Summary
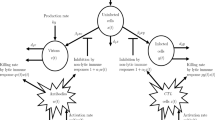
A continuous harvest effort can lead a population to extinction. How an “unconscious” immune system would perpetrate such an effort in order to eliminate a self-replicating antigen (a pathogen) becomes an intriguing problem if the system responses are functions of the pathogen population: the responses cannot be a continuous effort as the pathogen vanishes. On theoretical grounds, we show some qualities an immune response must have to support pathogen elimination. Then, three specific mechanisms are addressed: a pathogen-independent positive feedback loop among the responding cells of the system (e.g., B-lymphocyte and T-helper); the persistence of antigen bound to presenting cells; and the programmed expansion/contraction of a pool of responding cells. The maintenance of responding cells due to these mechanisms is the essential feature to the effective clearance of self-replicating agents. Thus, evolutionarily, the primary function of a helper lymphocyte would be to amplify a response and the primary function of memory would be the very elimination of pathogens.
Similar content being viewed by others
References
Antia, R., Koella, J., 1994. A model of non-specific immunity. J. Theor. Biol. 168, 141–150.
Bachmann, M.F., Kundig, T.M., Hengartner, H., Zinkernagel, R.M., 1997. Protection against immunopathological consequences of a viral infection by activated but not resting cytotoxic T cells: T cell memory without “memory T cells”? Proc. Natl. Acad. Sci. USA 94, 640–645.
Badovinac, V.P., Tvinnereim, A., Harty, J.T., 2000. Regulation of antigen-specific CD8+T cell homeostasis by perforin and interferon-γ. Science 290, 1354–1357.
Bevan, M.J., Goldrath, A.W., 2000. T-cell memory: you must remember this. Curr. Biol. 10, R338-R340.
Bocharov, G., Romanyukha, A., 1994. Mathematical model of antiviral immune response III. Influenza A virus infection. J. Theor. Biol. 167, 323–360.
Boman, H.G., 2000. Innate immunity and the normal microflora. Immunol. Rev. 173, 5–16.
Bunce, C., Bell, E.B., 1997. CD45RC isoforms define two types of CD4 memory T cells, one of which depends on persisting antigen. J. Exp. Med. 185, 767–776.
Busch, D.H., Pilip, I.M., Vijh, S., Pamer, E.G., 1998. Coordinate regulation of complex T cell populations responding to bacterial infection. Immunity 8, 353–362.
Chowdhury, D., 1993. A unified model of immune response II: continuum approach. J. Theor. Biol. 165, 135–159.
Ciurea, A., Hunziker, L., Klenerman, P., Hengartner, H., Zinkernagel, R.M., 2001. Impairment of CD4+T cell responses during chronic virus infection prevents neutralizing antibody responses against virus escape mutants. J. Exp. Med. 193, 297–305.
De Boer, R., Perelson, A., 1995. Towards a general function describing T cell proliferation. J. Theor. Biol. 175, 567–576.
Dibrov, B.F., Livshits, M., Volkenstein, M.V., 1977. Mathematical model of immune processes. J. Theor. Biol. 65, 609–631.
Facciotti, M.C., Schmideil, W., 1995. The new concept of minimum cell viability and its consequences on bioprocess design and operation. Braz. J. Chem. Eng. 12, 22–31.
Ferenci, T., 1999. Growth of bacterial cultures 50 years on: towards an uncertainty principle instead of contants in bacterial growth kinetics. Res. Microbiol. 150, 431–438.
Fishman, M., Perelson, A., 1994. Th1/Th2 cross regulation. J. Theor. Biol. 170, 25–56.
Fishman, M., Perelson, A., 1999. Th1/Th2 differentiation and cross-regulation. Bull. Mater. Biol. 61, 403–436.
Hogan, R.J., Usherwood, E.J., Zhong, W., Roberts, A., Dutton, R.W., Harmsen, A.G., Woodland, D.L. 2001a. Activated antigen-specific CD8+T cells persist in the lungs following recovery from respiratory virus infections. J. Immunol. 166, 1813–1822.
Hogan, R.J., Zhong, W., Usherwood, E.J., Cookenham, T., Roberts, A., Woodland D.L., 2001b. Protection from respiratory virus infections can be mediated by antigen-specific CD4(+) T cells that persist in the lungs. J. Exp. Med. 193, 981–986.
Hou, S., Hyland, L., Ryan, K.W., Portner, A., Doherty, P.C., 1994. Virus-specific CD8+T-cell memory determined by clonal burst size. Nature 369, 652–654.
Hsu, E., 1998. Mutation, selection and memory in B lymphocytes of exothermic vertebrates. Immunol. Res. 162, 25–36.
Kaufman, M., Thomas, R., 1987. Model analysis of the bases of multistationarity in the humoral immune response. J. Theor. Biol. 129, 141–162.
Kaufman, M., Urbain, J., Thomas, R., 1985. Towards a logical analysis of the immune-response. J. Theor. Biol. 114, 527–561.
Koverova-Kovar, K., Egli, T., 1998. Growth kinetics of suspended microbial cells: from single-substrate-controlled growth to mixed-substrate kinetics. Microbiol. Mol. Biol. Rev. 62, 646–666.
Langman, R.E., 1989. The Immune System: Evolutionary Principles Guide our Understanding of this Complex Biological Defense System. Academic Press, San Diego.
Le Morvan, C., Troutaud, D., Deschaux, P., 1998. Differential effects of temperature on specific and nonspecific immune defenses in fish. J. Exp. Biol. 201, 165–168.
Marshall, D., Turner, S., Belz, G., Wingo, S., Andreansky, S., Sangster, M., Riberdy, J., Liu, T., Tan, M., Doherty, P., 2001. Measuring the diaspora for virus-specific CD8+T cells. Proc. Natl. Acad. Sci. USA 98, 6313–6318.
Maruyama, M., Lam, K.P., Rajewsky, K., 2000. Memory B-cell persistence is independent of persisting immunizing antigen. Nature 407, 636–642.
McLean, A., 1994. Modelling T cell memory. J. Theor. Biol. 170, 63–74.
Mercado, R., Vijh, S., Allen, S.E., Kerksiek, K., Pilip, I.M., Pamer, E.G., 2000. Early programming of T cell populations responding to bacterial infection. J. Immunol. 165, 6833–6839.
Monteiro, L.H.A., 2002. Sistemas Dinâmicos. Editora-Livraria da Física, São Paulo.
Murray, J.D., 1993. Mathematical Biology, 2nd Ed., Springer, Berlin.
Strogatz, S. H., 1994. Nonlinear Dynamics and Chaos. Addison-Wesley, Reading.
Thomsen, A.R., Nansen, A., Andreasen, S.Ø., Wodraz, D., Christensen, J.P., 2000. Host factors influencing viral persistence. Phil. Trans. R. Soc. London series B 355, 1031–1041.
Wodarz, D., 2001. Cytotoxic T-lymphocyte memory, virus clearance and antigenic heterogeneity. Proc. R. Soc. London series B 268, 429–436.
Wodarz, D., May, R.M., Nowak, M.A., 2000. The role of antigen-independent persistence of memory cytotoxic T lymphocytes. Int. Immunol. 12, 467–477.
Wodarz, D., Jansen, V.A.A., 2001. The role of T cell help for anti-viral CTL responses. J. Theor. Biol. 211, 419–432.
Zinkernagel, R.M., Hengartner, H., 2001 Regulation of the immune response by antigen. Science 293, 251–253.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Chaui-Berlinck, J.G., Barbuto, J.A.M. & Monteiro, L.H.A. Conditions for pathogen elimination by immune systems. Theory Biosci. 123, 195–208 (2004). https://doi.org/10.1016/j.thbio.2004.01.001
Issue Date:
DOI: https://doi.org/10.1016/j.thbio.2004.01.001