012) Water structure-forming capabilities are temperature shifted for different models. J Phys Chem B 116:7538–7543. https://doi.org/10.1021/jp303583f
Darden T, York D, Pedersen L (1993) Particle mesh Ewald: An N⋅log(N) method for Ewald sums in large systems. J Chem Phys 98:10089–10092. https://doi.org/10.1063/1.464397
Olsson MHM, Søndergaard CR, Rostkowski M, Jensen JH (2011) PROPKA3: consistent treatment of internal and surface residues in empirical pKa predictions. J Chem Theory Comput 7:525–537. https://doi.org/10.1021/ct100578z
Dolinsky TJ, Nielsen JE, McCammon JA, Baker NA (2004) PDB2PQR: an automated pipeline for the setup of Poisson-Boltzmann electrostatics calculations. Nucleic Acids Res 32:W665–W667. https://doi.org/10.1093/nar/gkh381
Lindorff-Larsen K, Piana S, Palmo K et al (2010) Improved side-chain torsion potentials for the Amber ff99SB protein force field. Proteins. https://doi.org/10.1002/prot.22711
Kumari R, Kumar R, Lynn A (2014) g_mmpbsa—a GROMACS tool for high-throughput MM-PBSA calculations. J Chem Inf Model 54:1951–1962. https://doi.org/10.1021/ci500020m
Gangarapu S, Marcelis ATM, Zuilhof H (2013) Accurate pKa calculation of the conjugate acids of alkanolamines, alkaloids and nucleotide bases by quantum chemical methods. ChemPhysChem 14:990–995. https://doi.org/10.1002/cphc.201201085
Rebollar-Zepeda AM, Galano A (2012) First principles calculations of pK a values of amines in aqueous solution: application to neurotransmitters. Int J Quantum Chem 112:3449–3460. https://doi.org/10.1002/qua.24048
Casasnovas R, Fernández D, Ortega-Castro J et al (2011) Avoiding gas-phase calculations in theoretical pKa predictions. Theor Chem Acc 130:1–13. https://doi.org/10.1007/s00214-011-0945-5
Albert A, Goldacre R, Phillips J (1948) 455. The strength of heterocyclic bases. J Chem Soc. https://doi.org/10.1039/jr9480002240
Warhurst DC (2003) Hydroxychloroquine is much less active than chloroquine against chloroquine-resistant Plasmodium falciparum, in agreement with its physicochemical properties. J Antimicrob Chemother 52:188–193. https://doi.org/10.1093/jac/dkg319
Lundborg M, Lindahl E (2015) Automatic GROMACS topology generation and comparisons of force fields for solvation free energy calculations. J Phys Chem B 119:810–823. https://doi.org/10.1021/jp505332p
Karttunen M, Rottler J, Vattulainen I, Sagui C (2008) Chap. 2 Electrostatics in biomolecular simulations: where are we now and where are we heading? Current Topics in Membranes. Elsevier Inc, pp 49–89
Reynolds CA, Essex JW, Richards WG (1992) Atomic charges for variable molecular conformations. J Am Chem Soc 114:9075–9079. https://doi.org/10.1021/ja00049a045
Cornell WD, Cieplak P, Bayly CI et al (1995) A second generation force field for the simulation of proteins, nucleic acids, and organic molecules. J Am Chem Soc 117:5179–5197. https://doi.org/10.1021/ja00124a002
Basma M, Sundara S, Çalgan D et al (2001) Solvated ensemble averaging in the calculation of partial atomic charges. J Comput Chem 22:1125–1137. https://doi.org/10.1002/jcc.1072
Gillmor S, Craik CS, Fletterick RJ (1997) Structural determinants of specificity in the cysteine protease cruzain. Protein Sci 6:1603–1611. https://doi.org/10.1002/pro.5560060801
Kerr ID, Lee JH, Farady CJ et al (2009) Vinyl sulfones as antiparasitic agents and a structural basis for drug design. J Biol Chem 284:25697–25703. https://doi.org/10.1074/jbc.M109.014340
Schrödinger LLC (2016) The PyMOL molecular graphics system. Schrödinger LLC version 1. http://www.pymol.org
McGrath ME, Eakin AE, Engel JC et al (1995) The crystal structure of cruzain: a therapeutic target for Chagas’ disease. J Mol Biol 247:251–259. https://doi.org/10.1006/jmbi.1994.0137
Chen YT, Brinen LS, Kerr ID et al (2010) In vitro and in vivo studies of the trypanocidal properties of WRR-483 against Trypanosoma cruzi. PLoS Negl Trop Dis 4:e825. https://doi.org/10.1371/journal.pntd.0000825
Brinen LS, Hansell E, Cheng J et al (2000) A target within the target: probing cruzain’s P1′ site to define structural determinants for the Chagas’ disease protease. Structure 8:831–840. https://doi.org/10.1016/S0969-2126(00)00173-8
Gillmor SA (1998) Chapter 3: X-ray structures of complexes of cruzain with designed covalent inhibitors. Enzyme-ligand interactions, inhibition and specificity, pp 50–80
Genheden S, Ryde U (2015) The MM/PBSA and MM/GBSA methods to estimate ligand-binding affinities. Expert Opin Drug Discov 10:449–461. https://doi.org/10.1517/17460441.2015.1032936
Thompson DC, Humblet C, Joseph-McCarthy D (2008) Investigation of MM-PBSA rescoring of docking poses. J Chem Inf Model 48:1081–1091. https://doi.org/10.1021/ci700470c
Negri M, Recanatini M, Hartmann RW (2011) Computational investigation of the binding mode of bis(hydroxylphenyl)arenes in 17β-HSD1: molecular dynamics simulations, MM-PBSA free energy calculations, and molecular electrostatic potential maps. J Comput Aided Mol Des 25:795–811. https://doi.org/10.1007/s10822-011-9464-7
Sun H, Li Y, Shen M et al (2014) Assessing the performance of MM/PBSA and MM/GBSA methods. 5. Improved docking performance using high solute dielectric constant MM/GBSA and MM/PBSA rescoring. Phys Chem Chem Phys 16:22035–22045. https://doi.org/10.1039/C4CP03179B
Lu M-C, Yuan Z-W, Jiang Y-L et al (2016) A systematic molecular dynamics approach to the study of peptide Keap1–Nrf2 protein–protein interaction inhibitors and its application to p62 peptides. Mol BioSyst 12:1378–1387. https://doi.org/10.1039/C6MB00030D
Aldeghi M, Bodkin MJ, Knapp S, Biggin PC (2017) Statistical analysis on the performance of molecular mechanics Poisson-Boltzmann surface area versus absolute binding free energy calculations: bromodomains as a case study. J Chem Inf Model 57:2203–2221. https://doi.org/10.1021/acs.jcim.7b00347
Acknowledgements
The authors would like to thank Brazilian funding agencies Conselho Nacional do Desenvolvimento Científico e Tecnológico, Coordenação de Aperfeiçoamento de Pessoal de Nível Superior, Fundação de Amparo à Pesquisa do Estado de Minas Gerais and Fundação Carlos Chagas Filho de Amparo à Pesquisa do Estado do Rio de Janeiro. RSF, RBO and PGP hold CNPq research fellowships (Bolsa de Produtividade em Pesquisa). This research was enabled in part by support provided by Compute Canada (http://www.computecanada.ca).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Conflict of interest
The authors declare that they have no conflict of interest.
Electronic supplementary material
Below is the link to the electronic supplementary material.
Rights and permissions
About this article
Cite this article
Martins, L.C., Torres, P.H.M., de Oliveira, R.B. et al. Investigation of the binding mode of a novel cruzain inhibitor by docking, molecular dynamics, ab initio and MM/PBSA calculations. J Comput Aided Mol Des 32, 591–605 (2018). https://doi.org/10.1007/s10822-018-0112-3
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1007/s10822-018-0112-3