Abstract
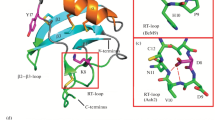
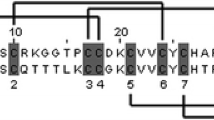
In the present paper, a study of classical and insect α-scorpion toxins is described. A homology model of the classical α-toxin LqqV from Leiurus quinquestriatus quinquestriatus was developed. The model was compared to stable and energetically favourable conformations of AaHII from Androctonus australis Hector and LqhαIT from Leiurus quinquestriatus hebraeus, which are the most active α-toxins in mammals and insects. The conformations were retrieved from molecular dynamics simulations of known structures. The model of LqqV shows a C-terminal conformation similar to LqhαIT. This is mainly caused by electrostatic interactions between Lys10/Lys60 and Glu59, which are comparable to the cation-π interactions of Tyr10 and Arg64 in LqhαIT. During the simulations the structures of AaHII and LqqV were stabilised through electrostatic interactions between Glu32 and Lys50 and especially the loop adjacent to the α-helix is affected, which is in contrast to LqhαIT. When the molecular electrostatic potentials of the toxins were studied, a possibly important difference between the classical α-toxins and the insect α-toxin LqhαIT was found in the area around Lys30 and Arg56 of AaHII, where a positive potential is missing in LqhαIT. A large negative potential caused by Asp3, Glu15 and Asp19 in LqhαIT is also unique for this toxin. It is proposed that Arg18, which is important for activity of LqhαIT, restricts the negative potential in this area and is not essential for toxins where negatively charged residues in comparable positions are not present.
Abbreviations: AaHI-III, α-toxins I, II, III from Androctonus australis Hector; LqqV, α-toxin V from Leiurus quinquestriatus quinquestriatus; LqhαIT, insect α-toxin from Leiurus quinquestriatus hebraeus; BotIII from Buthus occitanus tunetanus; BomIII fromButhus occitanus mardochei; LqhII and LqhIII from Leiurus quinquestriatus hebraeus.
Similar content being viewed by others
References
Possani, L.D., Becerril, B., Delepierre, M. and Tytgat, J., Eur. J. Biochem., 264 (1999) 287.
Gordon, D., Savarin, P., Gurevitz, M. and Zinn-justin, S., Toxicol. Toxin. Rev., 17 (1998) 131.
Rogers, J.C., Qu, Y., Tanada, T.N., Scheuer, T. and Catterall, W.A., J. Biol. Chem., 271 (1996) 15950.
Cestele, S., Qu, Y., Rogers, J.C., Rochat, H., Scheuer, T. and Catterall, W.A., Neuron, 21 (1998) 919.
Gordon, D., Martin-eauclaire, M.F., Cestele, S., Kopeyan, C., Carlier, E., Khalifa, R.B., Pelhate, M. and Rochat, H., J. Biol. Chem., 271 (1996) 8034.
Sautiere, P., Cestele, S., Kopeyan, C., Martinage, A., Drobecq, H., Doljansky, Y. and Gordon, D., Toxicon, 36 (1998) 1141.
Kharrat, R., Darbon, H., Rochat, H. and Granier, C., Eur. J. Biochem., 181 (1989) 381.
Kharrat, R., Darbon, H., Granier, C. and Rochat, H., Toxicon, 28 (1990) 509.
Darbon, H., Jover, E., Couraud, F. and Rochat, H., Int. J. Pept. Protein Res., 22 (1983) 179.
El Ayeb, M., Darbon, H., Bahraoui, E.M., Vargas, O. and Rochat, H., Eur. J. Biochem., 155 (1986) 289.
Zilberberg, N., Froy, O., Loret, E., Cestèle, S., Arad, D., Gordon, D. and Gurevitz, M., J. Biol. Chem., 272 (1997) 4810.
Gilles, N., Krimm, I., Bouet, F., Froy, O., Gurevitz, M., Lancelin, J.M. and Gordon, D., J. Neurochem., 75 (2000) 1735.
Clackson, T. and Wells, J.A., Science, 267 (1995) 383.
Bogan, A.A. and Thorn, K.S., J. Mol. Biol., 280 (1998) 1.
Cunningham, B.C. and Wells, J.A., J. Mol. Biol., 234 (1993) 554.
Sheinerman, F.B., Norel, R. and Honig, B., Curr. Opin. Struct. Biol., 10 (2000) 153.
Bermann, H.M., Westbrook, J., Feng, Z., Gilliland, G., Bhat, T.N., Weissig, H., Shindyalov, I.N. and Bourne, P.E., Nucleic Acids Res., 28 (2000) 235. (http://www.rcsb.org/pdb/)
Smith, G.D., Blessing, R.H., Ealick, S.E., Fontecilla-camps, J.C., Hauptman, H.A., Housset, D. and Langs, D.A., Acta Crystallogr., D53 (1997) 551.
Tugarinov, V., Kustanovich, I., Zilberberg, N., Gurevitz, M. and Anglister, J., Biochemistry, 36 (1997) 2414.
Menez, A., Toxicon, 36 (1998) 1557.
Philippopoulos, M. and Lim, C., Proteins, 36 (1999) 87.
Catterall, W.A., Neuron, 26 (2000) 13.
Bezanilla, F., Physiol. Rev., 80 (2000) 555.
Jiang, Y., Lee, A., Chen, J., Ruta, V., Cadene, M., Chait, B.T. and Mackinnon, R., Nature, 423 (2003) 33.
Jiang, Y., Ruta, V., Chen, J., Lee, A. and Mackinnon, R., Nature, 423 (2003) 42.
Schweitz, H., Vincent, J.P., Barhanin, J., Frelin, C., Linden, G., Hugues, M. and Lazdunski, M., Biochemistry, 20 (1981) 5245.
Khera, P.K., Benzinger, G.R., Lipkind, G., Drum, C.L., Hanck, D.A. and Blumenthal, K.M., Biochemistry, 34 (1995) 8533.
Li, H.M., Wang, D.C., Zeng, Z.H., Jin, J. and Hu, R.Q., J. Mol. Biol., 261 (1996) 415.
Housset, D., Habersetzer-rochat, C., Astier, J.P. and Fontecilla-camps, J.C., J. Mol. Biol., 238 (1994) 88.
INSIGHTII/DISCOVER/HOMOLOGY/DELPHI, Biosym Technologies, Inc./Molecular Simulations (MSI), San Diego, CA (Version 98.0 and 2000).
Laskowski, R.A., Macarthur, M.W., Moss, D.S. and Thornton, J.M., J. Appl. Crystallogr., 26 (1993) 283.
Kelley, L.A., Gardner, S.P. and Sutcliffe, M.J., Protein Eng., 10 (1997) 737.
Kelley, L.A., Gardner, S.P. and Sutcliffe, M.J., Protein Eng., 9 (1996) 1063.
Thompson, J.D., Higgins, D.G. and Gibson, T.J., Nucleic Acids Res., 22 (1994) 4673. (http://www2.ebi.ac.uk/clustalw)
Bairoch, A. and Boeckmann, B., Nucleic Acids Res., 19 Suppl (1991) 2247. (http://www.expasy.ch/sprot/sptot-top.html)
Landon, C., Cornet, B., Bonmatin, J.M., Kopeyan, C., Rochat, H., Vovelle, F. and Ptak, M., Eur. J. Biochem., 236 (1996) 395.
Krimm, I., Gilles, N., Sautiere, P., Stankiewicz, M., Pelhate, M., Gordon, D. and Lancelin, J.M., J. Mol. Biol., 285 (1999) 1749.
Author information
Authors and Affiliations
Rights and permissions
About this article
Cite this article
Bendels, S., Höltje, HD. Comparison of a 3D-model of the classical α-scorpion toxin V from Leiurus quinquestriatus quinquestriatus with other scorpion toxins. J Comput Aided Mol Des 18, 119–133 (2004). https://doi.org/10.1023/B:jcam.0000030035.00229.eb
Issue Date:
DOI: https://doi.org/10.1023/B:jcam.0000030035.00229.eb